SECURITIES AND EXCHANGE COMMISSION
Proxy Statement Pursuant to Section 14(a) of the Securities
Exchange Act of 1934 (Amendment No. )
Filed by the Registrant☒x
Filed by a Party other than the Registrant☐o
Check the appropriate box:
|
| |
o☒ | | Preliminary Proxy Statement |
o☐ | | Confidential, for Use of the Commission Only (as permitted by Rule14a-6(e)(2)) |
x☐ | | Definitive Proxy Statement |
o☐ | | Definitive Additional Materials |
o☐ | | Soliciting Material Pursuant to §240.14a-12§240.14a-12 |

EXELIXIS, INC.
(NameExact name of the Registrantregistrant as Specified In Its Charter)
specified in its charter)(Name of Person(s) Filing Proxy Statement, if other than the Registrant)
Payment of Filing Fee (Check the appropriate box):
|
| | | | | |
x☒ | | | No fee required. |
o☐ | | | Fee computed on table below per Exchange Act Rules14a-6(i)(1) and0-11. |
| | |
(1) | | | (1 | ) | | Title of each class of securities to which transaction applies: |
| |
(2) | (2 | ) | | Aggregate number of securities to which transaction applies: |
| |
(3) | | (3 | ) | | Per unit price or other underlying value of transaction computed pursuant to Exchange ActRule 0-11 (set forth the amount on which the filing fee is calculated and state how it was determined): |
| |
(4) | (4 | ) | | Proposed maximum aggregate value of transaction: |
|
☐ | |
o | Fee paid previously with preliminary materials. |
|
☐ | |
o |
| Check box if any part of the fee is offset as provided by Exchange Act Rule0-11(a)(2) and identify the
filing for which the offsetting fee was paid previously. Identify the previous filing by registration
statement number, or the Form or Schedule and the date of its filing. |
| | |
(1) | | | (1 | ) | | Amount Previously Paid: |
| �� |
(2) | (2 | ) | | Form, Schedule or Registration Statement No.: |

DEAR FELLOW STOCKHOLDERS,
Whether you are a recent investor or have been with Exelixis for a longer portion of our25-year history, thanks for your confidence in our company. With your support, Exelixis made important progress on our mission to help cancer patients recover stronger and live longer in 2018, our second consecutive year of profitability on an operating basis. Our performance was made possible by increased U.S. uptake of our lead medicine, CABOMETYX®, as well as strong growth in collaboration revenues from our partners advancing our products – COMETRIQ®, CABOMETYX®, COTELLIC®, and now MINNEBRO™ – around the world. We are proud that we continued to maintain our profitability even as we made extensive investments in internal drug discovery, productin-licensing, clinical development, and corporate infrastructure that will support our growth into the future.
As stewards of Exelixis’ business, the Board of Directors recognizes that as the company grows, so do the performance expectations and responsibilities imposed on its leadership. We have, therefore, worked with the management team to conduct a thorough review of our governance and compensation practices to ensure that they are appropriate for a company of Exelixis’ scale and growth potential. In the course of this review, we sought to obtain critical input and perspective from our stockholders. These engagement efforts resulted in the following significant governance and compensation enhancements, including:
Declassification of the Board of Directors through amendment of our certificate of incorporation – if this change is approved by the stockholders, the Board of Directors will be declassified and all directors will stand for election annually, beginning at the 2020 Annual Meeting of Stockholders;
Adoption of a more objective framework to guide the evaluation of annual executive bonuses;
Adoption of a Policy for Recoupment of Variable Compensation (also known as a clawback policy);
Incorporation of individual and corporate performance components into the long-term incentive compensation program; and
Adoption of stock ownership guidelines for executives and members of the Board of Directors.
We hope you will consider these enhancements favorably, as well as the additional updates and proposals in this Proxy Statement, as you cast your votes at the 2019 Annual Meeting of Stockholders, which will be held at our headquarters located at 1851 Harbor Bay Parkway, Alameda, California 94502 on May 22, 2019, beginning at 8 a.m. Pacific Time. The following notice of our Annual Meeting contains details of the business to be conducted at the Annual Meeting. Only stockholders of record (or beneficial owners who have obtained a proxy from their broker) at the close of business on March 25, 2019, will be entitled to notice of, and to vote at, the Annual Meeting.
This coming November, Exelixis will mark the 25th anniversary of its founding. Now based at our new campus in Alameda, we have come a long way from our early days as a model systems genetics company spun out of academia and based in Cambridge, Massachusetts. We owe our successes to smart science, effective collaboration, and a resilient culture and team that has withstood the clinical and business challenges that come with our industry—and emerged stronger and better for them. As we move through 2019, we are excited about and grateful for the continued opportunity to introduce medicines that can give patients and their families hope for the future. As we continue in our efforts, you have our commitment that we will remain engaged and aligned with all of our stakeholders.
Very truly yours,
| | | | |
Stelios Papadopoulos, PhD Chairman of the Board | | Charles Cohen, PhD Chairman of the Compensation Committee | | |
210 East Grand Ave.
South San Francisco, CA 94080
|
| |
NOTICE OF ANNUAL MEETING OF STOCKHOLDERS
TO BE HELD ON MAY 25, 2016
PRELIMINARY COPY | |  |
1851 Harbor Bay Parkway
Alameda, CA 94502
NOTICEOF ANNUAL MEETING
OF STOCKHOLDERS
To be held on MAY 22, 2019
To the Stockholders of Exelixis, Inc.:
NOTICE IS HEREBY GIVEN that the Annual Meeting of Stockholders of Exelixis, Inc., a Delaware corporation (“Exelixis”)(Exelixis), will be held on Wednesday, May 25, 2016,22, 2019, at 8:00 a.m., local time, at Exelixis’ offices located at 210 East Grand Avenue, South San Francisco,1851 Harbor Bay Parkway, Alameda, California 9408094502 for the following purposes:
|
| |
| 1. | To elect the threefive Class II nominees for director named in the Proxy Statement accompanying this Notice to hold office until the 20192022 Annual Meeting of Stockholders. |
| 2. | To ratify the selection by the Audit Committee of the Board of Directors of Ernst & Young LLP as Exelixis’ independent registered public accounting firm for the fiscal year ending December 30, 2016.January 3, 2020. |
| 3. | To approve an amendmentthe proposal of Exelixis’ Board of Directors to amend Exelixis’ Amended and restatementRestated Certificate of Incorporation to declassify the Exelixis, Inc. 2000 Employee Stock Purchase Plan (the “2000 Purchase Plan”)Board of Directors to increaseprovide for annual elections by the number2020 Annual Meeting of shares of common stock reserved for issuance under the 2000 Purchase Plan by 5,000,000 shares and make certain other changes as described in Proposal 3. A copy of the 2000 Purchase Plan, as amended, is attached to the Proxy Statement accompanying this Notice as Appendix A.Stockholders. |
| 4. | To approve, on an advisory basis, the compensation of Exelixis’ named executive officers, as disclosed in the Proxy Statement accompanying this Notice. |
| 5. | To conduct any other business properly brought before the meeting. |
These items of business are more fully described in the Proxy Statement accompanying this Notice.
We are mailing to most of our stockholders a Notice of Internet Availability of Proxy Materials (the “Notice”)Notice) instead of a paper copy of this Proxy Statement and our 20152018 Annual Report. The Notice contains instructions on how to access those documents over the Internet. The Notice also contains instructions on how to request a paper copy of our proxy materials, including this Proxy Statement, our 20152018 Annual Report and a form of proxy card or voting instruction card. All stockholders who do not receive a Notice will receive a paper copy of the proxy materials by mail. We believe that this process will allow us to provide our stockholders with the information they need in a more timely manner, while reducing the environmental impact and lowering the costs of printing and distributing our proxy materials.
The record date for the Annual Meeting is March 31, 2016.25, 2019. Only stockholders of record at the close of business on that date may vote at the meeting or any postponement or adjournment thereof.
|
|
Important notice regarding the availability of proxy materials for the Annual Meeting of Stockholders to be held on May 25, 2016,22, 2019, at 8:00 a.m., local time, at Exelixis’ offices located at 210 East Grand Avenue, South San Francisco,1851 Harbor Bay Parkway, Alameda, CA 94080. 94502.The Proxy Statement and 2018 Annual Report to stockholders are available
at www.exel-annualstockholdermeeting.com. www.exel-annualstockholdermeeting.com.
The Board of Directors recommends that you vote “FOR” Proposal Nos.1-4 identified above. |
By Order of the Board of Directors
JEFFREY J. HESSEKIEL
Executive Vice President and General Counsel
Alameda, California
April , 2019
YOUR VOTE IS IMPORTANT
WHETHER OR NOT YOU PLAN TO ATTEND THE 2019 ANNUAL MEETING OF STOCKHOLDERS, TO ENSURE THAT YOU ARE REPRESENTED AT THE MEETING AND TO ENSURE THAT A QUORUM IS PRESENT, WE URGE YOU TO VOTE YOUR PROXY ONLINE, BY TELEPHONE OR BY RETURNING A PROXY CARD BY MAIL AS INSTRUCTED IN THE NOTICE OF AVAILABILITY OF PROXY MATERIALS. EVEN IF YOU HAVE VOTED BY PROXY, YOU MAY STILL VOTE IN PERSON IF YOU ATTEND THE ANNUAL MEETING. PLEASE NOTE, HOWEVER, THAT IF YOU HOLD YOUR SHARES THROUGH A BROKER, BANK OR OTHER NOMINEE, THEN THAT ENTITY IS THE STOCKHOLDER OF RECORD, AND YOU WILL NEED TO FOLLOW THE INSTRUCTIONS ON THE INSTRUCTION FORM THEY SEND TO YOU AND THEY WILL VOTE YOUR SHARES AS YOU DIRECT, OR YOU MUST OBTAIN A PROXY ISSUED IN YOUR NAME FROM THAT ENTITY TO VOTE YOUR SHARES.
Table of Contents
TABLE OF CONTENTS
South San Francisco, California
April 13, 2016
|
| Proposal 4: Advisory Vote on the Compensation of the Named Executive Officer | | | 31 | |
YOUR VOTE IS IMPORTANT
WHETHER OR NOT YOU PLAN TO ATTEND THE 2016 ANNUAL MEETING OF STOCKHOLDERS, TO ENSURE THAT YOU ARE REPRESENTED AT THE MEETING AND TO ENSURE THAT A QUORUM IS PRESENT, YOU ARE URGED TO VOTE YOUR PROXY ONLINE, BY TELEPHONE OR BY RETURNING A PROXY CARD BY MAIL AS INSTRUCTED IN THE NOTICE OF AVAILABILITY OF PROXY MATERIALS. EVEN IF YOU HAVE VOTED BY PROXY, YOU MAY STILL VOTE IN PERSON IF YOU ATTEND THE ANNUAL MEETING. PLEASE NOTE, HOWEVER, THAT IF YOU HOLD YOUR SHARES THROUGH A BROKER, BANK OR OTHER NOMINEE, THEN THAT ENTITY IS THE HOLDER OF RECORD AND YOU WILL NEED TO FOLLOW THE INSTRUCTIONS ON THE INSTRUCTION FORM THEY SEND TO YOU AND THEY WILL VOTE YOUR SHARES AS YOU DIRECT, OR YOU MUST OBTAIN A PROXY ISSUED IN YOUR NAME FROM THAT ENTITY TO VOTE YOUR SHARES.
Required Vote and Board of Director Recommendation | | | 31 | |
210 East Grand Ave.
South San Francisco, CA 94080
Q2019 Proxy Statement UESTIONSi
ANDProxy Statement | Questions and Answers about these Proxy Materials and Voting
| | |
PRELIMINARY COPY | |  |
1851 Harbor Bay Parkway
Alameda, CA 94502
PROXY STATEMENT
FORTHE 2019 ANNUAL MEETINGOF STOCKHOLDERS
MAY 22, 2019
NSWERSQUESTIONS AND ANSWERS ABOUT THESE PROXY MATERIALS AND VOTING
ABOUT THESE PROXY MATERIALSAND VOTING
Why am I receiving these materials?
We have made these materials available to you on the Internet or, upon your request, have delivered printed versions of these materials to you by mail because the Board of Directors or the Board,(the Board), of Exelixis, Inc. (sometimes referred to as “we,” “us”“us,” the “company” or “Exelixis”) is soliciting your proxy to vote at the 20162019 Annual Meeting of Stockholders or the Annual Meeting,(Annual Meeting), including at any adjournments or postponements of the meeting. You are invitedThe Annual Meeting will be held on Wednesday, May 22, 2019, at 8:00 a.m., local time, at our offices at the address set forth above. We invite you to attend the Annual Meeting to vote on the proposals described in this proxy statement.Proxy Statement. However, you do not need to attend the Annual Meeting to vote your shares. Instead, you may simply complete, sign and return a proxy card, or follow the instructions below or in the Notice of Internet Availability of Proxy Materials described below to submit your proxy over the telephone or on the Internet.
We intend to send or make available these materials to stockholders on April 13, 2016.
, 2019.What is included in these proxy materials?
These proxy materials include:
The Notice of the 2016 Annual Meeting of Stockholders;
The Proxy Statement for the Annual Meeting; and
Our Annual Report on Form 10-K for the year ended January 1, 2016, as filed with the Securities and Exchange Commission, or SEC, on February 29, 2016, or the Annual Report.
| ›› | The Notice of the Annual Meeting; |
| ›› | The Proxy Statement for the Annual Meeting; and |
| ›› | Our Annual Report on Form10-K for the fiscal year ended December 28, 2018, as filed with the Securities and Exchange Commission (SEC) on February 22, 2019 (Annual Report). |
If you requested printed versions by mail, these proxy materials also include the proxy card or voting instruction form for the Annual Meeting.
Why did I receive aone-page notice in the mail regarding Internet availability of proxy materials instead of a full set of printed proxy materials?
Pursuant to rules adopted by the SEC, we have elected to use the Internet as the primary means of furnishing proxy materials to our stockholders this year. This method allows us to deliver the proxy materials to you more quickly, lowers our costs significantly and helps to conserve natural resources. Accordingly, we are sending a Notice of Internet Availability of Proxy Materials or Notice(Notice of Availability,Availability) to our stockholders who have not asked us to provide proxy materials in printed form. All stockholders receiving a Notice of Availability can request a printed set of proxy materials. Moreover, all stockholders can access the proxy materials atwww.exel-annualstockholdermeeting.com, irrespective of whether they receive a Notice of Availability or a printed copy of the proxy materials. Instructions on how to access the proxy materials on the Internet or how to request a printed copy may be found in the Notice of Availability and in this Proxy Statement.
In addition, a stockholder may ask to receive proxy materials in printed form by mail or electronically by email on an ongoing basis. We encourage stockholders to take advantage of thisthe option to receive proxy materials electronically by email to help reduce the environmental impact of our annual meeting and to reduce costs associated with the physical printing and mailing of materials in line with our cost-containment strategies.materials. If you choose to receive future proxy materials by email, you will receive an email message next year with instructions containing a link to those materials and a link to the proxy voting website. Your election to receive proxy materials by email will remain in effect until you terminate it.
2019 Proxy Statement 1
Who may vote at the Annual Meeting?
Only stockholders of record at the close of business on March 31, 2016,25, 2019, the Record Date, will be entitled to vote at the Annual Meeting. On the Record Date, there were 228,654,405301,346,561 shares of common stock outstanding and entitled to vote.
Stockholder of Record: Shares Registered in Your Name
If on March 31, 2016,25, 2019, your shares were registered directly in your name with our transfer agent, Computershare Trust Company, N.A., then you are a stockholder of record. As a stockholder of record, you may vote in person at the meetingAnnual Meeting or vote by proxy. Whether or not you plan to attend the Annual Meeting, we urge you to vote by proxy over the telephone or on the Internet as instructed below, or complete and mail the proxy card if you received printed materials.
Beneficial Owner: Shares Registered in the Name of a Broker, Bank or Bank
Other Stockholder of RecordIf on March 31, 2016,25, 2019, your shares were held in an account at a brokerage firm, bank, dealer, or other similar organization, then you are the beneficial owner of shares held in “street name”name,” and these proxy materials are being forwarded to you by that organization. The organization holding your shares is considered to be the stockholder of record for purposes of voting at the Annual Meeting. As a beneficial owner, you have the right to direct your broker, bank or other agentstockholder of record regarding how to vote the shares in your account.account, and you will receive instructions from your broker, bank or other stockholder of record that must be followed in order for your broker, bank or other stockholder of record to vote your shares per your instructions. You are also invited to attend the Annual Meeting. However, since you are not the stockholder of record, you may not vote your shares in person at the meeting unless you request and obtain a valid proxy from your broker, bank or other agent.
stockholder of record giving you the right to vote such shares in person at the Annual Meeting.What am I voting on?
There are four matters scheduled for a vote at the 2016 Annual Meeting. They are as follows:
Election of the three Class II nominees for director named herein to hold office until the 2019 Annual Meeting of Stockholders;
Ratification of Ernst & Young LLP as our independent registered public accounting firm for the fiscal year ending December 30, 2016;
Approval of an amendment and restatement of the Exelixis, Inc. 2000 Employee Stock Purchase Plan, or the 2000 Purchase Plan, to increase the number of shares of common stock reserved for issuance under the 2000 Purchase Plan by 5,000,000 shares and make certain other change as described in Proposal 3; and
Advisory approval of the compensation of our named executive officers, as disclosed in this Proxy Statement.
| ›› | Election of the five Class II nominees for director named herein to hold office until the 2022 Annual Meeting of Stockholders; |
| ›› | Ratification of Ernst & Young LLP as our independent registered public accounting firm for the fiscal year ending January 3, 2020; |
| ›› | Approval of an amendment to Exelixis’ Amended and Restated Certificate of Incorporation (Certificate of Incorporation) to declassify the Board to provide for annual elections by the 2020 Annual Meeting of Stockholders; and |
| ›› | Advisory approval of the compensation of our named executive officers, as disclosed in this Proxy Statement. |
How do I vote?
Whether or not you plan to attend the meeting, we urge you to vote by proxy to ensure your vote is counted. You may still attend the meeting and vote in person even if you have already voted by proxy.
Stockholder of Record: Shares Registered in Your Name
If you are a stockholder of record, you have four ways to vote.
| | | | |

In Person | | ›› | | To vote in person, come to the Annual Meeting and we will give you a ballot when you arrive. You must bring valid photo identification such as a driver’s license or passport and may be asked to provide proof of stock ownership, such as your account statement, as of the Record Date, March 25, 2019. |
| | |

Via Internet | | ›› | | To vote on the Internet, go to www.investorvote.com/EXEL and follow the instructions provided in the Notice of Availability. Your vote must be received by 11:59 p.m., Eastern Time, on May 21, 2019, to be counted. |
| | |

By Telephone | | ›› | | To vote by telephone, request a paper or email copy of the proxy materials by following the instructions provided in the Notice of Availability and call the number provided with the proxy materials to transmit your voting instructions. Your vote must be received by 11:59 p.m. Eastern Time, on May 21, 2019, to be counted. |
| | |

By Mail | | ›› | | To vote by mail, request a paper copy of the proxy materials by following the instructions provided in the Notice of Availability and complete, sign and date the proxy card enclosed with the paper copy of the proxy materials and return it promptly in the envelope that will be provided. If you return your signed proxy card to us before the Annual Meeting, we will vote your shares as you direct. |
In person.2 To vote in person, come to the Annual MeetingExelixis, Inc.
Proxy Statement | Questions and we will give you a ballot when you arrive. You must bring valid photo identification such as a driver’s license or passportAnswers about these Proxy Materials and may be asked to provide proof of stock ownership, such as your account statement, as of the Record Date, March 31, 2016.Voting
Via the Internet. To vote on the Internet, go to www.investorvote.com/EXEL and follow the instructions provided in the Notice of Availability. Your vote must be received by 11:59 p.m., Eastern Time, on May 24, 2016, to be counted.
By Telephone. To vote by telephone, request a paper or email copy of the proxy materials by following the instructions provided in the Notice of Availability and call the number provided with the proxy materials to transmit your voting instructions. Your vote must be received by 11:59 p.m. Eastern Time, on May 24, 2016, to be counted.
By Mail. To vote by mail, request a paper copy of the proxy materials by following the instructions provided in the Notice of Availability and complete, sign and date the proxy card enclosed with the paper copy of the proxy materials and return it promptly in the envelope that will be provided. If you return your signed proxy card to us before the Annual Meeting, we will vote your shares as you direct.
Beneficial Owner: Shares Registered in the Name of Broker, Bank or Bank (i.e.Other Stockholder of Record (i.e., “Street Name”)
If you are a beneficial owner of shares registered in the name of your broker, bank, or other agent,stockholder of record, you should have received the Notice of Availability containing voting instructions from that organization rather than from us. SimplyYou must follow these instructions for your bank, broker or other agentstockholder of record to vote your shares per your instructions. Alternatively, many brokers and banks provide the means to grant proxies to vote shares by telephone and via the Internet. If your shares are held in an account with a broker, bank or bankother stockholder of record providing such a service, you may grant a proxy to vote those shares by telephone or over the Internet as instructed by your broker, bank or bank.other stockholder of record. To vote in person at the Annual Meeting, you must obtain a valid legal proxy from
your broker, bank or other agent.stockholder of record. Follow the instructions from your broker, bank or bankother stockholder of record included with these proxy materials, or contact your broker, bank or bankother stockholder of record to request a proxy form.
legal proxy. |
|
We provide Internet proxy voting to allow you to vote your shares online, with procedures designed to ensure the authenticity and correctness of your proxy vote instructions. However, please be aware that you must bear any costs associated with your Internet access, such as usage charges from Internet access providers and telephone companies. |
How many votes do I have?
On each matter to be voted upon, you have one vote for each share of common stock you own as of the Record Date, March 31, 2016.
25, 2019.How are proxies voted?
All shares represented by valid proxies received prior to the taking of the vote at the Annual Meeting will be voted and, where a stockholder specifies by means of a proxy a choice with respect to any matter to be acted upon, the shares will be voted in accordance with the stockholder’s instructions.
What if I return a proxy card but do not make specific choices?
If you are a stockholder of record and you return a signed and dated proxy card without marking any voting selections, your shares will be voted on the proposals as follows:
“For” the election of Mr. Feldbaum and Drs. Garber and Marchesi as described in Proposal 1;
“For” the ratification of our selection of Ernst & Young LLP as our independent registered public accounting firm for the fiscal year ending December 30, 2016 as described in Proposal 2;
"For" the amendment and restatement of the Exelixis, Inc. 2000 Employee Stock Purchase Plan, or 2000 Purchase Plan, to increase the number of shares of common stock reserved for issuance under the 2000 Purchase Plan by 5,000,000 shares and make certain other changes as described in Proposal 3; and
“For” the advisory approval of the compensation of our named executive officers as described in Proposal 4.
| ›› | “For” the election of Drs. Freire, Garber and Marchesi, Mr. Feldbaum and Ms. Smith as described in Proposal 1; |
| ›› | “For” the ratification of our selection of Ernst & Young LLP as our independent registered public accounting firm for the fiscal year ending January 3, 2020, as described in Proposal 2; |
| ›› | “For” the approval of the amendment to the Certificate of Incorporation to declassify the Board to provide for annual elections by the 2020 Annual Meeting of Stockholders, as described in Proposal 3; and |
| ›› | “For” the advisory approval of the compensation of our named executive officers as described in Proposal 4. |
If any other matter is properly presented at the Annual Meeting, your proxyholder (one of the individuals named on your proxy card) will vote your shares using his or her best judgment.
If you are a beneficial owner of shares heldregistered in streetthe name of your broker, bank or other stockholder of record and you do not provide the organization that holdsbroker, bank or other stockholder of record holding your shares with specific voting instructions, then, under applicable rules,your broker, bank or other stockholder of record will determine if it has the organization that holdsdiscretionary authority to vote on the particular matter. If you are a beneficial owner whose shares are held of record by a broker and you do not provide voting instructions, your shares may generallywill not be voted on any proposal on which the broker does not have discretionary authority to vote. This is called a “brokernon-vote.” In these cases, the broker can register your shares as being present at the Annual Meeting for purposes of determining the presence of a quorum, but will not be able to vote on “routine”those matters but cannot vote on “non-routine” matters. for which specific authorization is required under the rules of the New York Stock Exchange.
If you are a beneficial owner whose shares are held of record by a broker, your broker has discretionary voting authority under the organization that holds your shares does not receive instructions from you on howrules of the New York Stock Exchange to vote your shares on a non-routine matter, that organization will informProposal No. 2 (the ratification of the inspectorappointment of election that itErnst & Young LLP as our independent registered public accounting firm for the fiscal year ending January 3, 2020), even if your broker does not receive voting instructions from you. However, your broker does not have thediscretionary authority to vote on this matter with respect to your shares. This is generally referred to asProposal No. 1, Proposal No. 3 or Proposal No. 4 without voting instructions from you, in which case a “broker non-vote.” Proposal 2 constitutes a “routine” management proposal,brokernon-vote will occur and thus, if you do not give your broker or nominee specific instructions, your broker or nominee will nevertheless have the authority to vote your shares with respect to this proposal, but will not have the authority to vote your shares with respect to Proposalsbe voted on Proposal No. 1, Proposal No. 3 or 4, which constitute “non-routine” proposals.Proposal No. 4.
2019 Proxy Statement 3
Who is paying for this proxy solicitation?
We are soliciting proxies and will bear the entire cost of soliciting proxies, including the preparation, printing and mailing of the Notice of Availability, the Notice of Annual Meeting, the Proxy Statement, the proxy card and any additional information furnished to stockholders. We have engaged Morrow Sodali LLC, located at 470 West Ave, Stamford, Connecticut 06902, to assist in the solicitation of proxies from shareholders for a fee of $13,000 plus reimbursement of customaryout-of-pocket expenses. Copies of solicitation materials will be furnished to banks, brokerage houses, fiduciaries and custodians holding in their names shares of our common stock beneficially owned by others to forward to such beneficial owners. We may reimburse persons representing beneficial owners of our common stock for their costs of forwarding solicitation materials to such beneficial owners. Original solicitation of proxies by mail may be supplemented by telephone, telegram or personal solicitation by our directors, officers or other regular employees. No additional compensation will be paid to directors, officers or other regular employees for such services.
What does it mean if I receive more than one Notice of Availability or proxy card?
If you receive more than one Notice of Availability or proxy card, your shares are registered in more than one name or are registered in different accounts. Please follow the instructions on each Notice of Availability or proxy card to ensure that all of your shares are voted.
Can I change my vote after submitting my proxy?
Yes. You can revoke your proxy at any time before the final vote at the Annual Meeting. You may revoke your proxy in the following ways:
Stockholder of Record: Shares Registered in Your Name
Your proxy may be revoked by filing with the Secretary of Exelixis at our principal executive office, Exelixis, Inc., 210 East Grand Avenue, South San Francisco, California 94080, either (1) a written notice of revocation or (2) a duly executed proxy card bearing a later date.
Your proxy may also be revoked by granting a subsequent proxy by telephone or on the Internet (your latest telephone or Internet proxy is the one that is counted).
Your proxy may also be revoked by attending the Annual Meeting and voting in person. Attendance at the Annual Meeting will not, by itself, revoke your proxy.
| ›› | Your proxy may be revoked by filing with the Secretary of Exelixis at our principal executive office, Exelixis, Inc., 1851 Harbor Bay Parkway, Alameda, California 94502, either (1) a written notice of revocation or (2) a duly executed proxy card bearing a later date. |
| ›› | Your proxy may also be revoked by granting a subsequent proxy by telephone or on the Internet (your latest telephone or Internet proxy is the one that is counted). |
| ›› | Your proxy may also be revoked by attending the Annual Meeting and voting in person. Attendance at the Annual Meeting will not, by itself, revoke your proxy. |
Beneficial Owner: Shares Registered in the Name of Broker, Bank or Bank
If your shares are held by your broker or bank as nominee or agent, you should follow the instructions provided by your broker or bank to revoke any prior voting instructions.
Other Stockholder of Record | ›› | If your shares are held by your broker or bank as nominee or agent, you should follow the instructions provided by your broker or bank to revoke any prior voting instructions. |
What is the quorum requirement for the Annual Meeting?
A majority of the outstanding shares entitled to vote at the Annual Meeting must be present at the Annual Meeting, either in person or by proxy, in order to hold a valid meeting. This is called a “quorum.”
If you are a stockholder of record, your shares will be counted towards the quorum only if you vote in person at the meeting or have properly voted by proxy on the Internet, by telephone or by submitting a proxy card.card by mail or at the Annual Meeting. You may vote “For,” “Against” or “Abstain” for each proposal.with respect to Proposal Nos. 1, 2, 3 and 4. Abstentions will be counted towards the number of shares considered to be present at the meeting for purposes of determining whether a quorum is present.
If you are a beneficial owner holding your shares in “street name” then only the broker, bank or bankother stockholder of record can vote your shares unless you obtain a valid proxy from the broker, bank or bank.other stockholder of record. See “What if I return a proxy card but do not make specific choices?” above. Shares represented by “brokernon-votes” will be counted in determining whether there is a quorum present. Votes will be counted by the inspector of election appointed for the Annual Meeting. If there is no quorum, either the chairman of the Annual Meeting or the holders of a majority of shares present at the Annual Meeting in person or represented by proxy may adjourn the Annual Meeting to another date.
4 Exelixis, Inc.
Proxy Statement | Questions and Answers about these Proxy Materials and Voting
How many votes are needed to approve each proposal, how are votes counted, and how are votes counted?
Proposal 1-Election of Directors: Directors in an uncontested election, such as this one, are elected by majority vote. Each of the three Class II nominees must receive “For” votes from the holders of a majority of shares cast with respect to such director (i.e., the number of shares voted “For” a director must exceed the number of shares voted “Against” that director). Abstentionsabstentions and brokernon-votes if any, are not counted for purposes of electing directors and will have no effect on the results of this vote.
treated? | ›› | Proposal1-Election of Directors: Directors in an uncontested election, such as this one, are elected by a majority of the votes cast. Each of the five Class II nominees must receive “For” votes from the holders of a majority of shares cast with respect to such director (i.e., the number of shares voted “For” a director must exceed the number of shares voted “Against” that director). Abstentions and brokernon-votes, if any, are not counted for purposes of electing directors and will have no effect on the results of this vote. |
| ›› | Proposal2-Ratification of Ernst & Young LLP:The affirmative vote of a majority of shares present in person or represented by proxy at the Annual Meeting and entitled to vote on the proposal is required to ratify the selection of Ernst & Young LLP as our independent registered public accounting firm for the fiscal year ending January 3, 2020. Abstentions will have the effect of votes against this proposal. Brokers generally have discretionary authority to vote on the ratification of our independent accounting firm; thus we do not expect any brokernon-votes on this proposal. |
| ›› | Proposal3-Approval of Amendment to Certificate of Incorporation to Declassify the Board:The affirmative vote of at leastsixty-six andtwo-thirds percent(66-2/3%) of the shares issued and outstanding and entitled to vote on the proposal is required to approve the amendment to our Certificate of Incorporation to declassify the Board to provide for annual elections by the 2020 Annual Meeting of Stockholders. Abstentions and brokernon-votes, if any, will have the effect of votes against this proposal. |
| ›› | Proposal4-Advisory Vote on Executive Compensation: The affirmative vote of a majority of shares present in person or by represented proxy at the Annual Meeting and entitled to vote on the proposal is required to approve thenon-binding, advisory vote on executive compensation. Abstentions will be counted toward the tabulation of votes cast on the proposal and will have the same effect as votes against this proposal. Brokernon-votes will have no effect and will not be counted towards the vote total. Since the vote is advisory, it is not binding on the Board or on us. Nevertheless, the views expressed by our stockholders, whether through this vote or otherwise, are important to the Compensation Committee and the Board and, accordingly, the Compensation Committee and Board intend to consider the results of this vote in making determinations in the future regarding executive compensation arrangements. Your vote will serve as an additional tool to guide the Compensation Committee and Board in continuing to improve the alignment of our executive compensation programs with business objectives and performance and with the interests of our stockholders. |
Proposal 2-Ratification of Ernst & Young LLP: The affirmative vote of a majority of shares present in person or represented by proxy at the Annual Meeting and entitled to vote on the proposal is required to ratify the selection of Ernst & Young LLP as our independent registered public accounting firm for the fiscal year ending December 30, 2016. Abstentions will have the effect of votes against this proposal. Brokers generally have discretionary authority to vote on the ratification of our independent accounting firm; thus we do not expect any broker non-votes on this proposal. To the extent there are any broker non-votes, they will have no effect on the results of this vote.
Proposal 3-Amendment and Restatement of the 2000 Purchase Plan: The affirmative vote of a majority of shares present in person or by represented proxy at the Annual Meeting and entitled to vote on the proposal is required to approve the amendment and restatement of the 2000 Purchase Plan. Abstentions will be counted toward the tabulation of votes cast on the proposal and will have the same effect as votes against this proposal. Broker non-votes will have no effect and will not be counted towards the vote total.
Proposal 4-Advisory Vote on Executive Compensation: The affirmative vote of a majority of shares present in person or by represented proxy at the Annual Meeting and entitled to vote on the proposal is required to approve the non-binding, advisory vote on executive compensation. Abstentions will be counted toward the tabulation of votes cast on the proposal and will have the same effect as votes against this proposal. Broker non-votes will have no effect and will not be counted towards the vote total. Since the vote is advisory, it is not binding on the Board or on us. Nevertheless, the views expressed by our stockholders, whether through this vote or otherwise, are
important to management and the Board and, accordingly, the Compensation Committee and Board intend to consider the results of this vote in making determinations in the future regarding executive compensation arrangements. Your vote will serve as an additional tool to guide the Compensation Committee and Board in continuing to improve the alignment of our executive compensation programs with business objectives and performance and with the interests of our stockholders.
Do I have dissenters’ rights?
No. We are organized as a corporation under Delaware law. Under the Delaware General Corporation Law, our stockholders are not entitled to dissenters’ rights with respect to any of the proposals set forth in this Proxy Statement and we will not independently provide the stockholders with any such rights.
How can I find out the results of the voting at the Annual Meeting?
Preliminary voting results will be announced at the Annual Meeting. In addition, final voting results will be published in a current report on Form8-K that we expect to file within four business days after the Annual Meeting. If final voting results are not available to us in time to file a Form8-K within four business days after the Annual Meeting, we intend to file a Form8-K to publish preliminary results and, within four business days after the final results are known to us, file an additional Form8-K to publish the final results.
Will other matters be voted on at the Annual Meeting?
We are not aware of any matters to be presented at the Annual Meeting other than those described in this Proxy Statement. If any other matters not described in the Proxy Statement are properly presented at the meeting, proxies will be voted in accordance with the best judgment of the proxyholders.
What proxy materials are available on the Internet?
This Proxy Statement and our 2015 Annual Report are available atwww.exel-annualstockholdermeeting.com.
2019 Proxy Statement www.exel-annualstockholdermeeting.com.5
What is the deadline for submitting stockholder proposals for the 20172019 Annual Meeting?
To be considered for inclusion in the 20172020 proxy materials, your proposal must be submitted in writing by December 14, 2016,13, 2019, to Exelixis’ Secretary at Exelixis, Inc., 210 East Grand Avenue, South San Francisco,1851 Harbor Bay Parkway, Alameda, California 94080,94502, and you must comply with all applicable requirements of Rule14a-8 promulgated under the Securities Exchange Act of 1934, as amended.amended (Exchange Act). However, if our 20172020 Annual Meeting of Stockholders is held before April 25, 2017,22, 2020, or after June 24, 2017,21, 2020, then the deadline will be a reasonable time prior to the time that we make our proxy materials available to our stockholders, either online or in printed form.
If you wish to submit a proposal or nominate a director at the 20172020 Annual Meeting of Stockholders, but you are not requesting that your proposal or nomination be included in next year’s proxy materials, you must submit your proposal in writing, in the manner set forth in our Bylaws, to Exelixis’ Secretary at Exelixis, Inc., 210 East Grand Avenue, South San Francisco,1851 Harbor Bay Parkway, Alameda, California 94080,94502, to be received no earlier than the close of business on February 24, 2017,22, 2020, and no later than the close of business on March 26, 2017.23, 2020. However, if our 20172020 Annual Meeting of Stockholders is held before April 25, 2017,22, 2020, or after June 24, 2017,21, 2020, then you must notify Exelixis’ Secretary, in writing, not earlier than the close of business on the 90th day prior to the date of the 20172020 Annual Meeting of Stockholders and not later than the close of business on the later of (i) the 60th day prior to the date of the 20172020 Annual Meeting of Stockholders or (ii) if we publicly announce the date of the 20172020 Annual Meeting of Stockholders fewer than 70 days prior to the date of the 20172020 Annual Meeting of Stockholders, the 10th day following the day that we first make such public announcement of the date of the 20172020 Annual Meeting of Stockholders. We also advise you to review our Bylaws, which contain additional requirements about advance notice of stockholder proposals and director nominations. The chairperson of the 20172020 Annual Meeting of Stockholders may determine, if the facts warrant, that a matter has not been properly brought before the meeting and, therefore, may not be considered at the meeting. In addition, if you do not also comply with the requirements ofunless prohibited by Rule14a-4(c)(1) promulgated under the Securities Exchange Act, of 1934, as amended, our management will have discretionary authority to vote all shares for which it has proxies for any such stockholder proposal or director nomination, including in opposition to such stockholder proposal or director nomination.
How may I obtain a printed copy of the Proxy Materials?
Instructions on how to obtain a printed copy of the proxy materials are set forth in the Notice of Availability.
6 Exelixis, Inc.
Where can I obtain directionsProxy Statement | Questions and Answers about these Proxy Materials and Voting
Directions to the Annual Meeting?
Meeting1851 Harbor Bay Parkway
Alameda, CA 94502
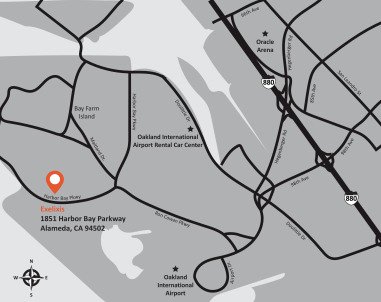
| | | | | | |
| From Oakland International Airport | | | | | From the City of San Francisco |
| Via Ron Cowan Parkway | | | | | | Via Highway 80 East (Bay Bridge) |
• Leave Oakland International Airport via Airport Drive • Merge right toward Ron Cowan Parkway • Turn left at first light onto Ron Cowan Parkway • Continue straight at third light onto Harbor Bay Parkway • 1851 Harbor Bay Parkway parking lot will be at second driveway on your right after passing North Loop Road. | | | | | | • Highway 80 East (over Bay Bridge) • After Bay Bridge, use right two lanes to take exit toward Highway 880 South (toward Oakland International Airport) • Continue onto Highway 880 South • Take exit 35 for 98th Avenue (toward Oakland International Airport) • Turn right at first light onto 98th Avenue • 98th Avenue becomes Bessie Coleman Avenue • Merge right onto Ron Cowan Parkway • Continue straight at second light onto Harbor Bay Parkway • 1851 Harbor Bay Parkway parking lot will be at second driveway on your right after passing North Loop Road. |
Directions to our Annual Meeting may also be found on our website at:www.exelixis.com/about/locations-and-directions.
2019 Proxy Statement www.exelixis.com/about/locations-and-directions7
PROPOSAL 1
ELECTIONOF CLASS II DIRECTORSIRECTORS
Our Certificate of Incorporation and Bylaws provide that the Board is divided into three classes, with each class having a three-year term. As of the date of this Proxy Statement, the Board has ten members -- three Class I directors, three Class II directors and four Class III directors. The term of office for each of the three directors in Class II will expire at the Annual Meeting. Each of the director nomineesas set forth in this Proxy Statement is currently a director of Exelixis who was previously elected by the stockholders. table below:
| | | | | | | | | | | | | | | | | | |
Director Nominees | | Class | | Age | | Position | | Director
Since | | | Current Term
Expires | | | Expiration of
Term For
Which
Nominated | |
Carl B. Feldbaum, Esq. | | II | | 75 | | Director | | | 2007 | | | | 2019 | | | | 2022 | |
Maria C. Freire, Ph.D. | | II | | 64 | | Director | | | 2018 | | | | 2019 | | | | 2022 | |
Alan M. Garber, M.D., Ph.D. | | II | | 63 | | Director | | | 2005 | | | | 2019 | | | | 2022 | |
Vincent T. Marchesi, M.D., Ph.D. | | II | | 83 | | Director | | | 2001 | | | | 2019 | | | | 2022 | |
Julie Anne Smith | | II | | 48 | | Director | | | 2016 | | | | 2019 | | | | 2022 | |
Continuing Directors | | Class | | Age | | Position | | Director
Since | | | Current Term
Expires | | | | |
Michael M. Morrissey, Ph.D. | | III | | 58 | | President and Chief Executive Officer | | | 2010 | | | | 2020 | | | | | |
Stelios Papadopoulos, Ph.D. | | III | | 70 | | Chairman of the Board | | | 1994 | | | | 2020 | | | | | |
George A. Scangos, Ph.D. | | III | | 70 | | Director | | | 1996 | | | | 2020 | | | | | |
Lance Willsey, M.D. | | III | | 57 | | Director | | | 1997 | | | | 2020 | | | | | |
Charles Cohen, Ph.D. | | I | | 68 | | Director | | | 1995 | | | | 2021 | | | | | |
George Poste, DVM, Ph.D., FRS | | I | | 74 | | Director | | | 2004 | | | | 2021 | | | | | |
Jack L. Wyszomierski, Ph.D., FRS | | I | | 63 | | Director | | | 2004 | | | | 2021 | | | | | |
If elected at the Annual Meeting, each of these director nominees would serve until the 20192022 Annual Meeting of Stockholders and until histhe director’s successor is elected and has qualified, or, if sooner, until the director’s death, resignation or removal.
Each of Drs. Garber and Marchesi, and Mr. Feldbaum, were previously elected by our stockholders. Ms. Smith and Dr. Freire were appointed by our Board in 2016 and 2018, respectively, and are being elected by our stockholders for the first time at the Annual Meeting. Ms. Smith was recommended for election to our Board by a third-party search firm, and Dr. Freire was recommended for election to our Board by several of thenon-employee directors.
As this is an uncontested election, directorseach director will be elected by a majority of the votes present in person or represented by proxy and entitled to votecast at the Annual Meeting.meeting with respect to the election of that director. A majority of the votes cast means that the number of shares voted “for” a director must exceed the number of votes cast “against” that director. If any nominee becomes unavailable for election as a result of an unexpected occurrence, your shares will be voted for the election of such substitute nominee as the Board, after receiving the recommendation of the Nominating and Corporate Governance Committee of the Board, may propose. Each person nominated for election has agreed to serve if elected, and we have no reason to believe that any nominee will be unable to serve.
AllOur Corporate Governance Guidelines require that all director nominees set forth in this Proxy Statement have tendered an irrevocable resignation as a director conditioned uponupon: (i) such director failing to receive a majority of the votes present in person or represented by proxy and entitled to votecast at the Annual Meeting,Meeting; and (ii) acceptance by the Board of such resignation. If a director nominee who is serving as a director at the time of the election does not receive a majority of the votes present in person or represented by proxy and entitled to votecast at the Annual Meeting, then the Board will take the following actions:
The Nominating and Corporate Governance Committee will act to determine whether to accept the director’s conditional resignation and will submit such recommendation for prompt consideration by the Board. The Board will act on the Nominating and Corporate Governance Committee’s recommendation within ninety days following certification of the stockholder vote. In making their decision, the Nominating and Corporate Governance Committee will evaluate the best interests of Exelixis and its stockholders and shall consider all factors and information deemed relevant. The director who tenders his or her conditional resignation shall not participate in the Nominating and Corporate Governance Committee’s recommendation or Board action regarding whether to accept the conditional resignation of such director.
8 Exelixis, Inc.
Proposal 1 | Election of Class II Directors
The Board will act on the Nominating and Corporate Governance Committee’s recommendation within ninety days following certification of the stockholder vote.
If the Board determines not to accept the conditional resignation of a director, the Board will promptly disclose its decision-making process and decision to reject the conditional resignation in a Form8-K furnished to the SecuritiesSEC.
The Board regularly evaluates the skills and Exchange Commission,experiences that it believes are desirable to be represented on the Board and best align with our strategic vision and business and operations. Below are certain qualifications, skills and experiences of our current directors that contribute the Board’s effectiveness as a whole.
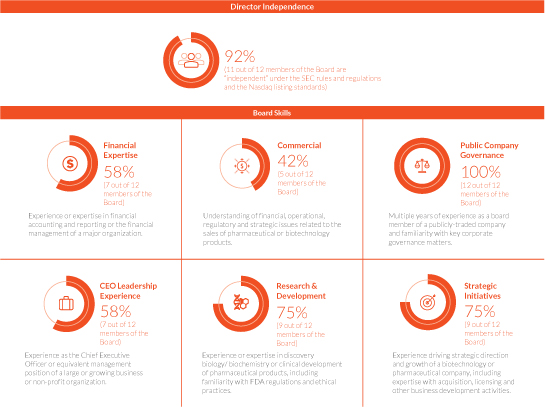
Director Independence 92% (11 out of 12 members of the Board are "independent" under the SEC rules and regulations and the Nasdaq listing standards) Board Skills CEO Leadership Experience 50% (6 out of 12 members of the Board) Public Company Governance 83 % (10 out of 12 members of the Board) Experience as the Chief Executive Officer or equivalent management position of a large or growing business or non-profit organization. Experience as a board member of another publicly-traded company and familiarity with key corporate governance matters. Research & Development 75% (9 out of 12 members of the Board) Commercial 42% (5 out of 12 members of the Board) Experience or expertise in discovery biology/biochemistry or clinical development of pharmaceutical products, including familiarity with FDA regulations ethical practices. Understanding of financial, operational, regulatory and strategic issues related to the sales of pharmaceutical or biotechnology products. Financial Expertise 50% (7 out of 12 members of the Board) Strategic Initiatives 67% (8 out of 12 members of the Board) Experience or expertise in financial accounting and reporting or the SEC.
Setfinancial management of a major organization. Experience driving strategic direction and growth of a biotechnology or pharmaceutical company, including expertise with acquisitions, licensing and other business development activities.In addition, set forth below is biographical information for each person nominated and each person whose term of office as a director will continue after the Annual Meeting. Incorporated within each biography is a description of the specific experience, qualifications, attributesskills and skillsexperiences of each director or director nominee that led our Board to conclude that the individual should serve as a director as of the date of this Proxy Statement.
2019 Proxy Statement 9
Class II Director Nominees
for Election for a Three-Year Term Expiring at the 20192022 Annual Meeting
| | |
Carl B. Feldbaum, Esq. President Emeritus, Biotechnology Innovation Organization | | Director since 2007 Age 75 Key Qualifications and Expertise: Our Board concluded that Mr. Feldbaum should continue to serve as a director of Exelixis due to his training as an attorney, his knowledge and experience with respect to the biotechnology, pharmaceutical and healthcare industries, his broad leadership experience resulting from service on various boards and as an executive officer and his knowledge and experience with policymaking, regulatory issues and other governmental matters. Committee Assignments: • Compensation Committee • Nominating and Corporate Governance Committee |
Carl B. Feldbaum, Esq., has been a director since February 2007. Mr. Feldbaum serves as a member emeritus of the board of directors of BIO Ventures for Global Health, a non-profit organization, and is president emeritus of the Biotechnology Innovation Organization (BIO), which represents more than 1,000 biotechnology companies, academic institutions and state biotechnology centers internationally. Mr. Feldbaum served as president of BIO from 1993 until his retirement in 2005. Prior to joining BIO, Mr. Feldbaum was chief of staff to Senator Arlen Specter of Pennsylvania. He also was president and founder of Palomar Corporation, a national security “think tank” in Washington, D.C. Before founding Palomar Corporation, Mr. Feldbaum was Assistant to the Secretary of Energy and served as the Inspector General for defense intelligence in the U.S. Department of Defense. Mr. Feldbaum served as a member of the board of directors of the following publicly-held companies: Actelion, Ltd, a biopharmaceutical company focused on the discovery, development and commercialization of innovative drugs for diseases with significant unmet medical needs (acquired by Johnson & Johnson in 2017), from 2005 to 2015; Trovagene, Inc., a precision medicine biotechnology company focused on the development of oncology therapeutics for improved cancer care, from 2014 to 2015; and Connetics Corporation, a specialty pharmaceutical company focused on the development and commercialization of innovative therapeutics for the dermatology market, from 2005 until its acquisition by Stiefel Laboratories, Inc. in 2006. Mr. Feldbaum holds an A.B. in Biology from Princeton University and a J.D. from the University of Pennsylvania Law School. |
| | |
Maria C. Freire, Ph.D. President and Executive Director, Foundation for the National Institutes of Health | | Director since 2018 Age 64 Key Qualifications and Expertise: Our Board concluded that Dr. Freire should continue to serve as a director of Exelixis due to her training as a scientist, her knowledge and experience with respect to medical research, the pharmaceutical industry and government healthcare policymaking, as well as her leadership experience in the public sector. Committee Assignments: • Nominating and Corporate Governance Committee • Research & Development Committee Current Public Company Boards: • Alexandria Real Estate Equities |
Maria C. Freire, Ph.D., has been a director since April 2018. Since November 2012, Dr. Freire has served as President and Executive Director and as a member of the board of directors of the Foundation for the National Institutes of Health. From March 2008 to November 2012, she served as President and as a member of the board of directors of the Albert and Mary Lasker Foundation. Prior to joining the Lasker Foundation, Dr. Freire served as President and Chief Executive Officer of the Global Alliance for TB Drug Development from 2001 to 2008 and Director of the Office of Technology Transfer at the National Institutes of Health from 1995 to 2001. Dr. Freire has served on the board of directors of Alexandria Real Estate Equities, Inc. a publicly-held urban office real estate investment trust uniquely focused on collaborative life science and technology campuses, since April 2012, and has served on the boards of numerous national and international organizations, including the Science Board of the U.S. Food and Drug Administration, the World Health Organization Commission on Intellectual Property Rights, Innovation and Public Health and the United Nations Secretary General’s High Level Panel on Access to Medicines. Dr. Freire is also a member of the National Academy of Medicine and the Council on Foreign Relations, and she is the recipient of numerous awards, including a 2017 Gold Stevie Award for “Woman of the Year,” the U.S. Department of Health and Human Services Secretary’s Award for Distinguished Service, the Arthur S. Fleming Award and the Bayh-Dole Award. Dr. Freire holds a Ph.D. in Biophysics from the University of Virginia and a B.S. from the Universidad Peruana Cayetano Heredia in Lima, Peru. |
10 age 72, has been a director since February 2007. Mr. Feldbaum serves as a member of the board of directors of BIO Ventures for Global Health, a non-profit organization, and is president emeritus of the Biotechnology Industry Organization (BIO), which represents more than 1,000 biotechnology companies, academic institutions and state biotechnology centers internationally. Mr. Feldbaum served as president of BIO from 1993 until his retirement in 2005. Prior to joining BIO, Mr. Feldbaum was chief of staff to Senator Arlen Specter of Pennsylvania. He also was president and founder of Palomar Corporation, a national security “think tank” in Washington, D.C. Before founding Palomar Corporation, Mr. Feldbaum was Assistant to the Secretary of Energy and served as the Inspector General for defense intelligence in the U.S. Department of Defense. Mr. Feldbaum served as a member of the board of directors of the following companies: Actelion, Ltd, a biopharmaceutical company, from 2005 to 2015; Trovagene,Exelixis, Inc. from 2014 to 2015; and Connetics Corporation from 2005 until its acquisition by Stiefel Laboratories, Inc. in 2006. Mr. Feldbaum holds an A.B. in Biology from Princeton University and a J.D. from the University of Pennsylvania Law School. Our Board has concluded that Mr. Feldbaum should continue to serve as a director of Exelixis as of the date of this
Proposal 1 | Class II Director Nominees
| | |
Alan M. Garber, M.D., Ph.D. Provost of Harvard University | | Director since 2005 Age 63 Key Qualifications and Expertise: Our Board concluded that Dr. Garber should continue to serve as a director of Exelixis due to his training as a physician and economist, his knowledge and experience with respect to the life sciences, healthcare and pharmaceutical industries, and his knowledge and experience with policymaking, regulatory issues and other governmental matters. Committee Assignments: • Nominating and Corporate Governance Committee (chair) • Research & Development Committee Current Public Company Boards: • Vertex Pharmaceuticals Incorporated |
Alan M. Garber, M.D., Ph.D., has been a director since January 2005. He became Provost of Harvard University, Mallinckrodt Professor of Health Care Policy at Harvard Medical School, and a Professor in the Harvard Kennedy School of Government and in the Department of Economics at Harvard in September 2011. Before moving to Harvard, from 1998 until August 2011, he was the Henry J. Kaiser Jr. Professor, a Professor of Medicine, and a Professor (by courtesy) of Economics, Health Research and Policy, and of Economics in the Graduate School of Business at Stanford University. Dr. Garber also served as the Director of the Center for Primary Care and Outcomes Research and the Center for Health Policy at Stanford. During his tenure at Stanford University, Dr. Garber also served as a Senior Fellow at the Freeman Spogli Institute for International Studies and as a staff physician at the VA Palo Alto Health Care System. Dr. Garber has served as a member of the board of directors of Vertex Pharmaceuticals Incorporated, a publicly-held biotechnology company focused on developing and commercializing therapies for the treatment of cystic fibrosis, since June 2017. Dr. Garber is a member of the National Academy of Medicine, the American Society of Clinical Investigation, the Association of American Physicians and the American Academy of Arts and Sciences. He is a Fellow of the American College of Physicians, the Royal College of Physicians and the American Association for the Advancement of Science. Dr. Garber is also a Research Associate with the National Bureau of Economic Research and served as founding Director of its Health Care Program for nineteen years. He has also served as a member of the National Advisory Council on Aging at the National Institutes of Health, as a member of the Board of Health Advisers of the Congressional Budget Office and as Chair of the Medicare Evidence Development and Coverage Advisory Committee at the Centers for Medicare and Medicaid Services. Dr. Garber previously served on the editorial board of acclaimed scientific journals and has received numerous awards and honors. Dr. Garber holds an A.B. summa cum laude, an A.M. and a Ph.D., all in Economics, from Harvard University, and an M.D. with research honors from Stanford University. |
| | |
Vincent T. Marchesi, M.D., Ph.D. Professor of Pathology and Cell Biology, Yale University | | Director since 2001 Age 83 Key Qualifications and Expertise: Our Board concluded that Dr. Marchesi should continue to serve as a director of Exelixis due to his training as a physician and scientist and his research and experience in the fields of healthcare and life sciences, with a particular focus on biotechnology. Committee Assignments: • Compensation Committee • Research & Development Committee |
Vincent T. Marchesi, M.D., Ph.D., has been a director since May 2001. Since 1973, Dr. Marchesi has been a Professor of Pathology and Cell Biology at Yale University and, since 1991, the Director of the Boyer Center for Molecular Medicine at Yale University. In 1982, Dr. Marchesi co-founded Molecular Diagnostics, Inc., a diagnostic development company. Dr. Marchesi was formerly Chair of Pathology at the Yale-New Haven Hospital. Dr. Marchesi holds an M.D. from Yale University and a Ph.D. from Oxford University, and is a member of the National Academy of Sciences and the Institute of Medicine. |
2019 Proxy Statement due to his training as an attorney, his knowledge and experience with respect to the biotechnology, pharmaceutical and healthcare industries, his broad leadership experience resulting from service on various boards and as an executive officer and his knowledge and experience with policymaking, regulatory issues and other governmental matters.
Alan M. Garber, M.D., Ph.D., age 60, has been a director since January 2005. He became Provost of Harvard University, Mallinckrodt Professor of Health Care Policy at Harvard Medical School, and a Professor in the Harvard Kennedy School of Government and in the Department of Economics at Harvard in September 2011. Before moving to Harvard, from 1998 until August 2011, he was the Henry J. Kaiser Jr. Professor, a Professor of Medicine, and a Professor (by courtesy) of Economics, Health Research and Policy, and of Economics in the Graduate School of Business at Stanford University. Dr. Garber also served as the Director of the Center for Primary Care and Outcomes Research and the Center for Health Policy at Stanford. During his tenure at Stanford University, Dr. Garber also served as a Senior Fellow at the Freeman Spogli Institute for International Studies and as a staff physician at the VA Palo Alto Health Care System. Dr. Garber is a member of the National11
| | |
Julie Anne Smith President and Chief Executive Officer, E-Scape Bio, Inc. | | Director since 2016 Age 48 Key Qualifications and Expertise: Our Board concluded that Ms. Smith should continue to serve as a director of Exelixis due to her knowledge and experience with respect to biotechnology, healthcare and pharmaceutical industries and her broad leadership experience resulting from service as an executive in the pharmaceutical industry. Committee Assignments: • Audit Committee • Compensation Committee Current Public Company Boards: • Audentes Therapeutics, Inc. |
Julie Anne Smith has been a director since September 2016. Since August 2018, Ms. Smith has served as President and Chief Executive Officer and as a member of the board of directors ofE-Scape Bio, Inc., a privately held biotechnology company developing novel small-molecule therapeutics for patients with inherited forms of neurodegenerative diseases. From July 2017 until June 2018, Ms. Smith served as President and Chief Executive Officer and as a member of the board of directors of Nuredis, Inc., a privately-held biotechnology company developing novel therapies to treat inherited neurodegenerative diseases caused by nucleotide repeat expansions. Prior to Nuredis, she served as President and Chief Executive Officer of Raptor Pharmaceutical Corp. from January 2015 until the company’s acquisition by Horizon Pharma plc in October 2016, where she also served as Executive Vice President and Chief Operating Officer from 2012 to 2014. From 2008 to 2012, Ms. Smith served as Chief Commercial Officer of Enobia Pharmaceuticals prior to the company’s acquisition by Alexion Pharmaceuticals, Inc. Previously, Ms. Smith served as Vice President of Commercial at Jazz Pharmaceuticals from 2006 to 2008, as Vice President, Global Marketing at Genzyme General from 2001 to 2005, and helped to establish the operations and business development function for the biotech start-up, Novazyme Pharmaceuticals, from 2000 to 2001. Ms. Smith began her industry career at Bristol-Myers Squibb Company in 1996. Ms. Smith has served on the board of directors of Audentes Therapeutics, Inc. a publicly-held clinical stage biotechnology company focused on developing and commercializing gene therapy products for patients suffering from serious, life-threatening rare diseases caused by single gene defects, since December 2016 and previously, Ms. Smith was a Director on the Health and Emerging Companies Sections of the BIO board. Ms. Smith holds a B.S. in biological and nutritional sciences from Cornell University. |
6THE BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” EACH NAMED NOMINEE.
Academy of Medicine, the American Society of Clinical Investigation, the Association of American Physicians and the Board on Science, Technology, and Economic Policy at the National Academies. He is a Fellow of the American College of Physicians and the Royal College of Physicians. Dr. Garber is also a Research Associate with the National Bureau of Economic Research and served as founding Director of its Health Care Program for nineteen years. He has also served as a member of the National Advisory Council on Aging at the National Institutes of Health, as a member of the Board of Health Advisers of the Congressional Budget Office and as Chair of the Medicare Evidence Development and Coverage Advisory Committee at the Centers for Medicare and Medicaid Services. Dr. Garber previously served on the editorial board of acclaimed scientific journals and has received numerous awards and honors. Dr. Garber holds an A.B. summa cum laude, an A.M. and a Ph.D., all in Economics, from Harvard University, and an M.D. with research honors from Stanford University. Our Board has concluded that Dr. Garber should continue to serve as a director of Exelixis as of the date of this Proxy Statement due to his training as a physician and economist, his knowledge and experience with respect to the life sciences, healthcare and pharmaceutical industries, and his knowledge and experience with policymaking, regulatory issues and other governmental matters.
Vincent T. Marchesi, M.D., Ph.D., age 80, has been a director since May 2001. Since 1973, Dr. Marchesi has been a Professor of Pathology and Cell Biology at Yale University and, since 1991, the Director of the Boyer Center for Molecular Medicine at Yale University. In 1982, Dr. Marchesi co-founded Molecular Diagnostics, Inc., a diagnostic development company. Dr. Marchesi was formerly Chair of Pathology at the Yale-New Haven Hospital. Dr. Marchesi holds an M.D. from Yale University and a Ph.D. from Oxford University, and is a member of the National Academy of Sciences and the Institute of Medicine. Our Board has concluded that Dr. Marchesi should continue to serve as director of Exelixis as of the date of this Proxy Statement due to his training as a physician and scientist and his research and experience in the fields of healthcare and life sciences, with a particular focus on biotechnology.
THE BOARDOF DIRECTORS RECOMMENDSA VOTE FOR EACH NAMED NOMINEE.
Class III Directors
Continuing in Office Until the 20172020 Annual Meeting
| | |
Michael M. Morrissey, Ph.D. President and Chief Executive Officer, Exelixis, Inc. | | Director since 2010 Age 58 Key Qualifications and Expertise: Our Board concluded that Dr. Morrissey should continue to serve as a director of Exelixis due to his leadership role as the President and Chief Executive Officer of Exelixis. Beyond his role as Exelixis’ principal executive officer, the Board also considered Dr. Morrissey’s extensive qualifications, including his training as a scientist, his significant knowledge and experience with respect to the biotechnology, healthcare and pharmaceutical industries, comprehensive leadership background resulting from service as an executive in the biotechnology industry, and his ability to bring historic knowledge and continuity to the Board. Committee Assignments: • None |
Michael M. Morrissey, Ph.D., has served as a director and as Exelixis’ President and Chief Executive Officer since July 2010. Dr. Morrissey has held positions of increasing responsibility at Exelixis since he joined the company in February 2000, including serving as President of Research and Development from January 2007 until July 2010. From 1991 to 2000, Dr. Morrissey held several positions at Berlex Biosciences, last holding the position of Vice President, Discovery Research. Earlier in his career, Dr. Morrissey served as a Senior Scientist and Project Team Leader in Medicinal Chemistry at CIBA-Geigy Corporation. He is the author of numerous scientific publications in medicinal chemistry and drug discovery and an inventor on 70 issued U.S. patents and 25 additional published U.S. patent applications. Dr. Morrissey holds a B.S. (Honors) in Chemistry from the University of Wisconsin and a Ph.D. in Chemistry from Harvard University. |
Michael M. Morrissey, Ph.D12., age 55, has served as a director and as Exelixis’ President and Chief Executive Officer since July 2010. Dr. Morrissey has held positions of increasing responsibility at Exelixis, since he joined the company in February 2000, including serving as President of Research and Development from January 2007 until July 2010. From 1991 to 2000, Dr. Morrissey held several positions at Berlex Biosciences (subsequently acquired by Bayer AG in 2006), last holding the position of Vice President, Discovery Research. He is the author of numerous scientific publications in medicinal chemistry and drug discovery and an inventor on 70 issued U.S. patents and 25 additional published U.S. patent applications. Dr. Morrissey holds a B.S. (Honors) in Chemistry from the University of Wisconsin and a Ph.D. in Chemistry from Harvard University. Our Board has concluded that Dr. Morrissey should continue to serve as a director of Exelixis as of the date of thisInc.
Proposal 1 | Class III Directors
| | |
Stelios Papadopoulos, Ph.D. Co-Founder and Chairman of the Board, Exelixis, Inc. | | Director since 1994 Age 70 Key Qualifications and Expertise: Our Board concluded that Dr. Papadopoulos should continue to serve as a director of Exelixis due to his training as a scientist, his knowledge and experience with respect to the biotechnology, healthcare and pharmaceutical industries, his broad leadership experience resulting from extensive service on various boards, his knowledge and experience with respect to finance matters, and his ability to bring historic knowledge and continuity to the Board. Committee Assignments: • Audit Committee • Research & Development Committee Current Public Company Boards: • Biogen Inc. (Chair) • Regulus Therapeutics, Inc. (Chair) |
Stelios Papadopoulos, Ph.D., a co-founder of Exelixis, has been a director since December 1994 and the Chairman of the Board since January 1998. Dr. Papadopoulos retired as Vice Chairman of Cowen & Co., LLC in August 2006 after six years as an investment banker with the firm, where he focused on the biotechnology and pharmaceutical sectors. Prior to joining Cowen & Co., he spent 13 years as an investment banker at PaineWebber, Incorporated, where he was most recently Chairman of PaineWebber Development Corp., a PaineWebber subsidiary focusing on biotechnology. He joined PaineWebber in April 1987 from Drexel Burnham Lambert, where he was a Vice President in the Equity Research Department covering the biotechnology industry. Prior to Drexel, he was a biotechnology analyst at Donaldson, Lufkin & Jenrette. Before coming to Wall Street in 1985, Dr. Papadopoulos was on the faculty of the Department of Cell Biology at New York University Medical Center. He continues his affiliation with New York University Medical Center as an Adjunct Associate Professor of Cell Biology. Dr. Papadopoulos was a co-founder of Anadys Pharmaceuticals, Inc., a publicly-held biopharmaceutical company dedicated to improving patient care by developing novel medicines for the treatment of hepatitis C, acquired by Hoffmann-La Roche Inc. in November 2011. Dr. Papadopoulos served as a member of the board of directors of Anadys Pharmaceuticals from 2000 to 2011 and as its chairman in 2011, prior to its acquisition. Dr. Papadopoulos has also served as a member of the board of directors of three other publicly-held companies: Biogen, Inc., a biopharmaceutical company focused on the treatment of serious diseases, since 2008 and as its chairman since 2014; Regulus Therapeutics Inc., a biopharmaceutical company focused on the development of medicines targeting microRNAs, since 2008, and as its chairman since 2013; and BG Medicine, Inc., a diagnostics company focused on the development and commercialization of cardiovascular diagnostic tests, from 2003 until 2018. Dr. Papadopoulos was also co-founder and member of the board of directors of Cellzome Inc., a privately-held drug discovery company acquired by GlaxoSmithKline in May 2012. In the not-for-profit sector, Dr. Papadopoulos is a co-founder and Chairman of Fondation Santé, a member of the board of visitors of Duke Health, and a member of the Global Advisory Board of the Duke Institute for Health Innovation. Dr. Papadopoulos holds an M.S. in Physics, a Ph.D. in Biophysics and an M.B.A. in Finance, all from New York University. |
| | |
George A. Scangos, Ph.D. Chief Executive Officer, Vir Biotechnology, Inc. | | Director since 1996 Age 70 Key Qualifications and Expertise: Our Board concluded that Dr. Scangos should continue to serve as a director of Exelixis due to his prior leadership role as our President and Chief Executive Officer. Beyond his prior role as our principal executive officer, the Board also considered Dr. Scangos’ extensive qualifications, including his training as a scientist, his significant knowledge and experience with respect to the biotechnology, healthcare and pharmaceutical industries, his comprehensive leadership background resulting from service on various boards and as an executive in the biotechnology industry, and his ability to bring historic knowledge and continuity to the Board. Committee Assignments: • Research & Development Committee Current Public Company Boards: • Agilent Technologies, Inc. |
George A. Scangos, Ph.D., has been a director since October 1996. Since January 2017, Dr. Scangos has served as Chief Executive Officer and as a member of the board of directors of Vir Biotechnology, Inc., a privately held biotechnology company focused on fighting infectious diseases. From July 2010 to December 2016, Dr. Scangos served as Chief Executive Officer and as a member of the board of directors of Biogen Inc., a publicly-held biopharmaceutical company focused on the treatment of serious diseases. Prior to joining the Biogen organization, from October 1996 to July 2010, Dr. Scangos served as our President and Chief Executive Officer. From September 1993 to October 1996, Dr. Scangos served as President of Biotechnology at Bayer Corporation, a pharmaceutical company, and was responsible for research, business and process development, manufacturing, engineering, and quality assurance. Dr. Scangos has served as a member of the board of directors of various publicly-held companies, including: Agilent Technologies, Inc., a global leader in life sciences, diagnostics and applied chemical markets, since 2014; and Anadys Pharmaceuticals, Inc., a biopharmaceutical company dedicated to improving patient care by developing novel medicines for the treatment of hepatitis C, from 2003 to 2010. Dr. Scangos has also served as a member of the board of directors of our former subsidiary, TaconicArtemis GmbH (previously known as Artemis Pharmaceuticals GmbH), until 2010 and Entelos, Inc. from 1997 to 2010, and he currently serves as a director of Decibel Therapeutics, Inc. and Fondation Santé. Dr. Scangos previously served as the Chair of the California Healthcare Institute (CHI), as a member of the Board of the Global Alliance for TB Drug Development, and as a member of the board of directors of BayBio. Dr. Scangos is also a member of the Board of Advisors of the University of California, San Francisco School of Pharmacy, and the National Board of Advisors of the University of California, Davis School of Medicine. Dr. Scangos was a Jane Coffin Childs Post-Doctoral Fellow at Yale University and a faculty member at Johns Hopkins University. Dr. Scangos currently holds an appointment as Adjunct Professor of Biology at Johns Hopkins University. Dr. Scangos holds a B.A. in Biology from Cornell University and a Ph.D. in Microbiology from the University of Massachusetts. |
2019 Proxy Statement due to his leadership role as the President and Chief Executive Officer of Exelixis. Beyond his role as Exelixis’ principal executive officer, the Board also considered Dr. Morrissey’s extensive qualifications, including his training as a scientist, his significant knowledge and experience with respect to the biotechnology, healthcare and pharmaceutical industries, comprehensive leadership background resulting from service as an executive in the biotechnology industry, and his ability to bring historic knowledge and continuity to the Board.
Stelios Papadopoulos, Ph.D., age 67, a co-founder of Exelixis, has been a director since December 1994 and the Chairman of the Board since January 1998. Dr. Papadopoulos retired as Vice Chairman of Cowen & Co., LLC in August 2006 after six years as an investment banker with the firm, where he focused on the biotechnology and pharmaceutical sectors. Prior to joining Cowen & Co., he spent 13 years as an investment banker at PaineWebber, Incorporated, where he was most recently Chairman of PaineWebber Development Corp., a PaineWebber subsidiary focusing on biotechnology. He joined PaineWebber in April 1987 from Drexel Burnham Lambert, where he was a Vice President in the Equity Research Department covering the biotechnology industry. Prior to Drexel, he was a biotechnology analyst at Donaldson, Lufkin & Jenrette. Before coming to Wall Street in 1985, Dr. Papadopoulos was on the faculty of the Department of Cell Biology at New York University Medical Center. He continues his affiliation with New York University Medical Center as an Adjunct Associate Professor of Cell Biology. Dr. Papadopoulos is a co-founder of Anadys Pharmaceuticals, Inc., a publicly-held drug discovery and development company acquired by Hoffmann-La Roche Inc. in November 2011. Dr. Papadopoulos served as a member of the board of directors of Anadys Pharmaceuticals from 2000 to 2011 and as its chairman in 2011, prior to its acquisition. Dr. Papadopoulos has also served as a member of the board of directors of three other publicly-held companies: BG Medicine, Inc., a diagnostics company focused on the development and commercialization of cardiovascular diagnostic tests, since 2003; Biogen, Inc., a biopharmaceutical company focused on the treatment of serious diseases, since 2008 and as its chairman since 2014; and Regulus Therapeutics Inc., a biopharmaceutical company focused on the development of medicines targeting microRNAs, since 2008, and as its chairman since 2013. Dr. Papadopoulos is a member of the board of directors of a privately-held biotechnology company, Joule Unlimited, Inc. Dr. Papadopoulos was also co-founder and member of the board of directors of Cellzome Inc., a privately-held drug discovery company acquired by GlaxoSmithKline in May 2012. In the not-for-profit sector, Dr. Papadopoulos is a co-founder and Chairman of Fondation Santé, a member of the board of visitors of Duke
University Medical Center, and a member of the Global Advisory Board of the Duke Institute for Health Innovation. Dr. Papadopoulos holds a Ph.D. in Biophysics and an M.B.A. in Finance, both from New York University. Our Board has concluded that Dr. Papadopoulos should continue to serve as a director of Exelixis as of the date of this Proxy Statement due to his training as a scientist, his knowledge and experience with respect to the biotechnology, healthcare and pharmaceutical industries, his broad leadership experience resulting from extensive service on various boards, his knowledge and experience with respect to finance matters, and his ability to bring historic knowledge and continuity to the Board.
| | |
Lance Willsey, M.D. Founding Partner, DCF Capital | | Director since 1997 Age 57 Key Qualifications and Expertise: Our Board concluded that Dr. Willsey should continue to serve as a director of Exelixis due to his skill as a physician, his knowledge and experience with respect to the life sciences and healthcare industries, and his knowledge and experience with respect to finance matters. Committee Assignments: • Compensation Committee • Research & Development Committee |
Lance Willsey, M.D., has been a director since April 1997. Dr. Willsey was a founding partner of DCF Capital, a hedge fund focused on investing in the life sciences, from July 1998 to March 2002, and currently is a consultant to institutional investors in the field of oncology. Since 2000, Dr. Willsey has served on the Visiting Committee of the Department of Genitourinary Oncology at the Dana-Farber Cancer Institute at Harvard Medical School. From July 1997 to July 1998, Dr. Willsey served on the Staff Department of Urologic Oncology at the Dana-Farber Cancer Institute. From July 1996 to July 1997, Dr. Willsey served on the Staff Department of Urology at Massachusetts General Hospital at Harvard University School of Medicine, where he was a urology resident from July 1992 to July 1996. From 2000 to 2010, Dr. Willsey served a member of the board of directors of Exact Sciences Corporation, a publicly-held molecular diagnostics company focused on the early detection and prevention of the deadliest forms of cancer. Dr. Willsey holds a B.S. in Physiology from Michigan State University and an M.S. in Biology and an M.D., both from Wayne State University. |
George A. Scangos, Ph.D., age 67, has been a director since October 1996. Since July 2010, Dr. Scangos has served as Chief Executive Officer and as a member of the board of directors of Biogen, Inc. From October 1996 to July 2010, Dr. Scangos served as our President and Chief Executive Officer. From September 1993 to October 1996, Dr. Scangos served as President of Biotechnology at Bayer Corporation, a pharmaceutical company, and was responsible for research, business and process development, manufacturing, engineering, and quality assurance. Dr. Scangos has served as a member of the board of directors of various publicly-held companies, including Anadys Pharmaceuticals, Inc. from 2003 to 2010 and Entelos, Inc. from 1997 to 2010. Dr. Scangos also served as a member of the board of directors of our former subsidiary, TaconicArtemis GmbH (previously known as Artemis Pharmaceuticals GmbH) until 2010. Dr. Scangos previously served as the Chair of the California Healthcare Institute (CHI), as a member of the Board of the Global Alliance for TB Drug Development, and as a member of the board of directors of BayBio. Dr. Scangos currently serves as a director of Agilent Technologies, Inc. and Fondation Santé. Dr. Scangos is also a member of the Board of Advisors of the University of California, San Francisco School of Pharmacy, and the National Board of Advisors of the University of California, Davis School of Medicine. Dr. Scangos was a Jane Coffin Childs Post-Doctoral Fellow at Yale University and a faculty member at Johns Hopkins University. Dr. Scangos currently holds an appointment as Adjunct Professor of Biology at Johns Hopkins University. Dr. Scangos holds a B.A. in Biology from Cornell University and a Ph.D. in Microbiology from the University of Massachusetts. Our Board has concluded that Dr. Scangos should continue to serve as a director of Exelixis as of the date of this Proxy Statement due to his prior leadership role as our President and Chief Executive Officer. Beyond his prior role as our principal executive officer, the Board also considered Dr. Scangos’ extensive qualifications, including his training as a scientist, his significant knowledge and experience with respect to the biotechnology, healthcare and pharmaceutical industries, his comprehensive leadership background resulting from service on various boards and as an executive in the biotechnology industry, and his ability to bring historic knowledge and continuity to the Board.
Lance Willsey, M.D., age 54, has been a director since April 1997. Dr. Willsey was a founding partner of DCF Capital, a hedge fund focused on investing in the life sciences, from July 1998 to March 2002, and currently is a consultant to institutional investors in the field of oncology. Since 2000, Dr. Willsey has served on the Visiting Committee of the Department of Genitourinary Oncology at the Dana Farber Cancer Institute at Harvard University School of Medicine. From July 1997 to July 1998, Dr. Willsey served on the Staff Department of Urologic Oncology at the Dana Farber Cancer Institute. From July 1996 to July 1997, Dr. Willsey served on the Staff Department of Urology at Massachusetts General Hospital at Harvard University School of Medicine, where he was a urology resident from July 1992 to July 1996. From 2000 to 2010, Dr. Willsey served a member of the board of directors of Exact Sciences Corporation, a publicly-held biotechnology company. Dr. Willsey holds a B.S. in Physiology from Michigan State University and an M.S. in Biology and an M.D., both from Wayne State University. Our Board has concluded that Dr. Willsey should continue to serve as a director of Exelixis as of the date of this Proxy Statement due to his skill as a physician, his knowledge and experience with respect to the life sciences and healthcare industries, and his knowledge and experience with respect to finance matters.
Class I Directors
Continuing in Office Until the 20182021 Annual Meeting
| | |
Charles Cohen, Ph.D. Chief Executive Officer, Perform Biologics, Inc. | | Director since 1995 Age 68 Key Qualifications and Expertise: Our Board concluded that Dr. Cohen should continue to serve as a director of Exelixis due to his training as a scientist, his knowledge and experience with respect to the biotechnology, pharmaceutical and healthcare industries, his broad leadership experience resulting from service on various boards and as a chief executive officer, and his knowledge and experience with respect to finance matters. Committee Assignments: • Audit Committee • Compensation Committee (chair) • Research & Development Committee |
Charles Cohen, Ph.D., has been a director since November 1995. Dr. Cohen is an independent investor and since March 2015, has served as Chief Executive Officer and as a member of the board of directors of Perform Biologics, Inc., a private biotechnology start-up company. Previously, Dr. Cohen served as Chief Executive Officer and as a member of the board of directors of On Target Therapeutics, LLC, a private biotechnology start-up company, from June 2015 to June 2017. From May 2007 to December 2015, Dr. Cohen was a managing director of Synthesis Capital (formerly Advent Healthcare Ventures), a venture capital firm. From 2003 to 2007, Dr. Cohen was Vice President of Advent International, a global private equity firm. From 2000 to 2002, Dr. Cohen was the Chief Executive Officer of Cellzome AG, a post-genomics biotechnology company. Prior to that time, Dr. Cohen co-founded Creative BioMolecules, Inc., a biotechnology company, in 1982 and was one of its directors and its Chief Executive Officer from 1985 to 1995. Dr. Cohen served as a member of the board of directors of the following publicly-held companies: Anadys Pharmaceuticals, Inc., a biopharmaceutical company dedicated to improving patient care by developing novel medicines for the treatment of hepatitis C, from 2000 to 2005; and Anesiva, Inc., a biopharmaceutical company focused on the development and commercialization of novel pharmaceutical products for pain management from 2005 to 2007. Dr. Cohen also served as the Chairman of the Supervisory Board of Cellzome AG, prior to its acquisition by GlaxoSmithKline in May 2012, and as the Chief Executive Officer of several other companies. Dr. Cohen received his Ph.D. from New York University School of Medicine. |
14 age 65, has been a director since November 1995. Dr. Cohen is an independent investor and the Chief Executive Officer of two biotechnology start-up companies, On Target Therapeutics, LLC and Perform Biologics,Exelixis, Inc. From May 2007 to December 2015, Dr. Cohen was a managing director of Synthesis Capital (formerly Advent Healthcare Ventures), a venture capital firm. From 2003 to 2007, Dr. Cohen was Vice President of Advent International, a global private equity firm. From 2000 to 2002, Dr. Cohen was the Chief Executive Officer of Cellzome AG, a post-genomics biotechnology company. Prior to that time, Dr. Cohen co-founded Creative BioMolecules, Inc., a biotechnology company, in 1982 and was one of its directors and its Chief Executive Officer from 1985 to 1995. Dr. Cohen served as a member of the board of directors of the following publicly-held biopharmaceutical companies: Anadys Pharmaceuticals, Inc., from 2000 to 2005, and Anesiva, Inc., from 2005 to 2007. Dr. Cohen serves on the board of directors and as a Chief Executive Officer of several Advent Healthcare Ventures privately-held portfolio companies. Dr. Cohen also served as the Chairman of the Supervisory Board of Cellzome AG, prior to its acquisition by GlaxoSmithKline in May 2012, and as the Chief Executive Officer of several other companies. Dr. Cohen received his Ph.D. from New York University School of Medicine. Our Board has concluded that Dr. Cohen should continue to serve as a director of Exelixis as of the date of this Proxy Statement due to his training as a scientist, his knowledge and experience with respect to the biotechnology, pharmaceutical and healthcare industries, his broad leadership experience resulting from service on various boards and as a chief executive officer, and his knowledge and experience with respect to finance matters.
George Poste, D.V.M., Ph.D., FRS,Proposal 1 | age 71, has been a director since August 2004. Since February 2009, Dr. Poste has been the Chief Scientist at Complex Adaptive Systems Initiative and Regents’ Professor and Del E. Webb Professor of Health Innovation at Arizona State University. From May 2003 to February 2009, Dr. Poste served as the director of the Biodesign Institute at Arizona State University. Dr. Poste has served as the Chief Executive Officer of Health Technology Networks, a consulting company that specializes in the application of genomic technologies and computing in healthcare, since 2000. From 1992 to 1999, he was the Chief Science and Technology Officer and President, R&D, of SmithKline Beecham Corporation, a pharmaceutical company. Dr. Poste served on the Defense Science Board of the U.S. Department of Defense from 2001 to 2010 and is a member of other organizations dedicated to advancing defenses against bioweapons and biowarfare. Since February 2003, Dr. Poste has served as a member of the board of directors of Monsanto Company, a publicly-held provider of agricultural products and solutions. From April 2000 until August 2009, Dr. Poste served as the Non-Executive Chairman of Orchid Cellmark, Inc., a publicly-held DNA forensics company. Dr. Poste currently serves as a Board Member of Caris Life Sciences, a privately held medical diagnostics company. Dr. Poste is a Fellow of the Royal Society, the UK Academy of Medical Sciences, Hoover Institution, Stanford University, and various other prestigious organizations and has been awarded honorary doctorates from several universities. Dr. Poste holds a D.V.M. in veterinary medicine and a Ph.D. in Virology from the University of Bristol, England. Our Board has concluded that Dr. Poste should continue to serve as a director of Exelixis as of the date of this Proxy Statement due to his training as a scientist, his knowledge and experience with respect to the life sciences, healthcare and pharmaceutical industries, his broad leadership experience resulting from service on various boards, and his knowledge and experience with policymaking, regulatory issues and other governmental matters.Class III Directors
| | |
George Poste, DVM, Ph.D., FRS Chief Scientist, Complex Adaptive Systems Initiative | | Director since 2004 Age 74 Key Qualifications and Expertise: Our Board concluded that Dr. Poste should continue to serve as a director of Exelixis due to his training as a scientist, his knowledge and experience with respect to the life sciences, healthcare and pharmaceutical industries, his broad leadership experience resulting from service on various boards, and his knowledge and experience with policymaking, regulatory issues and other governmental matters. Committee Assignments: • Nominating and Corporate Governance Committee • Research & Development Committee (chair) |
George Poste, DVM, Ph.D., FRS, has been a director since August 2004. Since February 2009, Dr. Poste has been the Chief Scientist at Complex Adaptive Systems Initiative and Regents’ Professor and Del E. Webb Professor of Health Innovation at Arizona State University. From May 2003 to February 2009, Dr. Poste served as the director of the Biodesign Institute at Arizona State University. Dr. Poste has served as the Chief Executive Officer of Health Technology Networks, a consulting company that specializes in the application of genomic technologies and computing in healthcare, since 2000. From 1992 to 1999, he was the Chief Science and Technology Officer and President, R&D, of SmithKline Beecham Corporation, a pharmaceutical company (later merged into GlaxoSmithKline plc). Dr. Poste served on the Defense Science Board of the U.S. Department of Defense from 2001 to 2010 and is a member of other organizations dedicated to advancing defenses against bioweapons and biowarfare. Dr. Poste has served as a member of the board of directors of Caris Life Sciences, a privately-held medical diagnostics company, since 2009. Previously, Dr. Poste served as a member of the board of directors of Monsanto Company, a publicly-held provider of agricultural products and solutions, from February 2003 until its acquisition by Bayer Aktiengesellschaft in June 2018, and as the Non-Executive Chairman of Orchid Cellmark, Inc., a publicly-held DNA forensics company, from April 2000 until August 2009. Dr. Poste is a Fellow of the Royal Society, the UK Academy of Medical Sciences, Hoover Institution, Stanford University, and various other prestigious organizations and has been awarded honorary doctorates from several universities. Dr. Poste holds a DVM in veterinary medicine and a Ph.D. in Virology from the University of Bristol, England. |
| | |
Jack L. Wyszomierski Former Executive Vice President and Chief Financial Officer, VWR International, LLC | | Director since 2004 Age 63 Key Qualifications and Expertise: Our Board concluded that Mr. Wyszomierski should continue to serve as a director of Exelixis due to his extensive financial reporting, accounting and finance experience, as well as his experience in the healthcare and life sciences industries. These qualities have also formed the basis for the Board’s decision to appoint Mr. Wyszomierski as a member and chairman of the Audit Committee. Committee Assignments: • Audit Committee (chair) • Nominating and Corporate Governance Committee Current Public Company Boards: • XOMA Corporation • Athersys, Inc. • SiteOne Landscape Supply, Inc. |
Jack L. Wyszomierski, has been a director since February 2004. From June 2004 to June 2009, Mr. Wyszomierski served as the Executive Vice President and Chief Financial Officer of VWR International, LLC, a supplier of laboratory supplies, equipment and supply chain solutions to the global research laboratory industry. From 1982 to 2003, Mr. Wyszomierski held positions of increasing responsibility within the finance group at Schering-Plough Corporation, a health care company, culminating with his appointment as Executive Vice President and Chief Financial Officer in 1996. Prior to joining Schering-Plough, he was responsible for capitalization planning at Joy Manufacturing Company, a producer of mining equipment, and was a management consultant at Data Resources, Inc. Mr. Wyszomierski has served: as a member of the board of directors of XOMA Corporation, a publicly-held company that licenses novel antibody therapeutics, since August 2010; as a member of the board of directors of Athersys, Inc., a publicly-held company engaged in the discovery and development of therapeutic product candidates, since June 2010; as a member of the board of directors of Solenis LLC, a privately-held manufacturer of chemical products, since August 2014; and as a member of the board of directors of SiteOne Landscape Supply, Inc., a publicly-held company that distributes landscape supply products, since April 2016. Mr. Wyszomierski previously served as a member of the board of directors of: Unigene Laboratories, Inc., a publicly-held biopharmaceutical company, from April 2010 to July 2013; and AssuraMed Holding, Inc., a privately-held distributor of home healthcare products, from January 2011 until its acquisition by Cardinal Health Inc. in March 2013. Mr. Wyszomierski holds a M.S. in Industrial Administration and a B.S. in Administration, Management Science and Economics from Carnegie Mellon University. |
Jack L. Wyszomierski, age 60, has been a director since February 2004. From June 2004 to June 2009, Mr. Wyszomierski served as the Executive Vice President and Chief Financial Officer of VWR International, LLC, a supplier of laboratory supplies, equipment and supply chain solutions to the global research laboratory industry. From 1982 to 2003, Mr. Wyszomierski held positions of increasing responsibility within the finance group at Schering-Plough Corporation, a health care company, culminating with his appointment as Executive Vice President and Chief Financial Officer in 1996. Prior to joining Schering-Plough, he was responsible for capitalization planning at Joy Manufacturing Company, a producer of mining equipment, and was a management consultant at Data Resources, Inc. Mr. Wyszomierski has served: as a member of the board of directors of XOMA Ltd., a publicly-held company that discovers, develops and manufactures novel antibody therapeutics, since August 2010; as a member of the board of directors of Athersys, Inc., a publicly-held company engaged in the discovery and development of therapeutic product candidates, since June 2010; and as a member of the board of directors of Solenis, LLC, a privately-held manufacturer of chemical products, since August 2014. Mr. Wyszomierski previously served as a member of the board of directors of: Unigene Laboratories, Inc., a publicly-held biopharmaceutical company, from April 2010 to July 2013; and AssuraMed Holding, Inc., a privately-held distributor of home healthcare products, from January 2011 until its acquisition by Cardinal Health Inc. in March 2013. Mr. Wyszomierski holds a M.S. in Industrial Administration and a B.S. in Administration, Management Science and Economics from Carnegie Mellon University. Our Board has concluded that Mr. Wyszomierski should continue to serve as director of Exelixis as of the date of this Proxy Statement due to his extensive financial reporting, accounting and finance experience, as well as his experience in the healthcare and life sciences industries. These qualities have also formed the basis for the Board’s decision to appoint Mr. Wyszomierski as a member and chairman of the Audit Committee.
Corporate Governance Guidelines
We have adopted written corporate governance guidelines,Corporate Governance Guidelines, which may be viewed atwww.exelixis.com under the caption “Investors & Media-Corporate Governance.Media—Corporate Governance—Corporate Governance Documents.” This document includes guidelines for determining director independence and qualifications for directors. Our Board regularly reviews, and modifies from time to time, our corporate governance guidelines,Corporate Governance Guidelines, Board committee charters and Board practices. Please note that information found on, or accessible through, our website is not a part of, and is not incorporated into, this Proxy Statement.
2019 Proxy Statement 15
Corporate Code of Conduct and Ethics
We have adopted a Corporate Code of Conduct, andwhich functions as our Code of Ethics under the SEC rules, that applies to all directors, officers and employees, including the principal executive officer principal financial officer and principal financial/accounting officer. The Corporate Code of Conduct and Ethics is posted on our website atwww.exelixis.com under the caption “Investors & Media-Corporate Governance.Media—Corporate Governance—Corporate Governance Documents.” We intend to satisfy the disclosure requirement under Item 5.05 of Form 8-K regarding an amendment to, or waiver from, a provision of this Corporate Code of Conduct and Ethics by posting such information on our website, at the address and location specified above and, to the extent required by the listing standards of the NASDAQNasdaq Stock Market, by filing a Current Report on Form 8-K with the SEC, disclosing such information.
Our Corporate Code of Conduct reflects our corporate values and describes how our officers, directors, employees and contractors are expected to conduct themselves when representing Exelixis. It also underscores our commitment to comply with laws that regulate our business activities as a biotechnology company. Our employees receive regular training on our Corporate Code of Conduct, which includes consequences of taking actions that would constitute a violation of our Corporate Code of Conduct.
Included in our Corporate Code of Conduct are procedures for employees to report potential violations to our Ethics Committee, which is chaired by our Chief Executive Officer and includes other members of our senior management team. To ensure our employees feel comfortable raising good faith questions or concerns with respect to our Corporate Code of Conduct or our other policies, these reports can be made confidentially (or anonymously) via our Ethics Hotline, and we maintain a strict policy against any retaliation or discrimination towards an employee who makes such a report.
Director Independence
We have adopted standards for director independence pursuant to NASDAQNasdaq listing standards, which require that a majority of the members of a listed company’s board of directors qualify as “independent,” as affirmatively determined by the
board of directors. An “independent director” means a person other than an officer or employee of Exelixis or one of our subsidiaries, or another individual having a relationship that, in the opinion of the Board, would interfere with the exercise of independent judgment in carrying out the responsibilities of a director. Consistent with these considerations, after review of all relevant transactions or relationships between each director, or any of his or her family members, and Exelixis, its senior management and its independent registered public accounting firm, the Board has affirmatively determined that Drs. Cohen, Freire, Garber, Marchesi, Papadopoulos, Poste, Scangos and Willsey, and Messrs. Wyszomierski and Feldbaum and Ms. Smith, who are nineeleven of the tentwelve members of the Board, represent a majority of the Board and are independent. In making this determination,independent under applicable SEC rules and the Board considered Dr. Cohen’s less than 10% ownership interest in Akarna Therapeutics, Ltd., or Akarna. In 2015, Exelixis entered into an asset purchase agreement pursuant with Akarna pursuant to which Akarna acquired certain assets, technologies and intellectual property relating to one of Exelixis’ terminated programs. After review of this relationship, the Board found that Dr. Cohen did not have a material or other disqualifying relationship with Exelixis.Nasdaq listing standards. Dr. Morrissey, our President and Chief Executive Officer, is not independent by virtue of his employment with Exelixis. In addition, the Board has also determined that: (i) all directors who serve on the Audit, Compensation and Nominating and Corporate Governance Committees are independent under applicable NASDAQNasdaq listing standards; and (ii) all members of the Audit Committee meet the independence requirements under the Securities Exchange Act of 1934, as amended.
Act.Board Leadership Structure
The Board does not have a formal policy on whether the role of chairman and chief executive officer should be separate or combined. Our corporate governance guidelinesCorporate Governance Guidelines provide that the Board will select its chairman and the chief executive officer in the manner it considers to be in the best interests of our company and those of our stockholders.company. Currently, we have an independent chairman of the board separate from the chief executive officer. The Board believes this bifurcated structure provides for sufficient independent oversight of management and strong Board leadership, while allowing for the effective management of company affairs. The Board believes that if the positions of chairman and chief executive officer are combined, the appointment of a lead independent director would be necessary for effective governance. Accordingly, our corporate governance guidelinesCorporate Governance Guidelines provide that if the roles are combined, the independent directors of the Board must appoint a lead independent director. Our corporate governance guidelinesCorporate Governance Guidelines further provide that the lead independent director would: (i) preside at all meetings of the Board at which the chairman is not present, including executive sessions of the independent directors; (ii) have the authority to call meetings of the independent directors; (iii) serve as the principal liaison on Board-wide issues between the independent directors and the chairman; and (iv) have such other authority and duties as the Board may from time to time determine. The Board believes that this flexible approach provides it with the ability to establish a leadership structure, based upon its judgment that is in the best interests of our company and those of our stockholders at any given time.
16 Exelixis, Inc.
Proposal 1 | Corporate Governance
Role of the Board in Risk Oversight
Management is responsible for identifying the various risks facing our company, including, without limitation, strategic, operational, financial, regulatory and regulatorycyber-security risks that may exist from time to time. Management is also charged with the responsibility of implementinghas implemented appropriate risk management structures, policies and procedures, and managingmanages our risk exposure on a day-to-day basis. While we dothe Board does not have a formal risk oversight policy, the Board does, as a whole and through its various committees, conductsoversee the proper functioning of our internal risk oversight function for our company.management processes. In its risk oversight role, the Board evaluates whether management has reasonable controls in place to address material risks currently facing our company and those we may face in the future. The Board and its committees meet at regularly scheduled and special meetings throughout the year at which theymanagement reports to the Board concerning the results of its risk management activities, as well as external changes that may change the levels of business risk to which we are presented with information regarding risks facing the company. exposed.
The Board delegates certain of its risk oversight responsibilities to its various committees as follows:
| ›› | Our Audit Committee oversees the management of risks related to financial reporting, trading in our securities, fraud, information technology and cyber-security, including a regular review of the existence or status of any such risks and evaluating the steps management has taken or proposes to take to mitigate them. Our Audit Committee also reviews any proposed related party transactions to ensure we do not engage in transactions that would create a conflict of interest or result in harm to us. |
| ›› | Our Compensation Committee oversees the management of risks related to our compensation policies and practices, including structuring and reviewing our executive compensation programs, considering whether such programs are in line with our strategic objectives and incentivizing appropriate risk-taking. |
| ›› | Our Nominating and Corporate Governance Committee oversees the management of risks related to our compliance programs, including healthcare compliance and data privacy, and corporate governance matters, including director independence and Board composition and succession. |
Also, the Board is presented with frequent business updates during monthly teleconferences among our Board and senior management. Following consideration of the information provided by management, the Board provides feedback, makes recommendations and, as needed, issues directives to management to address our risk exposure.
Prohibitions on Derivative, Hedging, Monetization and Other Transactions
We maintain an insider trading policy that applies to directors and employees, including our executive officers, which prohibits certain transactions in our stock, including short sales, puts, calls or other transactions involving derivative securities, hedging or monetization transactions, purchases of Exelixis securities on margin or borrowing against an account in which Exelixis securities are held, or pledging Exelixis stock as collateral for a loan.
There are no exceptions to these prohibitions, other than the pledging of Exelixis stock as collateral for a loan, which would require that such director or employee establish his or her financial capacity to repay the loan without resort to the pledged securities and obtains prior approval from management.Stockholder Communications with the Board
Security holders may send communications to the Board by mail at 210 East Grand Avenue, South San Francisco,1851 Harbor Bay Parkway, Alameda, California 94080,94502, by facsimile at(650) 837-7951 or by e-mail at info@exelixis.com, each of the foregoing sent “Attn: Board of Directors.”
Stock Ownership Guidelines for Non-Employee Directors
In February 2018, the Board adopted Stock Ownership Guidelines applicable to our directors and Named Executive Officers (as defined below) to further align their financial interests with those of our stockholders, as well as to promote sound corporate governance. For our non-employee directors, our Stock Ownership Guidelines provide an ownership target equal to the lesser of 3,000 shares or a value equivalent to three times the annual cash Board retainer. Under the guidelines, we expect non-employee directors to achieve their stock ownership targets within five years of becoming subject to these guidelines. The policy includes procedures for granting exemptions in the case of severe financial hardship. Ownership targets for our Named Executive Officers (including those serving on our Board) are described below under “Compensation Discussion and Analysis—Other Compensation Information—Stock Ownership Guidelines.”
2019 Proxy Statement 17
In determining ownership levels for each non-employee director under our Stock Ownership Guidelines, credit is provided for shares held outright (including shares owned through trusts, the Amended and Restated Exelixis, Inc. 401(k) Plan (401(k) Plan), or by a spouse), as well as 50% of the number of vested, but unexercised, stock options. No credit is provided for RSUs (as defined below) until they vest. The values for all shares determined to be held by our non-employee directors and Named Executive Officers are based on the 200-day average stock price as of the measurement date. As of February 12, 2019, all of our non-employee directors serving on the Board as of such date had met the required ownership targets.
Board Committees and Meetings
The Board held sixfive meetings and acted by unanimous written consent one time during 20152018, and all of our directors attended at least 75% of the total meetings of the Board and of the committees on which they served. The independent directors met four times in regularly scheduled executive sessions.
During 2015,2018, the Board had four standing committees: anthe Audit Committee,Committee; the Compensation Committee,Committee; the Nominating and Corporate Governance CommitteeCommittee; and the Research and& Development Committee. Committee membership in 20152018 was as follows:
|
| | | | | | | | |
| Board Member | | Audit Committee | | Compensation Committee | | Nominating and Corporate Governance Committee | | Research and Development Committee |
| Charles Cohen, Ph.D. | | Member | | Chair | | | | |
| Carl B. Feldbaum, Esq. | | | | | | Member | | |
| Alan M. Garber, M.D., Ph.D. | | | | | | Chair | | |
| Vincent T. Marchesi, M.D., Ph.D. | | | | Member | | | | Member |
| Stelios Papadopoulos, Ph.D. | | Member | | | | | | |
| George Poste, D.V.M., Ph.D., FRS | | | | | | Member | | Chair |
| George A. Scangos, Ph.D. | | | | | | | | |
| Lance Willsey, M.D. | | | | Member | | | | Member |
| Jack L. Wyszomierski | | Chair* | | | | | | |
| Number of Meetings Held in Fiscal 2015 | | 5 | | 13 | | 2 | | 2 |
*Designated by the Board as an “audit committee financial expert.”
| | | | | | | | |
Board Member | | Audit
Committee | | Compensation Committee | | Nominating
and Corporate
Governance
Committee | | Research & Development
Committee |
Charles Cohen, Ph.D. | | Member | | Chair | | | | Member |
Carl B. Feldbaum, Esq. | | | | Member | | Member | | |
Maria C. Freire, Ph.D.* | | | | | | Member | | Member |
Alan M. Garber, M.D., Ph.D. | | | | | | Chair | | Member |
Vincent T. Marchesi, M.D., Ph.D. | | | | Member | | | | Member |
Stelios Papadopoulos, Ph.D. | | Member | | | | | | Member |
George Poste, D.V.M., Ph.D., FRS | | | | | | Member | | Chair |
George A. Scangos, Ph.D. | | | | | | | | Member |
Julie A. Smith** | | Member | | Member | | | | |
Lance Willsey, M.D. | | | | Member | | | | Member |
Jack L. Wyszomierski | | Chair*** | | | | Member | | |
Number of Meetings Held in Fiscal 2018 | | 4 | | 10 | | 3 | | 3 |
| * | Dr. Freire became a director and was appointed to the Nominating and Corporate Governance Committee and the Research & Development Committee on April 5, 2018. |
| ** | On September 5, 2018, Ms. Smith moved from the Nominating and Corporate Governance Committee to the Audit Committee. |
| *** | Designated by the Board as an “audit committee financial expert.” |
Audit Committee
The Audit Committee of the Board oversees our corporate accounting and financial reporting process, ensures the integrity of our financial statements and has been designated as the Qualified Legal Compliance Committee within the meaning of Rule 205.2(k) of Title 17, Chapter II of the Code of Federal Regulations. The Audit Committee is composed entirely of independent directors and performs several functions, such as evaluatingincluding:
| | |
• Evaluating the performance, qualifications, compensation and continued engagement of the independent registered public accounting firm, as well as resolving any disagreements between the independent registered public accounting firm and management | | • Reviewing the sufficiency of our information technology resources and overseeing our program to identify, assess, manage and monitor cyber-security risks |
18 Exelixis, Inc.
Proposal 1 | Board Committees and assessing the qualifications of, the independent registered public accounting firm; determining whether to retain or terminate the existing independent registered public accounting firm or to appoint and engage a new independent registered public accounting firm; reviewing and approving the engagement of the independent registered public accounting firm to perform any proposed permissible services and appropriate compensation thereof; reviewing, providing oversight of, and approving related party transactions; establishing procedures, as required under applicable law, for the receipt, retention and treatment of complaints received by Exelixis regarding accounting, internal accounting controls or auditing matters and the confidential and anonymous submission by employees of concerns regarding questionable accounting or auditing matters; reviewing the financial statements to be included in our Annual Report on Form 10-K; discussing with management and the independent registered public accounting firm the results of the annual audit and the results of our quarterly financial statement reviews; and resolving any disagreements between the independent registered public accounting firm and management. The Audit Committee also has the specific responsibilities and authority necessary to comply with the listing standards of the NASDAQ Stock Market applicable to audit committees.
Meetings | | |
• Reviewing, providing oversight of, and approving related person transactions | | • Reviewing the financial statements for inclusion in our Annual Report on Form 10-K and preparing the Audit Committee’s report for inclusion in our Proxy Statement or Annual Report on Form 10-K |
| |
• Establishing procedures to receive and address complaints regarding accounting, internal accounting controls or auditing matters | | • Reviewing the results of the annual audit and the results of our quarterly financial statement reviews with management and the independent registered public accounting firm |
| |
• Overseeing our risk assessment and risk management processes with respect to financial reporting fraud and discussing and evaluating the steps management has taken or proposes to take to mitigate such risks | | • Maintaining compliance with SEC and Nasdaq rules applicable to audit committees |
| |
• Reviewing our tax strategy, the status of any material tax audits and proceedings and any other material tax matters | | |
The Board has determined that Mr. Wyszomierski is an “audit committee financial expert” as defined in applicable SEC rules.
The Audit Committee’s report is set forth in “Report of the Audit Committee” below. The Audit Committee has a written charter, which is available on our website atwww.exelixis.com under the caption “Investors & Media-Corporate Governance.Media—Corporate Governance—Committee Composition and Charters.”
The purposeCompensation Committee of the Compensation Committee is to: overseeBoard oversees our compensation policies, plans and programs; review and determine the compensation to be paid to officers and directors; work with management to address any conflict of interest with any compensation adviser engaged by management or the Compensation Committee; review with management our Compensation Discussion and Analysis, and to consider whether to recommend that it be included in our proxy statements and other filings; and prepare and review the Compensation Committee’s report included in our annual proxy statement or Annual Report on Form 10-K, as applicable, in accordance with applicable rules and regulations of the SEC.programs. The Compensation Committee reviews and recommends to the Board the compensation and benefitsis composed entirely of all officers, establishes and reviews general policies relating to compensation and benefits of employees, including executive officers,independent directors and performs such otherseveral functions,
regarding compensation as the Board may delegate. The Compensation Committee also administers the issuance of stock options and other awards under our stock plans.
including: | | |
• Reviewing and determining the compensation to be paid to executive officers | | • Reviewing our Compensation Discussion and Analysis and preparing the Compensation Committee’s report for inclusion in our Proxy Statement |
| |
• Addressing any conflict of interest with any compensation adviser engaged by management or the Compensation Committee | | • Establishing general policies relating to compensation and benefits of employees, including executive officers |
| |
• Evaluating director compensation and recommending any changes to the Board for approval | | • Administering the issuance of stock options and other awards under our stock plans |
The Compensation Committee’s report is set forth in “Compensation Committee Report” below. Information on the Compensation Committee’s processes and procedures for consideration of executive compensation are addressed in “Compensation Discussion and Analysis” below. For information regarding our processes and procedures for the consideration and determination of director compensation, please see “Compensation of Directors” below. The Compensation Committee has a written charter, which is available on our website atwww.exelixis.com under the caption “Investors & Media -Corporate Governance.Media—Corporate Governance—Committee Composition and Charters.” In accordance with its charter, the Compensation Committee also may delegate any of its authority or responsibility to the chairperson of the Compensation of the Committee or to a subcommittee composed of one or more members of the Compensation Committee and/or other members of the Board and/or officers of Exelixis.
Compensation Consultants.ManagementThe Compensation Committee retained Radford,Compensia, Inc. (Compensia), a national compensation consulting firm, serving technology and life sciences companies, as its external compensation consultant to assist withthe Compensation Committee in its duties related to executive and non-employee director compensation. Management retained Radford principally to provide benchmarkCompensia did not perform any work for us other than the executive compensation and industrynon-employee director compensation data for our senior management. For more detailed information regarding theconsulting services provided by Radford, please see “Compensation of Directors”to the Compensation Committee and
2019 Proxy Statement 19
reported directly to the Compensation Committee or through its chairperson. Fees paid to Compensia in 2018 did not exceed $120,000. See “Compensation Discussion and Analysis” below. In March 2015,for more information regarding the Compensation Committee assessed the independence of Radford pursuant to SEC rules and concluded that theCommittee’s engagement of Radford did not raise any conflicts of interest that would prevent Radford from serving as an independent consultant to management during 2015. The Compensation Committee performed a similar assessment of Radford in March 2016 and concluded that the engagement of Radford did not raise any conflicts of interest that would prevent Radford from continuing to serve as an independent consultant to management during 2016. In connection with each assessment, Radford provided a certification to the Compensation Committee regarding its independence and provision of services. Radford is an Aon Hewitt company and an affiliate of Aon plc which has provided insurance brokerage services to Exelixis since 2012. Aggregate fees paid to Aon Hewitt and its affiliates for 2015 did not exceed $120,000.
Compensia.Nominating and Corporate Governance Committee
The purpose of the Nominating and Corporate Governance Committee is to: overseeof the Board oversees all aspects of our corporate governance functions on behalf of the Board; make recommendations to the Board regarding corporate governance issues; identify, review and evaluate candidates to serve as directors; serve as a focal point for communication between such candidates, non-committee directors and management; recommend such candidates to the Board and make such other recommendations to the Board regarding affairs relating to the directors; and develop a set of corporate governance principles for Exelixis.Board. The Nominating and Corporate Governance Committee also oversees our healthcare compliance programs created to prevent fraudis composed entirely of independent directors and abuse in federal healthcare programs and non-compliance with applicable healthcare laws. performs several functions, including:
| | |
• Conducting periodic assessments of the performance of the Board and its committees and compliance with SEC and Nasdaq requirements for independence and expertise | | • Facilitating our CEO Succession Plan in the event our Chief Executive Officer is no longer able to serve in that position |
| |
• Identifying, reviewing and evaluating candidates to serve as directors and recommending qualified candidates to the Board | | • Developing a set of Corporate Governance Guidelines, as well as administering our Corporate Code of Conduct |
| |
• Ensuring effective communication between the Board, its committees and management, as well as receiving and reviewing communications from security holders to the Board | | • Overseeing our compliance programs, including healthcare compliance and data privacy, and corporate governance matters |
The Nominating and Corporate Governance Committee has a written charter, which is available on our website atwww.exelixis.com under the caption “Investors & Media-Corporate Governance.Media—Corporate Governance—Committee Composition and Charters.”
Director Qualifications; Diversity.Because we are a biopharmaceutical company with rapidly evolving and expanding clinical and commercial programs, the Board does not believe that it is appropriate to adopt, and the The Nominating and Corporate Governance Committee hasdoes not adopted, a formal policy with respect tohave a fixed set of minimum qualifications for its candidates for membership on the Board. Instead, in considering candidates for directorship, the Nominating and Corporate Governance Committee will generally consider all relevant factors, including the candidate’s applicable expertise and demonstrated excellence in his or her field, the usefulness of such expertise to us, the availability of the candidate to devote sufficient time and attention to the affairs of Exelixis, the existence of any relationship that would interfere with the exercise of the candidate’s independent judgment, and the candidate’s demonstrated character and judgment. In the review process, the Nominating and Corporate Governance Committee evaluates prospective candidates for directorship in the context of the existing membership of the Board (including the qualities and skills of the existing directors), our operating requirements and the long-term interests of our stockholders. TheWhile the Board does not have a formal policy with regard to the consideration of diversity in identifying director nominees, but the Nominating and Corporate Governance Committee believes that the factors considered above enable it to identify director candidates that possess a wide range of backgrounds,key industry knowledge as well as a variety of different professional and personal backgrounds, attributes (including, but not limited to, gender and national origin), experiences, skills and experiences.expertise such that the Board, as a group, can best fulfill its responsibilities to our stockholders.
The Nominating and Corporate Governance Committee regularly evaluates the needs of the Board with respect to skills and experiences that may be filled by a new director candidate. During the first quarter of 2018, after consultations with certain of our larger institutional stockholders and utilizing various internal resources, the Nominating and Corporate Governance Committee identified Dr. Maria Freire as a candidate for membership on the Board due to her training as a scientist, her knowledge and experience with respect to medical research, the pharmaceutical industry and government healthcare policymaking, as well as her leadership experience in the public sector; Dr. Freire was the elected to the Board in April 2018. In addition, the Nominating and Corporate Governance Committee is authorized to access external resources as it deems necessary or appropriate to fulfill its defined responsibilities, including engagement of executive search firms to help identify director candidates.
Director Nominations.The Nominating and Corporate Governance Committee considers and assesses all candidates recommended by our directors, officers and stockholders. Evaluations of candidates generally involve a review of background materials, internal discussions and interviews with selected candidates as appropriate. If, after its review, the Nominating and
20 Exelixis, Inc.
Proposal 1 | Board Committees and Meetings
Corporate Governance Committee supports a candidate, it would recommend the candidate for consideration by the full Board. The Nominating and Corporate Governance Committee considers stockholder recommendations for directors using the same criteria as potential nominees recommended by the members of the Nominating and Corporate Governance Committee or others. The Nominating and Corporate Governance Committee has not received any recommended nominations from any stockholder holding 5% or more of our common stock in connection with the 2015 Annual Meeting.
The Nominating and Corporate Governance Committee is authorized to access external resources as it deems necessary or appropriate to fulfill its defined responsibilities, including engagement of executive search firms to help identify director
candidates. In February 2016, the Nominating and Corporate Governance Committee engaged an executive search firm to assist the committee in identifying and recruiting potential candidates for membership on the Board.
Stockholders who wish to recommend individuals for consideration by the Nominating and Corporate Governance Committee to become nominees for election to the Board may do so by delivering a written recommendation to the Nominating and Corporate Governance Committee within the timeframe specified in our Bylaws that is applicable to matters to be brought before an Annual Meeting of Stockholders as set forth under “Questions and Answers About These Proxy Materials and Voting” above. Such communications should be sent to the following address: Exelixis, Inc., 210 East Grand Ave., South San Francisco,1851 Harbor Bay Parkway, Alameda, California 94080,94502, Attn: Nominating and Corporate Governance Committee of the Board. Submissions must include (a) the full name, age, business address and residence address of such person, (b) the principal occupation or employment of such person, (c) the class and number of shares of the proposed nominee,corporation which are beneficially owned by such person, (d) a description of all arrangements or understandings between the proposed nominee’s business experience for at leaststockholder and each nominee and any other person or persons (naming such person or persons) pursuant to which the previous five years, complete biographical information, a description of the proposed nominee’s qualifications as a director and a representation that the nominating stockholder is a beneficial or record owner of our stock. Any such submission mustnominations are to be accompaniedmade by the stockholder, (e) a statement whether such person, if elected, intends to comply with the corporation’s Corporate Governance Guidelines, including with respect to matters relating to the election of directors, and (f) any other information relating to such person that is required to be disclosed in solicitations of proxies for election of directors, or is otherwise required, in each case pursuant to Regulation 14A under the 1934 Act (including without limitation such person’s written consent ofto being named in the proposed nominee to be namedproxy statement, if any, as a nominee and to serveserving as a director if elected.
elected).Research and& Development Committee
The Research and& Development Committee which was established in January 2006, is responsible for advising Exelixis andof the Board onoversees various scientific matters related to our drug discovery and preclinical and clinical development programs. The Research & Development Committee is composed entirely of scientific importance as the Board, in consultation with management, may designate from time to time.
independent directors and performs several functions, including: | | |
• Overseeing our clinical development program and internal drug discovery activities | | • Evaluating and discussing trends in the oncology treatment landscape and potential effects on our pipeline strategy and other business needs |
| |
• Reviewing the progress of preclinical assets that we have in-licensed or acquired and evaluating potential future business development opportunities | | • Advising the Board on other matters of scientific importance as the Board, in consultation with management, may designate from time to time |
Annual Meeting; Attendance
The Board does not have a formal policy with respect to the attendance of its members at Annual Meetings of Stockholders. Dr. Morrissey was the only member of the Board in attendance at the 20152018 Annual Meeting of Stockholders.
2019 Proxy Statement 21
COMPENSATION OF DIRECTORS
Overview of Director Compensation
The compensation program for our non-employee directors is intended to be competitive and fair so that we can attract optimal talent to our Board and recognize the time and effort required of a director given the size and complexity of our operations. In accordance with its charter, our Compensation Committee is responsible for recommending to the Board for approval the annual compensation for our non-employee directors and acts on behalf of the Board in discharging the Board’s responsibilities with respect to overseeing our compensation policies for non-employee directors. To assist with the Compensation Committee’s and the Board’s review, Compensia, our external compensation consultant, prepares a comprehensive annual assessment of our non-employee director compensation program. The assessment includes benchmarking director compensation against the same 2018 peer group used for executive compensation purposes, an update in recent trends in director compensation and a review of related corporate governance best practices.
Recent Changes to Director Compensation
In February 2018, the Board approved changes to the non-employee director compensation program following consideration of market data prepared by the Compensation Committee’s independent compensation adviser, Compensia, and the recommendation of the Compensation Committee. The primary goal of these changes was to balance our ability to attract and retain the highest quality directors while aligning the program with the interests of our stockholders. As such, the Board sought to approximate the 50th percentile of total compensation for our non-employee directors with those in the 50th percentile of our 2018 peer group. The Board also sought to ensure that the pay structure appropriately reflected the scope of responsibilities and level of contribution provided by our non-employee directors. Among the changes to the program in 2018 were the following:
| ›› | For the cash component of our non-employee director compensation program, our non-employee directors no longer receive a “Meeting Fee” for attendance at each Board meeting or at each of their respective committee meetings. Instead, we pay a retainer to each non-employee director for service on the Board and standing committees during the fiscal year to reflect his or her responsibilities beyond attendance at Board and committee meetings. We only pay Meeting Fees for attendance at Board or committee meetings in excess of a pre-determined number of meetings for the fiscal year. |
| ›› | For the equity component of our non-employee director compensation program, the dollar value of the initial awards and annual awards our non-employee directors are eligible to receive was increased to approximately $680,000 and approximately $340,000, respectively. |
These two changes were effective beginning with the first quarter of fiscal 2018.
In addition, at our 2017 Annual Meeting, our stockholders approved a limit on the amount of non-employee director compensation under our 2017 Equity Incentive Plan (2017 Plan). The aggregate value of all compensation granted or paid to any individual solely for service as a non-employee director of the Board of Directors with respect to non-employee directors.
Compensation Arrangements; Cash and Restricted Stock Units (RSUs)any calendar year may not exceed $750,000 in Lieutotal value, calculating the value of Cash
Historically, eachany stock awards based on the grant date fair value of such awards for financial reporting purposes), or, with respect to the calendar year in which a non-employee director
has receivedis first appointed or elected to the Board, $1,500,000. This limit was not intended to serve as an increase in the annual amount of non-employee director compensation; rather, this action was approved for the purpose of limiting the amount of compensation the Board can approve for non-employee directors each year.Cash Compensation Arrangements
Each non-employee director receives an annual cash retainer and fees for his or her service on the Board, meeting attendance.as well as an additional annual cash retainer if he or she serves as the Chairman of the Board, on a committee or as the chair of a committee. In addition, chairs of our Board committees have received additional annual retainers and feesnon-employee directors receive Meeting Fees for attendance at each Board meeting or at each of their respective Committee meetings. However,committee meetings in excess of the pre-determined number of meetings for the fiscal year, which is included in the wakefull description of the company's September 2014 restructuring, the non-employee Board members determined that, in support of the company’s cash conservation measures, they would forego the cash component of their compensation, and instead, be paid that component in the form of equity for services rendered from the fourth quarter of 2014 through the end of 2015. The Board delegated authority to the Compensation Committee to research, develop, discuss and approve the design and terms of the new equity component.
The Compensation Committee reviewed several alternatives, including the grant of restricted stock units, or RSUs, in lieu of2018 cash compensation and determined that RSUs were the appropriate instrument to replace the director’s cash compensation. (An RSU award is the right to receive an amount of shares, or cash or any combination thereof equal to the fair market value of the shares covered by the RSU on the vesting date.) The Compensation Committee then reviewed eacharrangements for our non-employee director’s projected cash compensation for the fourth quarter of 2014 through the end of 2015, taking into account the number of regularly scheduled board and committee meetings anticipated during that period and the retainer payable in accordance with the Compensation Arrangements for Non-Employee Directors for Fiscal 2014 (as set forthdirectors in the table below), and usedbelow.22 Exelixis, Inc.
Compensation of Directors
The table below provides information regarding the closing price of the company’s common stock on September 30, 2014, of $1.53 to determine the number of RSUs to be granted to each non-employee director.
In determining the level of cash compensation arrangements for our non-employee directors for 2014, the Compensation Committee considered information provided by Radford,2018. Dr. Morrissey receives no compensation in his capacity as a compensation consultant retained by Exelixis to compile industry compensation data. The Compensation Committee, and then the full Board, reviewed, considered and discussed those materials and concluded that 2014 compensation should be targeted with respect to the peer group approved by the Board for compensation purposes at a level approximately the same (and not to exceed) thatmember of the 2014 peer group approved by the Board for cash and equity compensation for the company’s executive officers. The Board then established a special committee comprising Dr. Cohen to undertake a further review of, and make a final determination on behalf of the Board with respect to, 2014 non-employee director cash compensation. The special committee determined that the Board retainer payable to each non-employee director would increase from $20,000 in 2013 to $25,000 in 2014 (as set forth in the table below). For additional information
on our 2014 peer group, please see "Compensation of Executive Officers" included in our Proxy Statement filed with the SEC on April 16, 2015.
Board. |
| | | | | |
Compensation Arrangements for Non-Employee Directors for Fiscal 2014 |
Service | | Fee Type | | | |
Board | | Retainer Fee | | 25,000 |
50,000 | |
| | Additional Chair Retainer Fee | | 30,000 |
30,000 | |
| | Meeting Fee (1)(2) | | | Regular Meeting Fee2,500 | | 2,500 |
|
| | Special Meeting Fee (1) | | 1,000 |
|
Audit Committee | | Retainer Fee | | 6,000 |
10,000 | |
| | Additional Chair Retainer Fee | | 15,000 |
15,000 | |
| | Meeting Fee (2)(1)(3) | | | 1,000 |
|
Compensation Committee | | Retainer Fee | | 5,000 |
8,000 | |
| | Additional Chair Retainer Fee | | 10,000 |
12,000 | |
| | Meeting Fee (2)(1)(3) | | | 1,000 |
|
Nominating & Corporate Governance Committee | | Retainer Fee | | 5,000 |
5,000 | |
| | Additional Chair Retainer Fee | | 10,000 |
10,000 | |
| | Meeting Fee (2)(1)(4) | | | 1,000 |
|
Research & Development Committee | | Retainer Fee | | 10,000 |
5,000 | |
| | Additional Chair Retainer Fee | | 10,000 |
10,000 | |
| | | Meeting Fee (2)(1)(4) | | 5,000 |
|
| (1) | Meeting atfor which minutes are generated.generated count toward the meeting threshold to determine when Meeting Fees are to be paid. |
| |
| (2) | In-person meeting or teleconference at which minutes are generated.Meeting Fee paid for all meetings in excess of eight meetings. |
The following table sets forth the number| (3) | Meeting Fee paid for all meetings in excess of seven meetings. |
| (4) | Meeting Fee paid for all meetings in excess of four meetings. |
Equity Compensation Arrangements
Our non-employee directors are also eligible to receive equity as part of RSUs granted to each non-employee director (in lieu of cash compensation for the fourth quarter of 2014 through the end of 2015) who was serving ontheir Board service, including an initial award upon joining the Board and its committeesan annual award on October 14, 2014, the dateday following each Annual Meeting of grant:
|
| | |
Name | | Restricted Stock Units
Granted in Lieu of
Cash Compensation
(Sh)
|
Charles Cohen, Ph.D. | | 63,725 |
Carl B. Feldbaum, Esq. | | 40,033 |
Alan M. Garber, M.D., Ph.D. | | 48,611 |
Vincent T. Marchesi, M.D., Ph.D. | | 61,683 |
Stelios Papadopoulos, Ph.D. | | 69,444 |
George Poste, D.V.M., Ph.D., FRS | | 63,725 |
George A. Scangos, Ph.D. | | 29,412 |
Lance Willsey, M.D. | | 61,275 |
Jack L. Wyszomierski | | 55,964 |
The RSUs were grantedStockholders. Grants to our non-employee directors are made under the 2014 Equity Incentive Plan, or 2014 Equityour 2017 Plan, pursuant to the Non-Employee Director Equity Compensation Policy, oras amended (2017 Directors’ Policy, described below and vested as to one-fifth of the shares on each of January 2, 2015, April 3, 2015, October 2, 2015 and January 1, 2016.
In December 2015, the Compensation Committee met to discuss non-employee director cash compensation for 2016. Following a review of the compensation arrangement covering the period beginning with the fourth quarter of 2014 through the end of 2015 and with a primary motivation of cash preservation, the Compensation Committee recommended to the Board the continuation of a compensation program that provided for the issuance of RSUs in lieu of cash compensation for non-employee directors. The Board then reviewed the Compensation Committee's recommendation and determined that, in support of the company’s cash conservation measures, they would continue to forgo the cash component of their compensation, and instead,
be paid that component in the form of RSUs for services rendered for 2016. The Board delegated authority to the Compensation Committee to determine the amount of RSUs to be granted to each non-employee director.
The Compensation Committee then reviewed each non-employee director’s projected cash compensation for 2016, taking into account the number of regularly scheduled board and committee meetings anticipated during that period and the retainer payable in accordance with the Compensation Arrangements for Non-Employee Directors for Fiscal 2014 (as set forth in the table above). Given the company's financial position and cash conservation efforts, the Compensation Committee determined that it was appropriate to utilize the established compensation arrangements for non-employee directors for 2014 and 2015 in making its non-employee director compensation decisions for 2016. The Compensation Committee then used the closing price of the company’s common stock on December 31, 2015, of $5.64 to determine the number of RSUs to be granted to each non-employee director.
The following table sets forth the number of RSUs granted to each non-employee director (in lieu of cash compensation for 2016) who was serving on the Board and its committees on January 4, 2016, the date of grant:
|
| | |
Name | | Restricted Stock Units
Granted in Lieu of
Cash Compensation
(Sh)
|
Charles Cohen, Ph.D. | | 13,830 |
Carl B. Feldbaum, Esq. | | 8,688 |
Alan M. Garber, M.D.Policy), Ph.D. | | 10,550 |
Vincent T. Marchesi, M.D., Ph.D. | | 13,387 |
Stelios Papadopoulos, Ph.D. | | 15,071 |
George Poste, D.V.M., Ph.D., FRS | | 13,830 |
George A. Scangos, Ph.D. | | 6,383 |
Lance Willsey, M.D. | | 13,298 |
Jack L. Wyszomierski | | 12,146 |
The RSUs were granted under the 2014 Equity Plan pursuant to the Directors’ Policy and will vest as to one-fourth of the shares on each of April 1, 2016, July 1, 2016, September 30, 2016 and December 30, 2016, subject to the non-employee director’s continuous service through those dates.
Additional Equity Compensation Arrangements
Our non-employee directors are also eligible to receive stock options as part of their Board service. Grants to our non-employee directors are made under the 2014 Equity Plan pursuant to the Directors’ Policy, as adopted by the Board.
The Directors’ Policy became effective onTo address changes in the
datetrading price of
the 2014 Annual Meeting of Stockholders and provides for the automatic grant of options to purchase shares ofour common stock,
we utilize a value-based approach for determining the number of shares subject to non-employee
directors. Underdirector equity awards. During 2018, under the terms of the
2017 Directors’ Policy,
the aggregate value of each one-time initial award was approximately $680,000, and the aggregate value of each annual award was approximately $340,000. The value of each initial award and annual award is divided approximately evenly between a nonstatutory stock option and a restricted stock unit (RSU) award. However, each non-employee
directors are eligibledirector may instead elect to receive
the full value of his or her annual award in the form of a
one-timenonstatutory stock option.Under the 2017 Directors’ Policy, the total number of options and RSUs granted as part of each initial award and annual award is determined using a formula based upon the Black-Scholes Merton option to purchase 65,000 sharespricing model and the average of the daily closing sale prices of our common stock when they first joinfor the Boardtrading days during the 30-day calendar period ending on (and including) the last calendar day immediately prior to the relevant grant date . The value of each award, as determined in accordance with the 2017 Directors’ Policy, may be greater or lesser than the grant date fair value computed for financial reporting purposes and an annualreflected in the “Director Compensation Table” below. This is a result of the different calculation employed to determine the grant date fair value, which uses a formula based upon the Black-Scholes Merton option to purchase 40,000 sharespricing model and the closing sale price of our common stock on the day following each Annual Meeting of Stockholders.
grant date.Options granted under the 2014 Equity2017 Plan in accordance with the 2017 Directors’ Policy are not incentive stock options under the Internal Revenue Code of 1986, as amended or the Code.(the Code). The exercise price of automatic grants ofeach initial and annual stock optionsoption granted under the 2014 Equity2017 Plan is equal to 100% of the fair market value of a share of common stock on the grant date. Under the terms of the 2017 Directors’ Policy, the one-time initial options to purchase 65,000 shares are immediately exercisable, but shares issued upon early exercise are subject to a repurchase right and will vest at the rate of 25% of the underlying shares on the first anniversary of the grant date and monthly thereafter over the next three years. The annual grantsoptions are immediately exercisable, immediately but shares issued upon early exercise are subject to a repurchase right and will vest monthly in equal parts over a one-year period.
2019 Proxy Statement 23
As long as the option holdernon-employee director continues to serve with us or with an affiliate of us,ours, the option willoptions continue to vest and be exercisable during its term.their terms, and shares issued upon early exercise continue to vest. When the option holder’s service terminates, we will have the right to repurchase any unvested shares acquired upon exercise of the option at the original exercise price without interest. The post-termination exercise period for the vested portion of the options granted to our non-employee directors is generally set to terminate the earlier of three years after a non-employee director'sdirector’s service terminates or the remainder of the term of the option. option, as described in the form of option agreement for non-employee directors under the 2017 Plan (not to exceed seven years from the date of grant).
The options grantedinitial RSU awards vest at the rate of 25% of the underlying shares on each of the first four anniversaries of the grant date, and the annual RSU awards vest at the rate of 100% of the shares on the first anniversary of the grant date, in each case so long as the non-employee director continues to serve with us or with an affiliate of ours.
In the event of a change in control, 100% of the non-employee director’s outstanding and unvested equity awards will immediately vest, and any applicable repurchase rights we may have will terminate.
On April 5, 2018, Dr. Freire Joined the Board and became eligible to receive an initial equity award having an aggregate value equal to approximately $680,000, pursuant to the terms of the 2017 Directors’ Policy havePolicy. Accordingly, on April 5, 2018, the Board granted Dr. Freire a term that does not exceed seven years.
On May 28, 2015,one-time initial award, the day after our 2014 Annual Meetingvalue of Stockholders, we grantedwhich was divided approximately evenly between a nonstatutory stock option and an annualRSU award consisting of (i) an option to purchase 40,00028,240 shares of common stock to each of our non-employee directors, or the Annual Options. The Annual Options were granted at an exercise price per share of $3.13, which was equal to the fair market value of our common stock on the dateand (ii) an RSU award representing 14,120 shares of grant (based on the last reported sale price as quoted on the NASDAQ Global Select Market on the last trading day prior to the day of grant).
our common stock.Reimbursement of Expenses
The members of the Board are eligible for reimbursement of certain expenses incurred in connection with their attendance ofat Board meetings and their service on the Board in accordance with our policy.
Director Compensation Table
The following table shows compensation information for our non-employee directors for the fiscal year ended January 1, 2016.
December 28, 2018.Director Compensation for Fiscal 2015
|
| | | | | | | | | | | | |
| | |
Fees Earned or Paid in Cash ($) | | Stock Awards (Restricted Stock Units) ($)(1) | | Option Awards ($)(2) | |
Total ($) |
| Charles Cohen, Ph.D. | | — |
| | 77,490 |
| | 93,652 |
| | 171,142 |
|
| Carl B. Feldbaum, Esq. | | — |
| | 48,680 |
| | 93,652 |
| | 142,332 |
|
| Alan M. Garber, M.D., Ph.D. | | — |
| | 59,111 |
| | 93,652 |
| | 152,763 |
|
| Vincent T. Marchesi, M.D., Ph.D. | | — |
| | 75,007 |
| | 93,652 |
| | 168,659 |
|
| Stelios Papadopoulos, Ph.D. | | — |
| | 84,444 |
| | 93,652 |
| | 178,096 |
|
| George Poste, D.V.M., Ph.D., FRS | | — |
| | 77,490 |
| | 93,652 |
| | 171,142 |
|
| George A. Scangos, Ph.D. | | — |
| | 35,765 |
| | 93,652 |
| | 129,417 |
|
| Lance Willsey, M.D. | | — |
| | 74,510 |
| | 93,652 |
| | 168,162 |
|
| Jack L. Wyszomierski | | — |
| | 68,052 |
| | 93,652 |
| | 161,704 |
|
| | | | | | | | | | | | | | | | |
| | | Fees Earned or
Paid in Cash
($) | | | Stock Awards
(Restricted Stock
Units) ($)(1) | | | Option Awards
($)(2) | | | Total ($) | |
Charles Cohen, Ph.D. | | | 88,000 | | | | — | | | | 320,699 | | | | 408,699 | |
Carl B. Feldbaum, Esq. | | | 66,000 | | | | — | | | | 320,699 | | | | 386,699 | |
Maria C. Freire, Ph.D. | | | 45,000 | | | | 474,761 | (3) | | | 476,599 | (4) | | | 996,360 | |
Alan M. Garber, M.D., Ph.D. | | | 70,000 | | | | — | | | | 320,699 | | | | 390,699 | |
Vincent T. Marchesi, M.D., Ph.D. | | | 65,000 | | | | 162,709 | | | | 163,339 | | | | 391,048 | |
Stelios Papadopoulos, Ph.D. | | | 95,000 | | | | 162,709 | | | | 163,339 | | | | 421,048 | |
George Poste, D.V.M., Ph.D., FRS | | | 70,000 | | | | 162,709 | | | | 163,339 | | | | 396,048 | |
George A. Scangos, Ph.D. | | | 55,000 | | | | — | | | | 320,699 | | | | 375,699 | |
Julie A. Smith | | | 65,250 | | | | — | | | | 320,699 | | | | 385,949 | |
Lance Willsey, M.D. | | | 64,000 | | | | — | | | | 320,699 | | | | 384,699 | |
Jack L. Wyszomierski | | | 80,000 | | | | 162,709 | | | | 163,339 | | | | 406,048 | |
| |
| (1) | ThereOn May 24, 2018, each of Drs. Freire, Marchesi, Papadopoulos and Poste and Mr. Wyszomierski were no RSU awards granted to non-employee directors during fiscal 2015. On October 14, 2014, each non-employee director was granted an RSU that vested with respectaward pursuant to 1/5the 2017 Directors’ Policy, which accounted for approximately 50% of the shares at the endtotal value of the fourth quarter of fiscal 2014, and 1/5 of the shares at the end of each fiscal quarter of fiscal 2015. The RSU award was granted in lieu of cash fees that would otherwise have been paid for Board service for those five quarters.their annual award. Amounts shown in this column reflect the aggregate grant date fair value for the portion of the RSU award granted in fiscal 2014 that relates to services provided during fiscal 2015, as computed in accordance with Financial Accounting Standards Board (FASB), Accounting Standards or FASB, Codification Topic 718 or ASC 718. The aggregate grant date fair value for the total RSU award granted to each non-employee director in fiscal 2014 is as follows: Dr. Cohen - $96,862; Mr. Feldbaum - $60,850; Dr. Garber - $73,889; Dr. Marchesi - $93,758; Dr. Papadopoulos - $105,555; Dr. Poste - $96,862; Dr. Scangos - $44,706; Dr. Willsey - $93,138; and Mr. Wyszomierski - $85,065.(ASC 718). The assumptions used to calculate the value of RSUs are set forth in Note 107 of the Notes to Consolidated Financial Statements included in our Annual Report on Form 10-K for the fiscal year ended January 1, 2016,December 28, 2018, filed with the SEC on February 29, 2016.22, 2019. See “Compensation Arrangements; Cash and Restricted Stock Units (RSUs) in Lieu of Cash”“Equity Compensation Arrangements” above for a description of the RSU awards made to non-employee directors on October 14, 2014. The aggregateMay 24, 2018. |
24 Exelixis, Inc.
Compensation of Directors
| Only one RSU award was granted to each of Drs. Marchesi, Papadopoulos and Poste and Mr. Wyszomierski during fiscal 2018 and, accordingly, the grant date fair value forof that RSU award is reflected in the portiontable. The aggregate number of shares subject to all RSUs held by each of these non-employee directors as of December 28, 2018, is as follows: Dr. Freire—22,057 Dr. Marchesi—7,937; Dr. Papadopoulos—7,937; Dr. Poste—7,937; and Mr. Wyszomierski—7,937. None of the other non-employee directors received any RSU award granted in fiscal 2014 that relates to services providedawards during fiscal 2014 was reported in last year's proxy statement.2018, and none of the other non-employee directors held any outstanding RSU awards as of December 28, 2018. |
There were no RSUs held by any non-employee director as of January 1, 2016.
| |
| (2) | Amounts shown in this column do not reflect compensation actually received or amounts that may be realizedOn May 24, 2018, each non-employee director was granted an option to purchase our common stock pursuant to the 2017 Directors’ Policy. Each of Drs. Cohen, Garber, Scangos and Willsey, Mr. Feldbaum and Ms. Smith elected to receive 100% of their annual award in the future byform of options to purchase our common stock, while each of Drs. Freire, Marchesi, Papadopoulos and Poste and Mr. Wyszomierski elected to receive approximately 50% of their annual award in the non-employee directors.form of options to purchase our common stock and approximately 50% of their annual award in the form of an RSU award. Amounts shown in this column reflect the aggregate grant date fair value for the option awards granted in fiscal 20152018 as computed in accordance with FASB ASC 718. The assumptions used to calculate the value of option awards are set forth in Note 107 of the Notes to Consolidated Financial Statements included in our Annual Report on Form 10-K for the fiscal year ended January 1, 2016,December 28, 2018, filed with the SEC on February 29, 2016.22, 2019. See “Additional Equity“Equity Compensation Arrangements” above for a description of the option grants made to non-employee directors on May 28, 2015.24, 2018. There can be no assurance that the options will ever be exercised (in which case no value will actually be realized by the director) or that the value on exercise of stock options will be equal to the grant date fair value shown in this column. |
| With the exception of Dr. Freire, who was also granted a stock option award as part of her initial award in connection with her joining the Board on April 5, 2018, only one stock option award was granted to each non-employee director during fiscal 2018 and, accordingly, the grant date fair value of that stock option is reflected in the table. The aggregate number of shares subject to all stock options held by each of our current non-employee directors as of December 28, 2018, is as follows: Dr. Cohen—361,304; Mr. Feldbaum—274,704; Dr. Freire—44,114; Dr. Garber—302,864; Dr. Marchesi—218,814; Dr. Papadopoulos—339,601; Dr. Poste—227,222; Dr. Scangos—241,344; Ms. Smith—97,278; Dr. Willsey—352,050; and Mr. Wyszomierski—281,213. |
| (3) | On April 5, 2018, as part of her initial award in connection with her joining the Board, Dr. Freire was granted an RSU award representing 14,120 shares of our common stock. The grant date fair value of the RSU component of her initial award was $312,052, and the grant date fair value of the RSU component of her annual award was $162,709, in each case as computed in accordance with FASB ASC 718. The assumptions used to calculate the value of this option award is set forth in Note 7 of the Notes to Consolidated Financial Statements included in our Annual Report on Form 10-K for the fiscal year ended December 28, 2018, filed with the SEC on February 22, 2019. See “Equity Compensation Arrangements” above for a description of the initial award granted to Dr. Freire on April 5, 2018. |
| (4) | On April 5, 2018, as part of her initial award in connection with her joining the Board, Dr. Freire was granted an option to purchase 28,240 shares or our common stock. The grant date fair value of the option component of her initial award was $313,260, and the grant date fair value of the option component of her annual award was $163,339, in each case as computed in accordance with FASB ASC 718. The assumptions used to calculate the value of this option award is set forth in Note 7 of the Notes to Consolidated Financial Statements included in our Annual Report on Form 10-K for the fiscal year ended December 28, 2018, filed with the SEC on February 22, 2019. See “Equity Compensation Arrangements” above for a description of the initial award granted to Dr. Freire on April 5, 2018. There can be no assurance that the option will ever be exercised (in which case no value will actually be realized by the director) or that the value on exercise of the stock option will be equal to the grant date fair value described above. |
2019 Proxy Statement 25
Only one stock option award was granted to each non-employee director during fiscal 2015 and, accordingly, the grant date fair value of that stock option is reflected in the table. The aggregate number of shares subject to all stock options held by each of our current non-employee directors as of January 1, 2016, is as follows: Dr. Cohen - 316,646; Mr. Feldbaum - 259,796; Dr. Garber - 258,206; Dr. Marchesi - 281,254; Dr. Papadopoulos - 322,041; Dr. Poste - 260,912; Dr. Scangos - 886,684; Dr. Willsey - 307,392; and Mr. Wyszomierski - 263,653.
PROPOSAL 2
RATIFICATIONOF SELECTIONOF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRMATIFICATIONOF SELECTIONOF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
The Audit Committee of the Board has selected Ernst & Young LLP as Exelixis’ independent registered public accounting firm for the fiscal year ending December 30, 2016.January 3, 2020. The Board, on behalf of the Audit Committee, has further directed that management submit the selection of the independent registered public accounting firm for ratification by the stockholders at the Annual Meeting. Ernst & Young LLP has audited our financial statements for each of the fifteeneighteen fiscal years in the period ended January 1, 2016.December 28, 2018. Representatives of Ernst & Young LLP are expected to be present at the Annual Meeting. They will have an opportunity to make a statement if they so desire and will be available to respond to appropriate questions.
Neither our Bylaws nor other governing documents or law require stockholder ratification of the selection of Ernst & Young LLP as Exelixis’ independent registered public accounting firm. However, the Board is submitting the selection of Ernst & Young LLP to the stockholders for ratification as a matter of good corporate governance. If the stockholders fail to ratify the selection, the Audit Committee of the Board will reconsider whether or not to retain that firm. Even if the selection is ratified, the Audit Committee of the Board in its discretion may direct the appointment of a different independent registered public accounting firm at any time during the year if they determine that such a change would be in the best interests of Exelixis and its stockholders.
The affirmative vote of the holders of a majority of the shares present in person or represented by proxy and entitled to vote at the Annual Meeting is required to ratify the selection of Ernst & Young LLP. Abstentions will be counted toward the tabulation of votes cast on proposals presented to the stockholders and will have the same effect as a vote against this proposal. Broker non-votes are counted towards a quorum, but willBrokers generally have no effectdiscretionary authority to vote on the resultsratification of our independent accounting firm; thus, we do not expect any broker non-votes on this vote.
THE BOARDTHE BOARD OF DIRECTORS RECOMMENDS DIRECTORS RECOMMENDS A VOTE FOR PROPOSAL VOTE “FOR” PROPOSAL 2.
Principal Accountant Fees and Services
The aggregate fees billed by Ernst & Young LLP for the last two fiscal years for the services described below are as follows:
|
| | | | | | | | |
| | | Fiscal Year Ended |
| | | January 1, 2016 | | January 2, 2015 |
| Audit Fees (1) | | $ | 1,224,884 |
| | $ | 1,221,921 |
|
| Audit-Related Fees (2) | | 25,000 |
| | 80,000 |
|
| Tax Fees | | — |
| | — |
|
| All Other Fees (3) | | 1,995 |
| | — |
|
| Total Fees | | $ | 1,251,879 |
| | $ | 1,301,921 |
|
| | | | | | | | |
| | | Fiscal Year Ended | |
| | | December 28,
2018 | | | December 29,
2017 | |
Audit Fees (1) | | $ | 1,914,430 | | | $ | 1,658,395 | |
Audit-Related Fees (2) | | | 155,200 | | | | 220,000 | |
Tax Fees (3) | | | 416,273 | | | | 208,152 | |
All Other Fees (4) | | | 3,230 | | | | 1,995 | |
Total Fees | | $ | 2,489,133 | | | $ | 2,088,502 | |
| |
| (1) | “Audit fees” consist of fees billed for professional services rendered for the audit of our consolidated financial statements and review of the interim consolidated financial statements included in quarterly reports and services that are normally provided by Ernst & Young LLP in connection with statutory and regulatory filings and other engagements such as comfort letters, consents, and review of documents filed with the SEC. |
| |
| (2) | “Audit-related fees” consist of fees billed for assurance and related services that are reasonably related to the performance of the audit or review of our consolidated financial statements and are not reported under “Audit fees.” During fiscal 20152018 and 2014,2017 these services included consultations relating to various transactions. |
| |
| (3) | “Tax fees” include fees for tax compliance, tax advice and tax planning. |
| (4) | “All other fees” consist of fees for products and services other than the services described above. During fiscal 2015,2018 and 2017, these fees related to an onlineon-line subscription to an Ernst & Young LLP database. |
26 Exelixis, Inc.
Proposal 2 | Ratification of Selection of Independent Registered Public Accounting Firm
All fees described above were pre-approved by the Audit Committee. The Audit Committee has determined that the rendering of the services other than audit services by Ernst & Young LLP is compatible with maintaining the independence of the independent registered public accounting firm.
During 20152018 and 2014,2017, the Audit Committee of the Board pre-approved the audit and non-audit services to be performed by Exelixis’ independent registered public accounting firm, Ernst & Young LLP. Non-audit services are defined as services other than those provided in connection with an audit or a review of our financial statements. The Audit Committee pre-approves all audit and non-audit services rendered by Ernst & Young LLP. The Audit Committee generally pre-approves specified services in the defined categories of audit services, audit-related services, tax services and all other services up to
specified amounts. Pre-approval may also be given as part of the Audit Committee’s approval of the scope of the engagement of the independent registered public accounting firm or on an individual explicit case-by-case basis before the independent auditor is engaged to provide each service. The Audit Committee or its chairman, whom the Audit Committee has designated as a one-person subcommittee with pre-approval authority, pre-approved all audit fees, audit-related fees, tax fees and other fees in
20152018 and
2014.2017. Any pre-approvals by the subcommittee must be and were presented to the Audit Committee at its next scheduled meeting.
2019 Proxy Statement 27
REPORTOFTHE AUDIT COMMITTEEREPORTOF THE AUDIT COMMITTEE
The material in this report is not “soliciting material,” is not deemed “filed” with the Securities and Exchange Commission and is not deemed to be incorporated by reference in any filing of Exelixis under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing.
In connection with the audited consolidated financial statements for the fiscal year ended January 1, 2016,December 28, 2018, of Exelixis, Inc. (“Exelixis”), the Audit Committee of the Board of Directors of Exelixis has:
(1) reviewed and discussed the audited financial statements for the fiscal year ended January 1, 2016,December 28, 2018, with management of Exelixis;
(2) discussed with Ernst & Young LLP, Exelixis’ independent registered public accounting firm (“Ernst & Young”), the matters required to be discussed by Statement on Auditing Standard No. 16, 1301,Communications with Audit Committees, as adopted by the Public Company Accounting Oversight Board (“PCAOB”); and
(3) received the written disclosures and the letter from Ernst & Young required by applicable requirements of the PCAOB regarding Ernst & Young’s communications with the Audit Committee concerning independence, and has discussed with Ernst & Young that accounting firm’s independence.
Based on the foregoing, the Audit Committee has recommended to the Board of Directors that the audited financial statements be included in Exelixis’ Annual Report on Form 10-K for the fiscal year ended January 1, 2016.
December 28, 2018.Audit Committee:
Jack L. Wyszomierski, Chairman
PJulie A. Smith
ROPOSAL28 Exelixis, Inc.
APPROVALOF ANAMENDMENT| Declassification of the Board by the 2020 Annual Meeting of StockholdersPROPOSAL 3
DECLASSIFICATIONOFTHE BOARDBYTHE 2020 ANNUAL MEETINGOF STOCKHOLDERSNDRESTATEMENTOFTHEEXELIXIS, INC. 2000 EMPLOYEESTOCKPURCHASEPLAN
Currently, Article V.A of the Certificate of Incorporation provides for a classified board of directors divided into three classes of directors, with each class elected for three-year terms.
We take corporate governance matters seriously and are askingcommitted to achieving best corporate governance practices. We recognize, in particular through our ongoing stockholder engagement, the growing sentiment among stockholders that annual director election is one of the most meaningful tools available to them to evaluate and provide feedback on the performance of the Board. After considering the advantages and disadvantages of declassification, including through discussions with our stockholders to approve an amendment and restatementin light of the fact that the number of companies with classified boards continues to decline, the Board has determined it is in the best interests of Exelixis Inc. 2000 Employee Stock Purchase Plan (the “2000 Purchase Plan”) atand its stockholders to amend our Certificate of Incorporation and Bylaws to declassify the Annual Meeting. We refer to such amendment and restatement of the 2000 Purchase PlanBoard. This will result in this Proxy Statement as the “Amended 2000 Purchase Plan.” The Amended 2000 Purchase Plan was approveda fully declassified Board by the Compensation Committee on February 11, 2016, subject to approval by our stockholders.
The Amended 2000 Purchase Plan contains2020 Annual Meeting of Stockholders, at which time the following material changes from the 2000 Purchase Plan:
Subject to adjustmentcurrent term for certain changeseach director then in our capitalization, the maximum number of shares of our common stockoffice will expire, notwithstanding that such director may be issued under the Amended 2000 Purchase Plan will be 13,650,000 shares, which is an increase of 5,000,000 shares over the maximum number of shares of our common stockhave been elected for a term that may be issued under the 2000 Purchase Plan.
The Amended 2000 Purchase Plan provides that the Board and the Compensation Committee will have the discretion to structure an offering so that if the fair market value of our common stock on any purchase date during an offering is less than or equal to the fair market value of our common stock on the first day of the offering, then (i) that offering will terminate immediately following the purchase of shares on such purchase date, and (ii) the participants in such terminated offering will be automatically enrolled in a new offering that begins immediately after such purchase date. The 2000 Purchase Plan does not include such a provision.
The Amended 2000 Purchase Plan provides that upon certain changes in our capitalization, the purchase price of outstanding purchase rights will be appropriately adjusted. The 2000 Purchase Plan does not include such a provision.
In the Amended 2000 Purchase Plan, the definition of “corporate transaction” has been revised to include the acquisition of beneficial ownership of our securities representing at least 50% of the combined voting power entitled to vote in the election of members of the Board.
Approval of the Amended 2000 Purchase Plan by our stockholders will allow us to continue to provide our employees with the opportunity to acquire an ownership interest in Exelixis through their participation in the Amended 2000 Purchase Plan, thereby encouraging them to remain in our service and more closely aligning their interests with those of our stockholders.
If this Proposal 3 is approved by our stockholders, an additional 5,000,000 shares of our common stock will be available for issuance under the Amended 2000 Purchase Plan. As of March 31, 2016, a total of 1,046,959 shares of our common stock were available for issuance under the 2000 Purchase Plan. We do not maintain any other employee stock purchase plans. As of March 31, 2016, a total of 228,654,405 shares of our common stock were outstanding.
If this Proposal 3 is approved by our stockholders, the Amended 2000 Purchase Plan will become effective as ofextended beyond the date of the 2020 Annual Meeting. InMeeting of Stockholders. Thus, upon the event thatfiling of a certificate of amendment to our stockholders do not approve this Proposal 3, the Amended 2000 Purchase Plan will not become effective and the 2000 Purchase Plan will continue in its current form.
SummaryCertificate of Incorporation following stockholder approval of the Amended 2000 Purchase Plan
proposed amendment to our Certificate of Incorporation, beginning with the 2020 Annual Meeting of Stockholders, all directors would henceforth be elected annually. The
material featuresproposed amendment would not change the present number of directors or the Board’s authority to change that number and to fill any vacancies or newly created directorships. Vacancies which occur during the year may be filled by the Board for the remainder of the
Amended 2000 Purchase Plan are described below. The following descriptionfull term. In accordance with Delaware law, the proposed amendment would provide that once the Board is fully declassified as of the
Amended 2000 Purchase Plan2020 Annual Meeting of Stockholders, directors may be removed at any time, either with or without cause.Effect of Declassifying the Board
Classified boards provide protection against certain abusive takeover tactics and more time to solicit higher bids in a hostile takeover situation because it is more difficult to change a summary onlymajority of directors on the board in a single year. While the Board continues to believe that these are important considerations, the Board also considered potential advantages of declassification, including the ability of stockholders to evaluate directors annually.
Language of Proposed Amendment
If approved, the amendment would enable us to amend and is qualifiedrestate Article V of our Certificate of Incorporation as follows:
Article V.A, Section 3, of the Certificate of Incorporation be amended to read in its entirety by referenceas follows:
“3. Election of Directors. Subject to the complete textrights of the Amended 2000 Purchase Plan, whichholders of any series of Preferred Stock to elect additional directors under specified circumstances and the remaining provisions of this section 3, until the Corporation’s 2020 annual meeting of stockholders, the directors shall be divided into three classes designated as Class I, Class II and Class III, respectively. At each annual meeting of stockholders held following the closing of the initial public offering pursuant to an effective registration statement under the Securities Act of 1933, as amended, covering the offer and sale of Common Stock to the public (the Initial Public Offering) and prior to or at the Corporation’s 2019 annual meeting of stockholders, each director was elected for a three year term, expiring at the third annual meeting of stockholders following his or her election. Subject to the rights of the holders of any series of Preferred Stock to elect additional directors under specified circumstances, the term of each director then in office shall expire at the Corporation’s 2020 annual meeting of stockholders, notwithstanding that such director may have been elected for a term that extended beyond the date of such annual meeting. Subject to the rights of the holders of any series of Preferred Stock to elect additional directors under specified circumstances, commencing at the Corporation’s 2020 annual meeting of stockholders, each director elected at such meeting and at each annual meeting of stockholders thereafter to succeed those directors whose terms then expire shall be elected for a term expiring at the next annual meeting of stockholders following their election.
Notwithstanding the foregoing provisions of this section, each director shall serve until his or her successor is attached to thisduly elected and qualified or until his or her death, resignation or removal. No decrease in the number of directors constituting the Board of Directors shall shorten the term of any incumbent director.”
2019 Proxy Statement as Appendix A.29
Purpose
The purposeArticle V.A, Section 4, of the Amended 2000 Purchase Plan isCertificate of Incorporation be amended to provide a means by which our employeesread in its entirety as follows:
“4. Removal of Directors. Subject to the rights of the holders of any series of Preferred Stock then outstanding, any director or the entire Board of Directors may be given an opportunity to purchase sharesremoved by the holders of our common stock, to assist us in retaining the services of our employees, to secure and retain the services of new employees and to provide incentives for such persons to exert maximum efforts for our success. The rights to purchase common stock granted under the Amended 2000 Purchase Plan are intended to qualify as options issued under an “employee stock purchase plan” as that term is defined in Section 423(b)a majority of the Internal Revenue Codeshares then entitled to vote at an election of 1986, as amended, or the Code.
Administration
directors.”Effect on Bylaws
The Board has approved the powerbelow changes to administerExelixis’ Bylaws, contingent on the Amended 2000 Purchase Plan and may also delegate administrationeffectiveness of the Amended 2000 Purchase Planproposed amendment to a committee composedthe Certificate of two or more membersIncorporation:
Section 17 of the Board.Bylaws is amended to read in its entirety as follows:
“Section 17. Term of Office. The Board has delegated administrationterm of office of each director shall be as provided in the Certificate of Incorporation.”
Section 20 of the Amended 2000 Purchase PlanBylaws is amended to the Compensation Committee, but may,read in its entirety as follows:
“Section 20. Intentionally Omitted.”
Required Vote and Board of Directors Recommendation
The affirmative vote of at any time, revest in itself some or all of the powers previously delegated to the Compensation Committee. The Boardleast sixty-six and the Compensation Committee are each considered to be a Plan Administrator for purposes of this Proposal 3. The Plan Administrator has the final power to construe and interpret both the Amended 2000 Purchase Plan and the rights granted under it. The Plan Administrator has the power, subject to the provisions of the Amended 2000 Purchase Plan, to determine when and how rights to purchase our common stock will be granted, the provisions of each offering of such rights (which need not be identical), and whether employees of any of our parent or subsidiary companies will be eligible to participate in the Amended 2000 Purchase Plan.
Stock Subject to Amended 2000 Purchase Plan
Subject to adjustment for certain changes in our capitalization, the maximum number of shares of our common stock that may be issued under the Amended 2000 Purchase Plan will be 13,650,000 shares. If any rights granted under the Amended 2000 Purchase Plan terminate without being exercised in full, the shares of common stock not purchased under such rights will again become available for issuance under the Amended 2000 Purchase Plan. The shares of common stock issuable under the Amended 2000 Purchase Plan may be unissued shares or shares that have been bought on the open market.
Offerings
The Amended 2000 Purchase Plan will be implemented by offerings of rights to purchase our common stock to all eligible employees. The Plan Administrator will determine the duration of each offering period, provided that in no event may an offering period exceed 27 months. The Plan Administrator may establish separate offerings which vary in terms (although not inconsistent with the provisions of the Amended 2000 Purchase Plan or the requirements of applicable laws). Each offering period will have one or more purchase dates, as determined by the Plan Administrator prior to the commencement of the offering period. The Plan Administrator has the authority to alter the terms of an offering prior to the commencement of the offering period, including the duration of subsequent offering periods. When an eligible employee elects to join an offering period, he or she is granted a right to purchase shares of our common stock on each purchase date within the offering period. On the purchase date, all contributions collected from the participant are automatically applied to the purchase of our common stock, subject to certain limitations (which are described further below under “Eligibility”two-thirds percent (66-2/3%).
The Plan Administrator has the discretion to structure an offering so that if the fair market value of our common stock on any purchase date during an offering is less than or equal to the fair market value of our common stock on the first day of the offering, then (i) that offering will terminate immediately following the purchase of shares on such purchase date, and (ii) the participants in such terminated offering will be automatically enrolled in a new offering that begins immediately after such purchase date.
Eligibility
Any individual who is employed by us (or by any of our parent or subsidiary companies if such company is designated by the Plan Administrator as eligible to participate in the Amended 2000 Purchase Plan) may participate in offerings under the Amended 2000 Purchase Plan, provided such individual has been employed by us (or our parent or subsidiary, if applicable) for such continuous period preceding the first day of the offering period as the Plan Administrator may require, but in no event may the required period of continuous employment be equal to or greater than two years. In addition, the Plan Administrator may provide that an employee will not be eligible to be granted purchase rights under the Amended 2000 Purchase Plan unless such employee is customarily employed for more than 20 hours per week and five months per calendar year. The Plan Administrator may also provide in any offering that certain of our employees who are “highly compensated” as defined in the Code are not eligible to participate in the Amended 2000 Purchase Plan.
No employee will be eligible to participate in the Amended 2000 Purchase Plan if, immediately after the grant of purchase rights, the employee would own, directly or indirectly, stock possessing 5% or more of the total combined voting power or value of all classes of our stock or of any of our parent or subsidiary companies, including any stock which such employee may purchase under all outstanding purchase rights and options. In addition, no employee may purchase more than $25,000 worth of our common stock (determined based on the fair market value of the shares at the time such rights are granted) under all our employee stock purchase plansissued and any employee stock purchase plans of our parent or subsidiary companies for each calendar year during which such rights are outstanding.
As of March 31, 2016, all of our approximately 229 employees will be eligible to participate in the Amended 2000 Purchase Plan.
Participation in the Amended 2000 Purchase Plan
An eligible employee may enroll in the Amended 2000 Purchase Plan by delivering to us, prior to the date selected by the Plan Administrator as the beginning of an offering period, an agreement authorizing contributions which may not exceed either 15% of such employee’s earnings during the offering period or the maximum dollar amount designated by the Plan Administrator for the offering period. Each participant will be granted a separate purchase right for each offering in which he or she participates. Unless an employee’s participation is discontinued, his or her purchase right will be exercised automatically at the end of each purchase period at the applicable purchase price.
Purchase Price
The purchase price per share at which shares of our common stock are sold on each purchase date during an offering period will not be less than the lower of (i) 85% of the fair market value of a share of our common stock on the first day of the offering period or (ii) 85% of the fair market value of a share of our common stock on the purchase date. As of March 31, 2016, the closing price of our common stock as reported on the NASDAQ Global Select Market was $4.00 per share.
Payment of Purchase Price; Payroll Deductions
The purchase of shares during an offering period generally will be funded by a participant’s payroll deductions accumulated during the offering period. A participant may change his or her rate of contributions, as determined by the Plan Administrator in the offering. All contributions made for a participant are credited to his or her account under the Amended 2000 Purchase Planoutstanding and deposited with our general funds.
Purchase Limits
In connection with each offering made under the Amended 2000 Purchase Plan, the Plan Administrator may specify (i) a maximum number of shares of our common stock that may be purchased by any participant pursuant to such offering, (ii) a maximum number of shares of our common stock that may be purchased by any participant on any purchase date pursuant to such offering, (iii) a maximum aggregate number of shares of our common stock that may be purchased by all participants pursuant to such offering, and/or (iv) a maximum aggregate number of shares of our common stock that may be purchased by all participants on any purchase date pursuant to such offering. If the aggregate purchase of shares of our common stock issuable upon exercise of purchase rights granted under such offering would exceed any such maximum aggregate number, then the Plan Administrator will make a pro rata allocation of available shares in a uniform and equitable manner.
Withdrawal
Participants may withdraw from an offering by delivering a withdrawal form to us and terminating their contributions. Such withdrawal may be elected at any time prior to the end of an offering, except as otherwise provided by the Plan Administrator. Upon such withdrawal, we will distribute to the employee his or her accumulated but unused contributions without interest, and such employee’s right to participate in that offering will terminate. However, an employee’s withdrawal from an offering does not affect such employee’s eligibility to participate in any other offerings under the Amended 2000 Purchase Plan.
Termination of Employment
A participant’s rights under any offering under the Amended 2000 Purchase Plan will terminate immediately if the participant either (i) is no longer employed by us or any of our parent or subsidiary companies (subject to any post-employment participation period required by law) or (ii) is otherwise no longer eligible to participate. In such event, we will distribute to the participant his or her accumulated but unused contributions without interest.
Restrictions on Transfer
Rights granted under the Amended 2000 Purchase Plan are not transferable except by will, by the laws of descent and distribution, or if permitted by us, by a beneficiary designation. During a participant’s lifetime, such rights may only be exercised by the participant.
Changes in Capitalization
In the event of certain changes in our capitalization, the Plan Administrator will appropriately adjust: (i) the class(es) and maximum number of shares subject to the Amended 2000 Purchase Plan; (ii) the class(es) and number of shares subject to, and the purchase price applicable to outstanding purchase rights; and (iii) the class(es) and number of shares that are the subject of any purchase limits under each ongoing offering.
Effect of Certain Corporate Transactions
In the event of a corporate transaction (as defined in the Amended 2000 Purchase Plan and described below), (i) any surviving or acquiring corporation (or its parent company) will assume outstanding purchase rights granted under the Amended 2000 Purchase Plan or will substitute similar rights (including a right to acquire the same consideration paid to the stockholders in the corporate transaction) for such outstanding purchase rights, or (ii) if any surviving or acquiring corporation (or its parent company) refuses to assume such outstanding purchase rights or to substitute similar rights for such outstanding purchase rights, then, as determined by the Plan Administrator in its sole discretion, such outstanding purchase rights may continue in full force and effect or the participants’ accumulated contributions may be used to purchase shares of our common stock immediately prior to the corporate transaction under the ongoing offering and such purchase rights thereafter terminated.
For purposes of the Amended 2000 Purchase Plan, a corporate transaction generally will be deemed to occur in the event of: (i) a dissolution or liquidation of the Company; (ii) a sale, lease or other disposition of all or substantially all of the assets of the Company; (iii) a merger, consolidation or similar transaction in which the Company is not the surviving corporation; (iv) a reverse merger, consolidation or similar transaction in which the Company is the surviving corporation but the shares of common stock outstanding immediately prior to such transaction are converted into other property by virtue of such transaction; or (v) an acquisition of the beneficial ownership of securities of the Company representing at least 50% of the combined voting power entitled to vote
inon the
electionproposal is required to approve the amendment to the Certificate of
membersIncorporation to declassify the Board to provide for annual elections by the 2020 Annual Meeting of Stockholders. Abstentions and broker non-votes, if any, will have the effect of votes against this proposal.If our stockholders approve the proposed amendment to the Certificate of Incorporation, it will become effective upon filing with the Secretary of State of the Board.
Duration, Amendment and Termination
The Plan Administrator may amend or terminateState of Delaware a certificate setting forth the
Amended 2000 Purchase Plan at any time. However, except in regard to certain capitalization adjustments, any such amendment,
must be approved by our stockholders if such approval is required by applicable law or listing requirements.which we anticipate doing as soon as practicable following stockholder approval.THE BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” PROPOSAL 3.
30 Exelixis, Inc.
Any outstanding purchase rights granted before an amendment or terminationProposal 4 | Advisory Vote on the Compensation of the Amended 2000 Purchase Plan will not be materially impaired by any such amendment or termination, except (i) with the consent of the employee to whom such purchase rights were granted, (ii) as necessary to comply with applicable laws, listing requirements or governmental regulations (including Section 423 of the Code), or (iii) as necessary to obtain or maintain favorable tax, listing or regulatory treatment.
Notwithstanding anything in the Amended 2000 Purchase Plan or any offering to the contrary, the Plan Administrator will be entitled to: (i) establish the exchange ratio applicable to amounts withheld in a currency other than U.S. dollars; (ii) permit contributions in excess of the amount designated by a participant in order to adjust for mistakes in the processing of properly completed contribution elections; (iii) establish reasonable waiting and adjustment periods and/or accounting and crediting procedures to ensure that amounts applied toward the purchase of our common stock for each participant properly correspond with amounts withheld from the participant’s contributions; (iv) amend any outstanding purchase rights or clarify any ambiguities regarding the terms of any offering to enable such purchase rights to qualify under and/or comply with Section 423 of the Code; and (v) establish other limitations or procedures as the Plan Administrator determines in its sole discretion advisable that are consistent with the Amended 2000 Purchase Plan. Any such actions by the Plan Administrator will not be considered to alter or impair any purchase rights granted under an offering as they are part of the initial terms of each offering and the purchase rights granted under each offering.
Federal Income Tax Information
The following is a summary of the principal United States federal income tax consequences to participants and us with respect to participation in the Amended 2000 Purchase Plan. This summary is not intended to be exhaustive and does not discuss the income tax laws of any local, state or foreign jurisdiction in which a participant may reside. The information is based upon current federal income tax rules and therefore is subject to change when those rules change. Because the tax consequences to any participant may depend on his or her particular situation, each participant should consult the participant’s tax adviser regarding the federal, state, local, and other tax consequences of the grant or exercise of a purchase right or the sale
Named Executive Officers
or other disposition of common stock acquired under the Amended 2000 Purchase Plan. The Amended 2000 Purchase Plan is not qualified under the provisions of Section 401(a) of the Code and is not subject to any of the provisions of the Employee Retirement Income Security Act of 1974, as amended.
Rights granted under the Amended 2000 Purchase Plan are intended to qualify for favorable federal income tax treatment associated with rights granted under an employee stock purchase plan which qualifies under the provisions of Section 423 of the Code.
A participant will be taxed on amounts withheld for the purchase of shares of our common stock as if such amounts were actually received. Otherwise, no income will be taxable to a participant as a result of the granting or exercise of a purchase right until a sale or other disposition of the acquired shares. The taxation upon such sale or other disposition will depend upon the holding period of the acquired shares.
If the shares are sold or otherwise disposed of more than two years after the beginning of the offering period and more than one year after the shares are transferred to the participant, then the lesser of the following will be treated as ordinary income: (i) the excess of the fair market value of the shares at the time of such sale or other disposition over the purchase price; or (ii) the excess of the fair market value of the shares as of the beginning of the offering period over the purchase price (determined as of the beginning of the offering period). Any further gain or any loss will be taxed as a long-term capital gain or loss.
If the shares are sold or otherwise disposed of before the expiration of either of the holding periods described above, then the excess of the fair market value of the shares on the purchase date over the purchase price will be treated as ordinary income at the time of such sale or other disposition. The balance of any gain will be treated as capital gain. Even if the shares are later sold or otherwise disposed of for less than their fair market value on the purchase date, the same amount of ordinary income is attributed to the participant, and a capital loss is recognized equal to the difference between the sales price and the fair market value of the shares on such purchase date. Any capital gain or loss will be short-term or long-term, depending on how long the shares have been held.
There are no federal income tax consequences to us by reason of the grant or exercise of rights under the Amended 2000 Purchase Plan. We are entitled to a deduction to the extent amounts are taxed as ordinary income to a participant for shares sold or otherwise disposed of before the expiration of the holding periods described above (subject to the requirement of reasonableness and the satisfaction of tax reporting obligations).
Amended 2000 Purchase Plan Benefits
Participation in the Amended 2000 Purchase Plan is voluntary and each eligible employee will make his or her own decision regarding whether and to what extent to participate in the Amended 2000 Purchase Plan. In addition, the Board and the Compensation Committee have not granted any purchase rights under the Amended 2000 Purchase Plan that are subject to stockholder approval of this Proposal 3. Accordingly, the benefits or amounts that will be received by or allocated to our executive officers and other employees under the Amended 2000 Purchase Plan are not determinable. Our non-employee directors will not be eligible to participate in the Amended 2000 Purchase Plan.
2000 Purchase Plan Benefits
The following table sets forth, for each of the individuals and groups indicated, the total number of shares of our common stock that have been purchased under the 2000 Purchase Plan as of March 31, 2016.
|
| | | |
Name and Position | | Number of Shares |
Michael M. Morrissey, Ph.D.
President and Chief Executive Officer
| | 2,056 |
|
Christopher J. Senner
Executive Vice President and Chief Financial Officer
| | — |
|
Deborah Burke
Senior Vice President, Finance and Controller
| | 5,799 |
|
Jeffrey J. Hessekiel, J.D.
Executive Vice President, General Counsel and Secretary
| | — |
|
Peter Lamb, Ph.D.
Executive Vice President, Scientific Strategy and Chief Scientific Officer
| | 1,968 |
|
Gisela M. Schwab, M.D.
President, Product Development and Medical Affairs and Chief Medical Officer
| | 49,842 |
|
All current executive officers as a group | | 53,866 |
|
All current directors who are not executive officers as a group (1) | | 5,711 |
|
Each nominee for election as a director: | | |
Carl B. Feldbaum, Esq. | | — |
|
Alan M. Garber, M.D., Ph.D. | | — |
|
Vincent T. Marchesi, M.D., Ph.D. | | — |
|
Each associate of any executive officers, current directors or director nominees | | — |
|
Each other person who received or is to receive 5% of purchase rights | | — |
|
All employees, including all current officers who are not executive officers, as a group | | 994,817 |
|
____________________
| |
(1) | Includes shares purchased under the 2000 Purchase Plan by Dr. Scangos who served as our President and Chief Executive Officer from October 1996 to July 2010. |
Equity Compensation Plan Information
The following table provides certain information about our common stock that may be issued upon the exercise of stock options and other rights under all of our existing equity compensation plans as of January 1, 2016, which consists of our 2000 Equity Incentive Plan (the "2000 Equity Plan"), our 2000 Non-Employee Directors’ Stock Option Plan ("the Director Plan"), the 2000 Purchase Plan, our 2011 Equity Incentive Plan (the "2011 Equity Plan"), our 2014 Equity Incentive Plan (the "2014 Equity Plan") and our 401(k) Retirement Plan (the "401(k) Plan"):
|
| | | | | | | | | | | | |
| Plan Category | | Number of securities to be issued upon exercise of outstanding options, warrants and rights | | Weighted-average exercise price of outstanding options, warrants and rights (1) | | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) | |
| | | (a) | | (b) | | (c) | |
| Equity compensation plans approved by stockholders: | | | | | | | |
| 2000 Equity Plan | | 3,416,186 |
| | $ | 7.83 |
| | — |
| (2 | ) |
| Director Plan | | 1,898,906 |
| | $ | 6.55 |
| | — |
| (2 | ) |
| 2000 Purchase Plan | | — |
| | — |
| | 1,046,959 |
| |
| 2011 Equity Plan | | 5,450,205 |
| | $ | 5.53 |
| | — |
| (2 | ) |
| 2014 Equity Plan | | 17,662,745 |
| | $ | 2.95 |
| | 8,041,842 |
| |
| Equity compensation plans not approved by stockholders: | |
|
| | | | | |
| 401(k) Plan (3) | | — |
| | — |
| | 450,042 |
| |
| Total | | 28,428,042 |
| | $ | 4.19 |
| | 9,538,843 |
| |
____________________
| |
(1) | The weighted average exercise price does not take into account the shares subject to outstanding RSUs which have no exercise price. |
| |
(2) | The 2000 Equity Plan, Director Plan and 2011 Equity Plan have been superseded by the 2014 Equity Plan. Therefore, shares are no longer available for issuance pursuant to those equity compensation plans. |
| |
(3) | Represents shares of our common stock issuable pursuant to the 401(k) Plan. We sponsor a 401(k) Plan whereby eligible employees may elect to contribute up to the lesser of 50% of their annual compensation or the statutorily prescribed annual limit allowable under Internal Revenue Service regulations. The 401(k) Plan permits us to make matching contributions on behalf of all participants. We match 100% of the first 3% of participant contributions into the 401(k) Plan in the form of our common stock. |
PROPOSAL 4
ADVISORY VOTEONTHE COMPENSATIONOFTHE NAMED EXECUTIVE OFFICERSDVISORY VOTEONTHE COMPENSATIONOFTHE NAMED EXECUTIVE OFFICERS
Our stockholders are entitled to vote to approve, on an advisory basis, the compensation, as disclosed in this Proxy Statement, of our Chief Executive Officer, Chief Financial Officer and the other executive officers appearing in the table entitled “Summary Compensation Table for Fiscal 2015”Table” later in this Proxy Statement (collectively, the Named Executive Officers). This vote is not intended to address any specific item of compensation, but rather the overall compensation of our Named Executive Officers and the philosophy, policies and practices described in this Proxy Statement.
The compensation of our Named Executive Officers subject to the vote is disclosed in “Compensation Discussion and Analysis,” and the compensation tables and the related narrative disclosure contained in this Proxy Statement. This year, we have also included a discussion of our stockholder engagement effort focused on executive compensation during fiscal 2018, as well as a discussion of changes made by our Compensation Committee to the executive compensation program for fiscal 2018 in response to that engagement effort. As discussed in the “Compensation Discussion and Analysis” section of this Proxy Statement, the success of biopharmaceuticalbiotechnology companies is significantly influenced by their work forces. We believe it is critical to our business that we retain our core team of highly qualified employees, including our executive officers. Large pharmaceutical and biopharmaceuticalbiotechnology companies and strong local competitors have aggressively recruited our executives and other skilled employees, with the most critical positions at our company among those that are the most in demand. In light of these circumstances, we have designed our executive compensation program to help attract and retain highly qualified individuals with relevant experience in the biopharmaceuticalbiotechnology industry to manage the varied aspects of our evolving business. The primary objective of our executive compensation program is to retain and motivate our core team of highly qualified employees, including our Named Executive Officers, and align their compensation with our critical business objectives and performance, as well as with the interests of our stockholders. In support of this objective, our fiscal 2015 executive compensation program continues to reflect our decision to focus our proprietary resources and development and commercialization efforts primarily on cabozantinib, an internally-discovered inhibitor of multiple receptor tyrosine kinases, in various tumor indications.
The Board encourages our stockholders to review the compensation tables and read the disclosures set forth in the “Compensation Discussion and Analysis” section of this Proxy Statement to reviewthat describe our executive compensation program and the correlation between compensation and performance, as well as compensation actions taken in 2015.of our Named Executive Officers for fiscal 2018. The Board believes that our executive compensation program effectively aligns executive paycompensation with our performance and results in the attraction and retention of highly talented executives.
Accordingly, the Board recommends that our stockholders vote FOR the following resolution:
“RESOLVED, that the compensation paid to the our Named Executive Officers, as disclosed pursuant to Item 402 of Regulation S-K, including the Compensation Discussion and Analysis, compensation tables and narrative discussion, is hereby APPROVED.”
Required Vote and Board of Directors Recommendation
Advisory approval of Proposal 4 requires the affirmative vote of a majority of the shares present or represented by proxy and entitled to vote at the Annual Meeting. Abstentions will be counted toward the tabulation of votes cast on the proposal and will have the same effect as votes against this proposal. Broker non-votes will have no effecteffect.
Our stockholders have expressed a preference, and will not be counted towardsour Board has determined, to hold an advisory vote on executive compensation annually. We are presenting this Proposal 4 as required by Section 14A of the vote total.
Exchange Act. Our Board believes that approval of Proposal 4 is in our best interests and the best interests of our stockholders for the reasons stated above. Because the vote is advisory, it is not binding on the Board or on us. Nevertheless, the views expressed by our stockholders, whether through this vote or otherwise, are important to management and the Board and, accordingly, the Compensation Committee and Board intend to consider the results of this vote in making determinations in the future regarding executive compensation arrangements. Your vote will serve as an additional tool to guide the Compensation Committee and Board in continuing to improve the alignment of our executive compensation programs with business objectives and performance and with the interests of our stockholders.
THE BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” PROPOSAL 4.
T2019 Proxy Statement HE31
BOARDOF DIRECTORS RECOMMENDSA VOTE FOR PROPOSAL 4.
SSECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENTECURITY OWNERSHIPOF CERTAIN BENEFICIAL OWNERSAND MANAGEMENT
The following table sets forth certain information regarding the ownership of our common stock as of March 11, 2016,February 28, 2019, (except as noted) by: (i) each director and nominee for director; (ii) each of the executive officers named in the Summary Compensation Table; (iii) all current executive officers and directors of Exelixis as a group; and (iv) all those known by us to be beneficial owners of more than five percent of our common stock.
|
| | | | | | | |
| | | Beneficially Owned(1) |
| Name of Beneficial Owner | | Number of Shares of Common Stock | | Percentage of Total |
| Executive Officers and Directors | | | | |
| Michael M. Morrissey, Ph.D. (2) | | 3,661,688 |
| | 1.6 | % |
| Christopher J. Senner (3) | | 33,282 |
| | * | |
| Deborah Burke (4) | | 527,265 |
| | * | |
| Jeffrey J. Hessekiel, J.D. (5) | | 609,742 |
| | * | |
| Peter Lamb, Ph.D. (6) | | 1,062,111 |
| | * | |
| Gisela M. Schwab, M.D. (7) | | 1,868,892 |
| | * | |
| Charles Cohen, Ph.D. (8) | | 554,454 |
| | * | |
| Carl B. Feldbaum, Esq. (9) | | 302,001 |
| | * | |
| Alan M. Garber, M.D., Ph.D. (10) | | 309,454 |
| | * | |
| Vincent T. Marchesi, M.D., Ph.D. (11) | | 391,283 |
| | * | |
| Stelios Papadopoulos, Ph.D. (12) | | 1,549,208 |
| | * | |
| George Poste, D.V.M., Ph.D., FRS (13) | | 328,094 |
| | * | |
| George A. Scangos, Ph.D. (14) | | 2,215,701 |
| | * | |
| Lance Willsey, M.D. (15) | | 805,691 |
| | * | |
| Jack L. Wyszomierski (16) | | 322,653 |
| | * | |
| All current directors, executive officers as a group (14 persons) (17) | | 14,014,254 |
| | 5.9 | % |
| 5% Stockholders | | | | |
FMR LLC (18) 245 Summer Street Boston, Massachusetts 02210 | | 34,082,482 |
| | 14.9 | % |
T. Rowe Price Associates, Inc. (19) 100 E. Pratt Street Baltimore, Maryland 21202 | | 25,083,170 |
| | 11.0 | % |
Meditor Group Ltd. (20) Penboss Building 50 Parliament Street Hamilton HM12, Bermuda | | 22,070,213 |
| | 9.7 | % |
BlackRock, Inc. (21) 55 East 52nd Street New York, New York 10055 | | 15,871,810 |
| | 6.9 | % |
The Vanguard Group (22) 100 Vanguard Blvd. Malvern, Pennsylvania 19355 | | 15,619,701 |
| | 6.8 | % |
*Less than one percent
| | | | | | | | |
| | | Beneficially Owned (1) | |
Name of Beneficial Owner | | Number of
Shares of
Common Stock | | | Percentage
of Total | |
Executive Officers and Directors | | | | | | | | |
Michael M. Morrissey, Ph.D. (2) | | | 3,222,949 | | | | 1.1 | % |
Gisela M. Schwab, M.D. (3) | | | 1,860,620 | | | | * | |
Christopher J. Senner (4) | | | 646,671 | | | | * | |
Jeffrey J. Hessekiel, J.D. (5) | | | 882,434 | | | | * | |
Peter Lamb, Ph.D. (6) | | | 1,256,800 | | | | * | |
Charles Cohen, Ph.D. (7) | | | 356,033 | | | | * | |
Carl B. Feldbaum, Esq. (8) | | | 232,725 | | | | * | |
Maria C. Freire, Ph.D. (9) | | | 47,644 | | | | * | |
Alan M. Garber, M.D., Ph.D. (10) | | | 352,943 | | | | * | |
Vincent T. Marchesi, M.D., Ph.D. (11) | | | 303,477 | | | | * | |
Stelios Papadopoulos, Ph.D. (12) | | | 1,347,665 | | | | * | |
George Poste, D.V.M., Ph.D., FRS (13) | | | 310,620 | | | | * | |
George A. Scangos, Ph.D. (14) | | | 1,627,970 | | | | * | |
Julie A. Smith (15) | | | 98,041 | | | | * | |
Lance Willsey, M.D. (16) | | | 744,073 | | | | * | |
Jack L. Wyszomierski (17) | | | 375,057 | | | | * | |
All current directors, executive officers as a group (17 persons) (18) | | | 14,037,144 | | | | 4.5 | % |
5% Stockholders | | | | | | | | |
BlackRock, Inc. (19) 55 East 52nd Street New York, New York 10055 | | | 30,584,751 | | | | 10.2 | % |
The Vanguard Group (20) 100 Vanguard Blvd. Malvern, Pennsylvania 19355 | | | 27,335,719 | | | | 9.1 | % |
FMR LLC (21) 245 Summer Street Boston, Massachusetts 02210 | | | 21,317,889 | | | | 7.1 | % |
Meditor Group Ltd. (22) Wessex House, 3rd Floor 45 Reid Street Hamilton HM12, Bermuda | | | 15,966,038 | | | | 5.3 | % |
| (1) | This table is based upon information supplied by executive officers and directors and upon information gathered by us about principal stockholders known to us. Unless otherwise indicated in the footnotes to this table and subject to community property laws where applicable, we believe that each of the stockholders named in this table has sole voting |
32 Exelixis, Inc.
Security Ownership of Certain Beneficial Owners and Management
| and investment power with respect to the shares indicated as beneficially owned. Applicable percentages are based on 228,623,481300,996,666 shares outstanding on March 11, 2016,February 28, 2019, adjusted as required by rules promulgated by the SEC. The percentage of beneficial ownership as to any person as of a particular date is calculated by dividing the number of shares beneficially owned by such person, which includes the number of shares as to which such person has the right to acquire voting or investment power within 60 days of March 11, 2016,February 28, 2019, by the sum of the number of shares outstanding as of such date plus the number of shares as to which such person has the right to acquire voting or investment power within 60 days of March 11, 2016.February 28, 2019. Consequently, the denominator for calculating beneficial ownership percentages may be different for each beneficial owner. |
| |
| (2) | Includes 174,698193,998 shares held by Michael M. Morrissey and Meghan D. Morrissey, Trustees of the Morrissey Family Living Trust dated July 21, 1994, as amended. Also includes 3,471,3753,011,450 shares Dr. Morrissey has the right to acquire pursuant to options exercisable within 60 days of March 11, 2016.February 28, 2019. Also includes 15,61517,501 shares held by Mr.Dr. Morrissey under our 401(k) Retirement Plan, or 401(k) Plan, determined based upon information provided in plan statements. |
| |
| (3) | Includes 5551,595,713 shares Dr. Schwab has the right to acquire pursuant to options exercisable within 60 days of February 28, 2019. Also includes 14,644 shares held by Dr. Schwab under our 401(k) Plan, determined based upon information provided in plan statements. |
| (4) | Includes 554,062 shares Mr. Senner has the right to acquire pursuant to options exercisable within 60 days of February 28, 2019. Also includes 2,534 shares held by Mr. Senner under our 401(k) Plan, determined based upon information provided in plan statements. |
| |
(4) | Includes 500,075 shares Ms. Burke has the right to acquire pursuant to options exercisable and shares issuable pursuant to RSUs scheduled to vest within 60 days of March 11, 2016. Also includes 3,465 shares held by Ms. Burke under our 401(k) Plan, determined based upon information provided in plan statements. |
| |
| (5) | Represents 579,375Includes 693,374 shares Mr. Hessekiel has the right to acquire pursuant to options exercisable within 60 days of March 11, 2016.February 28, 2019. Also includes 864 shares held by Mr. Hessekiel under our 401(k) Plan, determined based upon information provided in plan statements. |
| |
| (6) | Includes 989,8121,172,092 shares Dr. Lamb has the right to acquire pursuant to options exercisable within 60 days of March 11, 2016.February 28, 2019. Also includes 14,94916,885 shares held by Dr. Lamb under our 401(k) Plan, determined based upon information provided in plan statements. |
| |
| (7) | Includes 1,724,187 shares Dr. Schwab has the right to acquire pursuant to options exercisable within 60 days of March 11, 2016. Also includes 12,689 shares held by Dr. Schwab under our 401(k) Plan, determined based upon information provided in plan statements. |
| |
(8) | Includes 316,646335,054 shares Dr. Cohen has the right to acquire pursuant to options exercisable within 60 days of March 11, 2016, 10,000February 28, 2019, 7,792 of which would be subject to repurchase by us, if so exercised, and 3,458 shares issuable pursuant to RSUs scheduled to vest within 60 days of March 11, 2016.exercised. |
| (8) | |
(9) | Includes 259,796224,204 shares Mr. Feldbaum has the right to acquire pursuant to options exercisable within 60 days of March 11, 2016, 10,000February 28, 2019, 7,792 of which would be subject to repurchase by us, if so exercised, and 2,172exercised. |
| (9) | Includes 3,530 shares issuable pursuant to RSUs scheduled tothat will vest within 60 days of March 11, 2016.February 28, 2019 and 44,114 shares Dr. Freire has the right to acquire pursuant to options exercisable within 60 days of February 28, 2019, 32,209 of which would be subject to repurchase by us, if so exercised. |
| |
| (10) | Includes 258,206291,614 shares Dr. Garber has the right to acquire pursuant to options exercisable within 60 days of March 11, 2016, 10,000February 28, 2019, 7,792 of which would be subject to repurchase by us, if so exercised, and 2,637 shares issuable pursuant to RSUs scheduled to vest within 60 days of March 11, 2016.exercised. |
| |
| (11) | Includes 281,254177,564 shares Dr. Marchesi has the right to acquire pursuant to options exercisable within 60 days of March 11, 2016, 10,000February 28, 2019, 3,969 of which would be subject to repurchase by us, if so exercised, and 3,346 shares issuable pursuant to RSUs scheduled to vest within 60 days of March 11, 2016.exercised. |
| |
| (12) | Includes 10,000 shares held by Fondation Santé, of which Dr. Papadopoulos is a co-trustee. Also includes 322,041313,351 shares Dr. Papadopoulos has the right to acquire pursuant to options exercisable within 60 days of March 11, 2016, 10,000February 28, 2019, 3,969 of which would be subject to repurchase by us, if so exercised, and 3,767 shares issuable pursuant to RSUs scheduled to vest within 60 days of March 11, 2016.exercised. |
| |
| (13) | Includes 260,912227,222 shares Dr. Poste has the right to acquire pursuant to options exercisable within 60 days of March 11, 2016, 10,000February 28, 2019, 3,969 of which would be subject to repurchase by us, if so exercised, and 3,457 shares issuable pursuant to RSUs scheduled to vest within 60 days of March 11, 2016.exercised. |
| |
| (14) | Includes 8,963 shares held by Dr. Scangos and Leslie S. Wilson, as Trustees of The Jennifer Wilson Scangos Trust, and 8,963 shares held by Dr. Scangos and Leslie S. Wilson, as Trustees of The Katherine Wilson Scangos Trust. Also includes 886,684241,344 shares Dr. Scangos has the right to acquire pursuant to options exercisable within 60 days of March 11, 2016, 10,000February 28, 2019, 7,792 of which would be subject to repurchase by us, if so exercised, and 1,595 shares issuable pursuant to RSUs scheduled to vest within 60 days of March 11, 2016.exercised. Also includes 5,669 shares held by Dr. Scangos under our 401(k) Plan, determined based upon information provided in plan statements. |
| |
| (15) | Includes 307,39297,278 shares Ms. Smith has the right to acquire pursuant to options exercisable within 60 days of February 28, 2019, 26,394 of which would be subject to repurchase by us, if so exercised. |
| (16) | Includes 325,800 shares Dr. Willsey has the right to acquire pursuant to options exercisable within 60 days of March 11, 2016, 10,000February 28, 2019, 7,792 of which would be subject to repurchase by us, if so exercised, and 3,324 shares issuable pursuant to RSUs scheduled to vest within 60 days of March 11, 2016.exercised. |
| (17) | |
(16) | Includes 263,653281,213 shares Mr. Wyszomierski has the right to acquire pursuant to options exercisable within 60 days of March 11, 2016, 10,000February 28, 2019, 3,969 of which would be subject to repurchase by us, if so exercised, and 3,036 shares issuable pursuant to RSUs scheduled to vest within 60 days of March 11, 2016.exercised. |
2019 Proxy Statement 33
| (18) | |
(17) | Total number of shares includes 4,093,3194,083,690 shares of common stock held by our current directors and executive officers as of March 11, 2016,February 28, 2019, and entities affiliated with such directors and executive officers. Also includes 10,421,4083,530 shares issuable pursuant to RSUs that will vest within 60 days of February 28, 2019. Also includes 9,881,411 shares our directors and executive officers have the right to acquire pursuant to options exercisable and 26,792 shares issuable pursuant to RSUs scheduled to vest within 60 days of March 11, 2016, 90,000February 28, 2019, 113,439 of which would be subject to repurchase by us, if so exercised. Also includes 52,94268,513 shares held by our current directors and executive officers under our 401(k) Plan, determined based upon information provided in plan statements. |
| (19) | |
(18) | FMR LLCBlackRock, Inc. reported that (a) it has sole voting power over 29,407,614 of such shares, and that it has sole dispositive power over all of the shares. The information is based solely on a Schedule 13G/A, filed with respect to 4,646,452the SEC on January 28, 2019, which provides information only as of December 31, 2018, and, consequently, the beneficial ownership of BlackRock may have changed between December 31, 2018 and February 28, 2019. |
| (20) | The Vanguard Group reported that it has sole voting power over 163,947 of such shares, shared voting power over 39,980 of these shares, (b)sole dispositive power over 27,162,112 of such shares and shared dispositive power over 173,607 of such shares. The information is based solely on a Schedule 13G/A, filed with the SEC on February 11, 2019, which provides information only as of December 31, 2018, and, consequently, the beneficial ownership of Vanguard may have changed between December 31, 2018 and February 28, 2019. |
| (21) | FMR LLC reported that it has sole voting power over 6,894,567 of such shares, and that FMR LLC and Abigail P. Johnson each have sole dispositive power ofover all of these shares and (c) Fidelity Growth Company Fund has sole voting power with respect to 14,514,689 of thesethe shares. Ms. Johnson is a Director, the Vice Chairman and the Chief Executive Officer and President of FMR LLC. FMR LLC also reports that FMR Co., Inc. beneficially owns 5% or greater of our common stock. The foregoing information is based solely on a Schedule 13G/A filed with the SEC on |
February 12, 2016, which provides information only as of December 31, 2015, and, consequently, the beneficial ownership of these reporting persons may have changed between December 31, 2015, and March 11, 2016.
| |
(19) | These shares are owned by various individual and institutional investors for which T. Rowe Price Associates, Inc., or Price Associates, serves as investment adviser. Price Associates reported that it has sole dispositive power over all of these shares and sole voting power over 3,316,900 of these shares. The foregoing information is based solely on a Schedule 13G/A filed with the SEC on February 12, 2016,13, 2019, which provides information only as of December 31, 2015,2018, and, consequently, the beneficial ownership of Price Associatesthese reporting persons may have changed between December 31, 2015,2018 and March 11, 2016.February 28, 2019. |
| (22) | |
(20) | These shares are beneficially owned by Meditor Group Ltd. (Meditor), or Meditor, and Meditor European Master Fund Ltd. (MEMF), or MEMF, an investment management client and subsidiary of Meditor. Meditor reported that it and MEMF each have shared voting and dispositive power over the shares. The foregoing information is based solely on a Schedule 13G/A filed with the SEC on January 25, 2016, which provides information only asall of December 31, 2015, and, consequently, the beneficial ownership of Meditor and MEMF may have changed between December 31, 2015, and March 11, 2016. |
| |
(21) | BlackRock, Inc. reported that it has sole dispositive power over all such shares and sole voting power over 15,418,533 of such shares. The information is based solely on a Schedule 13G/A filed with the SEC on February 10, 2016,January 2, 2019, which provides information only as of December 31, 2015,2018, and, consequently, the beneficial ownership of BlackRockMeditor and MEMF may have changed between December 31, 2015,2018 and March 11, 2016.February 28, 2019. |
| |
(22) | The Vanguard Group reported that it has sole voting power over 485,766 of such shares, sole dispositive power over 15,145,335 of such shares and shared dispositive power over 474,366 of such shares. The information is based solely on a Schedule 13G/A, filed with the SEC on February 10, 2016, which provides information only as of December 31, 2015, and, consequently, the beneficial ownership of Vanguard may have changed between December 31, 2015, and March 11, 2016. |
EXECUTIVE34 OExelixis, Inc.
Executive Officers
FFICERSEXECUTIVE OFFICERS
The following chart sets forth certain information regarding our executive officers as of March 31, 2016:
25, 2019: |
| | | | | |
Name | | Age | | Position |
Michael M. Morrissey, Ph.D. (1) | | 55 |
58 | | President and Chief Executive Officer |
Christopher J. Senner | | 48 |
| | Executive Vice President and Chief Financial Officer |
Jeffrey J. Hessekiel, J.D. | | 47 |
| | Executive Vice President, General Counsel and Secretary |
Peter Lamb, Ph.D. | | 55 |
| | Executive Vice President, Scientific Strategy and Chief Scientific Officer |
Gisela M. Schwab, M.D. | | 59 |
62 | | President, Product Development and Medical Affairs and Chief Medical Officer |
Christopher J. Senner | | 51 | | Executive Vice President and Chief Financial Officer |
Patrick J. Haley | | 43 | | Senior Vice President, Commercial |
Jeffrey J. Hessekiel, J.D. | | 50 | | Executive Vice President and General Counsel |
Peter Lamb, Ph.D. | | 58 | | Executive Vice President, Scientific Strategy and Chief Scientific Officer |
| (1) | Please see “Class III Directors Continuing in Office Untiluntil the 20172020 Annual Meeting” in this Proxy Statement for Dr. Morrissey’s biography. |
|
Gisela M. Schwab, M.D. President, Product Development and Medical Affairs and Chief Medical Officer |
Gisela M. Schwab, M.D., has served as President, Product Development and Medical Affairs and Chief Medical Officer since February 2016. Previously she served as Executive Vice President and Chief Medical Officer from January 2008 to February 2016 and as Senior Vice President and Chief Medical Officer from September 2006 to January 2008. From 2002 to 2006, she held the position of Senior Vice President and Chief Medical Officer at Abgenix, Inc., a human antibody-based drug development company. She also served as Vice President, Clinical Development, at Abgenix from 1999 to 2001. Prior to working at Abgenix, from 1992 to 1999, she held positions of increasing responsibility at Amgen Inc., most recently as Director of Clinical Research and Hematology/Oncology Therapeutic Area Team Leader. From August 2011 through July 2014, Dr. Schwab served as a member of the board of directors of Topotarget A/S, a publicly-held biopharmaceutical company. Since June 2012 she has served as a member of the board of directors of Cellerant Therapeutics, Inc. a privately-held biopharmaceutical company and since March 2015, she has served as a member of the board of directors of Nordic Nanovector A.S., a Norwegian biotechnology company. She received her Doctor of Medicine degree from the University of Heidelberg, trained at the University of Erlangen-Nuremberg and the National Cancer Institute and is board certified in internal medicine and hematology and oncology. |
|
Christopher J. Senner Executive Vice President and Chief Financial Officer |
Christopher J. Senner, has served as Executive Vice President and Chief Financial Officer (and in such capacity, as our principal financial officer and principal accounting officer, as defined under applicable securities laws) since July 2015. Prior to joining Exelixis, Mr. Senner served as Vice President, Corporate Finance for Gilead Sciences, Inc., a biopharmaceutical company, from March 2010 to July 2015, where he was accountable for controllership, tax, treasury and corporate and operational financial planning. Mr. Senner previously spent eighteen years at Wyeth, a pharmaceutical company acquired by Pfizer Inc. in 2009, in a variety of financial roles with increasing responsibility, most notably as Chief Financial Officer of Wyeth’s U.S. pharmaceuticals business and the BioPharma Business Unit. Mr. Senner holds a B.S. in Finance from Bentley College. |
|
P.J. Haley Senior Vice President, Commercial |
P.J. Haley, has served as the company’s Senior Vice President, Commercial since December 2016 and has held positions of progressive Commercial leadership since September 2010, serving as Vice President, Commercial from November 2014 to November 2016, Executive Director, Sales & Marketing from September 2013 to October 2014, Senior Director, Marketing from March 2012 to August 2013, and as Director, Marketing from September 2010 to February 2012. Prior to joining Exelixis, from 2007 to 2010, he held positions of increasing responsibility at Genentech, Inc., on the Avastin marketing team, most recently Group Product Manager. Between 2003 and 2007, Mr. Haley served in various sales and marketing roles at Amgen, Inc. He served as an analyst at PWC Securities, Lehman Brothers and Accenture from 1998 to 2001. Mr. Haley holds a Masters of Business Administration from University of Michigan, Ross School of Business, and a Bachelor of Arts in Art History and Medieval and Renaissance Studies from Duke University. |
2019 Proxy Statement , has served as Executive Vice President and Chief Financial Officer (and in such capacity, as our principal financial officer and principal accounting officer, as defined under applicable securities laws) since July 2015. Prior to joining Exelixis, Mr. Senner served as Vice President, Corporate Finance for Gilead Sciences, Inc., a biopharmaceutical company, from March 2010 to July 2015, where he was accountable for controllership and operational financial planning and analysis, including research and development, manufacturing, commercial operations, and tax and treasury planning. Mr. Senner previously spent eighteen years at Wyeth in a variety of financial roles with increasing responsibility, most notably as Chief Financial Officer of Wyeth's U.S. pharmaceuticals business and the BioPharma Business Unit. Mr. Senner and holds a Bachelor of Science in Finance from Bentley College. A prior disclosure indicating that Mr. Senner was a Certified Public Accountant was inaccurate due to an administrative error in the related filing. Mr. Senner is not a Certified Public Accountant.35
|
Jeffrey J. Hessekiel, J.D. Executive Vice President and General Counsel |
Jeffrey J. Hessekiel, J.D., has served as Executive Vice President and General Counsel since February 2014 and as its Secretary from October 2014 to September 2017. From 2012 to 2014, he held the position of Senior Counsel at Arnold & Porter Kaye Scholer LLP, where he advised emerging growth and public companies, primarily in the life sciences sector, on complex legal issues connected with strategic transactions, healthcare compliance programs and investigations, and regulatory matters. Prior to working with Arnold & Porter, from 2002 to 2012, he held positions of increasing responsibility at Gilead Sciences, Inc., most recently as Chief Compliance & Quality Officer where he was responsible for the creation and management of Gilead’s Corporate Compliance & Quality department. From 1998 to 2002, Mr. Hessekiel held the position of Associate, working on both litigation and corporate matters for Wilson Sonsini Goodrich and Rosati PC. Mr. Hessekiel also worked as an Associate focusing on litigation matters for Heller Ehrman LLP from 1996 to 1998. Prior to joining Heller Ehrman LLP, Mr. Hessekiel also worked for several international non-governmental organizations. Mr. Hessekiel received his J.D. from The George Washington University Law School and is admitted to practice in California. Mr. Hessekiel received a B.A. in Political Science from Duke University. |
|
Peter Lamb, Ph.D. Executive Vice President, Scientific Strategy and Chief Scientific Officer |
Peter Lamb, Ph.D., has served as Executive Vice President, Scientific Strategy and Chief Scientific Officer since February 2016. Previously, he served as Executive Vice President, Discovery Research and Chief Scientific Officer from September 2009 to February 2016, as Senior Vice President, Discovery Research and Chief Scientific Officer from January 2007 until September 2009, as Vice President, Discovery Pharmacology from December 2003 until January 2007 and as Senior Director, Molecular Pharmacology and Structural Biology from October 2000 until December 2003. Prior to joining Exelixis, from June 1992 until September 2000, Dr. Lamb held positions of increasing responsibility at Ligand Pharmaceuticals, a pharmaceutical company, most recently serving as Director of Transcription Research. Dr. Lamb has held post-doctoral research fellowships at the Carnegie Institution, Department of Embryology, with Dr. S.L. McKnight and the University of Oxford with Dr. N.J. Proudfoot, working in the field of gene regulation. He has authored numerous articles in the fields of gene expression, signal transduction and oncology, and is an author on multiple issued and pending U.S. patents. He has a Ph.D. in Molecular Biology from the ICRF/University of London and a B.A. in Biochemistry from the University of Cambridge. |
36 has served asExelixis, Inc.
Compensation of Executive Vice President and General Counsel since February 2014 and as its Secretary since October 2014. From November 2012 to February 2014, he held the position of Senior Counsel at Arnold & Porter LLP, where he advised emerging growth and public companies, primarily in the life sciences sector, on complex legal issues connected with strategic transactions, healthcare compliance programs and investigations, and regulatory matters. Mr. Hessekiel also served as Acting General Counsel of Achaogen, Inc., a clinical-stage biopharmaceutical company, from May 2012 to November 2012, where he advised the executive management team on matters related to clinical and government contracts, litigation management and employment law. Prior to working at Achaogen, from December 2002 to May 2012, he held positions of increasing responsibility at Gilead Sciences, Inc., most recently as Chief Compliance & Quality Officer where he was responsible for the creation and management of Gilead’s Corporate Compliance & Quality department. From October 1998 to November 2002, Mr. Hessekiel held the position of Associate, working on both litigation and corporate matters for Wilson Sonsini Goodrich and Rosati PC. Mr. Hessekiel also worked as an Associate focusing on litigation matters for Heller Ehrman LLP from August 1996 to July 1998. Prior to joining Heller Ehrman LLP, Mr. Hessekiel also worked for several international non-governmental organizations. Mr. Hessekiel received his J.D. from The George Washington University Law School and is admitted to practice in California. Mr. Hessekiel received a B.A. in Political Science from Duke University.
Peter Lamb, Ph.DOfficers ., has served as Executive Vice President, Scientific Strategy and Chief Scientific Officer since February 2016. Previously, he served as Executive Vice President, Discovery Research and Chief Scientific Officer from September 2009 to February 2016, as Senior Vice President, Discovery Research and Chief Scientific Officer from January 2007 until September 2009, as Vice President, Discovery Pharmacology from December 2003 until January 2007 and as Senior Director, Molecular Pharmacology and Structural Biology from October 2000 until December 2003. Prior to joining Exelixis, from June 1992 until September 2000, Dr. Lamb held positions of increasing responsibility at Ligand Pharmaceuticals, a pharmaceutical company, most recently serving as Director of Transcription Research. Dr. Lamb has held post-doctoral research fellowships at the Carnegie Institution, Department of Embryology, with Dr. S.L. McKnight and the University of Oxford with Dr. N.J. Proudfoot, working in the field of gene regulation. He has authored numerous articles in the fields of gene expression, signal transduction and oncology, and is an author on multiple issued and pending U.S. patents. He has a Ph.D. in Molecular Biology from the ICRF/University of London and a B.A. in Biochemistry from the University of Cambridge.
Gisela M. Schwab, M.D.,| has served as President, Product Development and Medical Affairs and Chief Medical Officer since February 2016. Previously she served as Executive Vice President and Chief Medical Officer from January 2008 to February 2016 and as Senior Vice President and Chief Medical Officer from September 2006 to January 2008. From 2002 to 2006, she held the position of Senior Vice President and Chief Medical Officer at Abgenix, Inc., a human antibody-based drug development company. She also served as Vice President, Clinical Development, at Abgenix from 1999 to 2001. Prior to working at Abgenix, from 1992 to 1999, she held positions of increasing responsibility at Amgen Inc., most recently as Director of Clinical Research and Hematology/Oncology Therapeutic Area Team Leader. From August 2011 through July 2014, Dr. Schwab served as a member of the board of directors of Topotarget A/S, a publicly-held biopharmaceutical company. Since
June 2012 she has served as a member of the board of directors of Cellerant Therapeutics, Inc. a privately-held biopharmaceutical company and since March 2015, she has served as a member of the board of directors of Nordic Nanovector A.S., a Norwegian biotechnology company. She received her Doctor of Medicine degree from the University of Heidelberg, trained at the University of Erlangen-Nuremberg and the National Cancer Institute and is board certified in internal medicine and hematology and oncology.
COMPENSATIONOF EXECUTIVE OFFICERS
Compensation Discussion and Analysis
COMPENSATION OF EXECUTIVE OFFICERS
Compensation Discussion and Analysis
This Compensation Discussion and Analysis (CD&A) explains ourthe strategy, design, of, and decision-making related to our compensation programs and practices for our principal executive officer, our principal financial officer and our three other most highly compensated executive officers, (collectively, "Namedwho we refer to collectively as our Named Executive Officers").Officers. This Compensation Discussion and AnalysisCD&A is intended to provide perspective on the compensation information contained in the tables that follow this discussion.
For fiscal 2015,2018, our Named Executive Officers were:
Michael M. Morrissey, Ph.D., President and Chief Executive Officer
Christopher J. Senner, Executive Vice President and Chief Financial Officer
Deborah Burke, Senior Vice President, Finance and Controller*
Jeffrey J. Hessekiel, J.D., Executive Vice President, General Counsel and Secretary
Peter Lamb, Ph.D., Executive Vice President, Scientific Strategy and Chief Scientific Officer
Gisela M. Schwab, M.D., President, Product Development and Medical Affairs and Chief Medical Officer
*Ms. Burke served as our Senior Vice President and Chief Financial Officer from September 2014 to July 2015. SEC rules require that we include her as a Named Executive Officer as she served as our principal financial officer for a portion of fiscal 2015.
| | |
Named Executive Officers | | Title |
Michael M. Morrissey, Ph.D. | | President and Chief Executive Officer |
Gisela M. Schwab, M.D. | | President, Product Development and Medical Affairs and Chief Medical Officer |
Christopher J. Senner | | Executive Vice President and Chief Financial Officer |
Jeffrey J. Hessekiel, J.D. | | Executive Vice President and General Counsel |
Peter Lamb, Ph.D. | | Executive Vice President, Scientific Strategy and Chief Scientific Officer |
While the principal purpose of this Compensation Discussion and AnalysisCD&A is to discuss the compensation of our Named Executive Officers, many of the programs discussed apply to other members of senior management who, together with the Named Executive Officers, are collectively referred to as our "executives."
executive officers.Executive Summary
Exelixis is a biopharmaceuticalOur Business
We are an oncology-focused biotechnology company that discovers, develops and commercializes small molecule therapies foraims to accelerate the treatment of cancer. Our business focuses predominantly on thediscovery, development and commercialization of new medicines fordifficult-to-treat cancers. Since we were founded in 1994, four products resulting from our discovery efforts have progressed through clinical development and received regulatory approval; three have a growing commercial presence in markets worldwide, and we expect that the fourth will soon enter the marketplace in Japan. Two are derived from cabozantinib, our flagship molecule, an internally-discovered inhibitor of multiple receptor tyrosine kinases in various tumor indications, but also includes the stewardship of various compounds we discovered in our laboratoriesincluding MET, AXL, VEGF receptors and have partnered with larger pharmaceutical manufacturersRET. These are: CABOMETYX® (cabozantinib) tablets approved for further developmentadvanced renal cell carcinoma (RCC) and if warranted, commercialization. Cabozantinib was firstpreviously treated hepatocellular carcinoma (HCC), and COMETRIQ® (cabozantinib) capsules approved in the United States and European Union for the treatment of progressive, metastatic medullary thyroid cancer, or MTC, and it is marketed for that indication in a distinct capsule formulation under the brand name COMETRIQ.
cancer. The year 2015 was an impactful one for our company, which translated into rapid achievement of our most important commercial, research and development, andtwo other business goals. In July 2015, we obtained positive clinical resultsproducts resulting from our phase 3 pivotal trial METEOR (Metastatic RCC Phase 3 Study Evaluating Cabozantinib vs. Everolimus). Following the positive results from METEOR, the U.S. Food and Drug Administration, or FDA, granted Breakthrough Therapy and Fast Track designations for cabozantinib in RCC. These data from METEOR ultimately formed the basisdiscovery efforts are: COTELLIC® (cobimetinib), an inhibitor of MEK approved as part of a New Drug Application, or NDA, submissioncombination regimen to the FDA, which was completed in December 2015. On January 27, 2016, the FDA granted priority review to the NDA,treat advanced melanoma and marketed under a collaboration with a Prescription Drug User Fee Act, or PDUFA, action date of June 22, 2016. Having been actively preparing for this commercial opportunity, we are presently ready from a staffing perspective to launch cabozantinib for the treatment of patients with advanced RCC in the United States should a positive regulatory decision come prior to the PDUFA action date.
The METEOR results also laid the ground work for our February 2016 exclusive licensing agreement with Ipsen Pharma SAS to commercialize cabozantinib in regions outside the United States, Canada and Japan. Beyond RCC and MTC, we continue to explore the clinical potential of cabozantinib in additional tumor types, including CELESTIAL (Cabozantinib Phase 3 Controlled Study In Hepatocellular Carcinoma), our phase 3 trial of cabozantinib in advanced hepatocellular carcinoma and earlier stage trials conducted through our Cooperative Research and Development Agreement, or CRADA, with the National Cancer Institute’s Cancer Therapy Evaluation Program, or NCI-CTEP, or our investigator sponsored trial program.
In 2015, we also made significant progress with respect to the clinical development, regulatory status and commercial potential of certain of our partnered compounds, notably, cobimetinib, a compound we out-licensed in 2006 to Genentech, Inc. (a member of the Roche Group). The FDA; and MINNEBROTM (esaxerenone), an oral,non-steroidal, selective blocker of the mineralocorticoid receptor approved cobimetinibfor the treatment of hypertension in Japan and licensed to Daiichi Sankyo Limited Company.2018 Financial Performance Highlights
We delivered strong financial results in fiscal 2018, increasing total revenue by 89% year over year and ending fiscal 2018 with a healthy cash and investments balance of $851.6 million. We also continued to execute on November 10, 2015, underour tactical plan to generate product and collaboration revenue to reinvest in our business in order to drive the brand name COTELLICexpansion and depth of our product offerings. Other key financial highlights from fiscal 2018 include:
| ›› | Cabozantinib franchise net product revenue of $619.3 million, resulting in our third consecutive year of record net product revenue; |
| ›› | Total revenue of $853.8 million, reflecting both strong product demand and significant financial contributions generated from our collaborations; |
| ›› | Total operating expenses of $415.0 million, up 45% from the prior fiscal year, demonstrating our commitment to investing in our product pipeline, while also exercising disciplined expense management; and |
| ›› | A second full year of operating profit, resulting in diluted earnings per share of $2.21, taking into account the release of substantially all of our valuation allowance against deferred tax assets, up from diluted earnings per share of $0.49 for the 2017 fiscal year. |
2019 Proxy Statement TM37
,Stock Price Performance
Our strong financial and corporate performance for the three-year period ended December 31, 2018 also resulted in combination with vemurafenib,a stock price that continued to significantly outperform our 2018 peer group (described below) as a treatment for patients with BRAF V600E or V600K mutation-positive advanced melanoma. COTELLIC in combination with vemurafenib has also been approved in Switzerland,whole and the European Union and Canada for use inRussell 3000 Index, as well as the same indication.
2015 Compensation Program Highlights
Our compensation decisions for 2015 reflected the positive clinical results from the METEOR trial and both the speed and success withStandard & Poor’s (S&P) 400 MidCap Index, to which we were
ableadded in July 2018.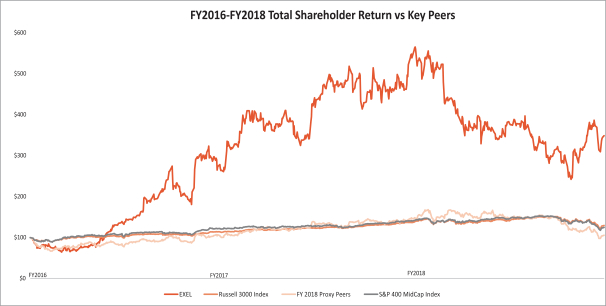
The foregoing graph compares, for the three-year period ended December 31, 2018, the cumulative total stockholder return for our common stock, the Russell 3000 Index, the S&P 400 MidCap Index and an average of the companies that form our 2018 peer group. The graph assumes that $100 was invested on December 31, 2015 in each of our common stock, the Russell 3000 Index, the S&P 400 MidCap Index and in each company in our 2018 peer group, and assumes reinvestment of any dividends. We believe it places our corporate performance in context and highlights the strength of our results relative to navigate the regulatory pathway for cabozantinibour 2018 peer group as a treatmentwhole and the market, despite the decline of our stock price during 2018. The stock price performance on the graph is not necessarily predictive of future stock price performance.
2018 Corporate Performance Highlights
In fiscal 2018, we continued to drive growth by executing on our strategy and substantially advanced toward our goal to become a fully integrated biotechnology company. When considering corporate performance factors, the Compensation Committee’s decision-making in 2018 was rooted in the understanding that our Named Executive Officers led our company to achieve this noteworthy success. Key highlights of our corporate performance included:
Successful Commercialization of CABOMETYX for Advanced RCC
| ›› | In the U.S., following the U.S. Food and Drug Administration’s (FDA) December 2017 approval of CABOMETYX’s expanded indication to include treatment for patients with previously untreated advanced RCC, we continued to promote CABOMETYX within its labeled indication and to educate physicians on its unique clinical profile. These efforts translated into strong product demand in 2018. |
| ›› | In markets outside the U.S., we continued to work closely with our partner Ipsen Pharma SAS (Ipsen) in support of its regulatory strategy and commercialization efforts for CABOMETYX as a treatment for advanced RCC. As a result of approvals in more than ten territories outside of the U.S., including the European Union (EU), Canada, Brazil, Taiwan, South Korea and Australia, CABOMETYX continued to grow both in sales revenue and the number of RCC patients benefiting from its clinical effect in 2018. |
38 Exelixis, Inc.
Compensation of Executive Officers | Compensation Discussion and Analysis
Navigation of Regulatory Pathways Toward Expansion of CABOMETYX Label as a Treatment for Patients with advanced RCC, including receiptHCC Who Have Previously Been Treated with Sorafenib
| ›› | Following acceptance of our supplemental new drug application in May 2018, on January 14, 2019, the FDA approved CABOMETYX as a treatment for patients with HCC who have been previously treated with sorafenib, an aggressive anddifficult-to-treat cancer that represents a significant unmet medical need and a market with the potential to grow significantly in the coming years. |
| ›› | In an effort to help make CABOMETYX available to HCC patients in the EU and worldwide, we worked closely with Ipsen on its regulatory strategy which culminated in the European Commission’s November 2018 approval of CABOMETYX as a treatment for HCC in adults who have previously been treated with sorafenib. This approval provided physicians in the EU with a second approved therapy for the second-line treatment of HCC. |
Late-Stage Evaluation of Breakthrough TherapyCabozantinib as a Monotherapy and Fast Track designationsin Combination with Immune Checkpoint Inhibitors
| ›› | In October 2018, we initiatedCOSMIC-311, a phase 3 pivotal trial evaluating cabozantinib in patients with radioactive iodine-refractory differentiated thyroid cancer (DTC) who have progressed after up to two prior vascular endothelial growth factor receptor (VEGFR)-targeted therapies. Cabozantinib demonstrated encouraging clinical activity in phase 1 and phase 2 studies in this disease setting, suggesting it may be a promising treatment option for patients who have progressed after prior VEGFR-targeted therapy. |
| ›› | In December 2018, we initiatedCOSMIC-312, a phase 3 pivotal trial evaluating cabozantinib in combination with atezolizumab versus sorafenib in previously untreated advanced HCC. Ipsen has opted to participate in this trial and will have access to the results to support potential future regulatory submissions outside of the U.S. and Japan. |
Expansion of Product Pipeline
| ›› | In December 2018, we filed an investigational new drug (IND) application for XL092, a next-generation oral TKI and the first compound to advance from our reinitiated discovery operations. The IND for XL092 was accepted by the FDA in January 2019 and in February 2019, we initiated a phase 1 dose escalation trial to evaluate the compound’s pharmacokinetics, safety and tolerability in patients with advanced solid tumors. |
| ›› | As part of our strategy to expand our product pipeline through thein-license of attractive, early-stage oncology assets, in January 2018, we entered into an exclusive collaboration and license agreement with StemSynergy Therapeutics, Inc. (StemSynergy) for the discovery and development of novel oncology compounds aimed to inhibit tumor growth and in May 2018, we entered into a collaboration and license agreement with Invenra, Inc. (Invenra) which is focused on developing next-generation biologics, to discover and develop multispecific antibodies for the treatment of cancer. |
Successful Management of Organizational Growth and Transition of Corporate Headquarters
| ›› | In support of the execution of our planned cabozantinib development activities and resumption of internal discovery efforts, we increased hiring activities, and as of the end of fiscal 2018, had 484 full-time employees (FTEs), a 30% year-over-year increase of our employee population. To enable the effective management of this growth, we implemented new and improved operational and financial systems, procedures and controls, and expanded employee training efforts. |
| ›› | In contemplation of our organizational growth, in June 2018, we completed the renovation and buildout of our new corporate headquarters in Alameda, California and successfully relocated our employee base in the same month. Our new headquarters are housed in astate-of-the-art facility that meets high building code standards for energy efficiency and environmental impact, and is designed to help maximize collaboration among employees and enable novel drug discovery. |
Enhancements to 2018 Executive Compensation Program
An integral part of the FDA. Further,review process with respect to our executive compensation program was our stockholder outreach initiative, aimed at creating a better understanding of the concerns and perspectives of our stockholder base.
2019 Proxy Statement 39
Stockholder Outreach
Each year, during the period following the filing of our Proxy Statement with the SEC and the date of our annual meeting, we engage with our stockholders to seek support for our annual meeting proposals and request feedback regarding our executive compensation program and other governance matters of importance to our stockholders. Stockholder feedback is then reported to the Compensation Committee, Nominating & Corporate Governance Committee and/or the entire Board for consideration. During this period in 2018, we reached out to stockholders representing more than 57% of our outstanding shares at that time. Participants at these meetings included members of the management team and the chair of our Compensation Committee. This outreach effort provided the opportunity to discuss executive compensation measures that are in the best interest of our stockholders and to convey our commitment to align pay and performance.
Stockholder Advisory Vote on Executive Compensation
Our stockholders are provided the opportunity to cast an annual advisory vote on our executive compensation program, and the Compensation Committee takes the results of this vote into account when determining the compensation of the company’s Named Executive Officers. At our annual meeting of stockholders held in May 2018, approximately 79% of the votes present and entitled to vote voted in favor of thesay-on-pay proposal, which was a decline from 97% in 2017. We gave thoughtful consideration to this development, and to better understand our stockholders’ perspective concerning our executive compensation, we greatly expanded our regular engagement activities. We were particularly focused on those stockholders who voted against oursay-on-pay proposal and reached out to all such stockholders holding more than 0.35% of outstanding shares. Members of the management team participated in these meetings, and the stockholder feedback received was shared and discussed with the Compensation Committee. As a result of this feedback, the Compensation Committee approved several changes to our executive compensation program in an effort to enhance the performance-based nature of the program, create better alignment with the interests of our stockholders, establish greater accountability and increase overall transparency.
In consideration of the constructive feedback from our stockholders and following thoughtful deliberation, the Compensation Committee and Board considered that we performed well in most of the other areas of corporate performance that they determined to be important, including, commercialization, research and development and attainment of our business goals for 2015. As part of the decision making process, the Compensation Committee also considered the challenges of operating with a reduced workforce in the wake of our restructuring in late 2014, the rapid evolution of the business following positive clinical results from METEOR in July 2015 and the need to retain the talent required to ensure the success of a commercial launch of cabozantinib in advanced RCC.
In light of the above, the Board and Compensation Committee took the following key actions with respect to
2015 compensation for our Named Executive Officers:
| | |
Stockholder Feedback Following 2018 Annual Meeting | | How We Responded |
Incorporate Performance Component(s) into Long-Term Incentive Program | | In September 2018, as part of our ongoing equity incentive compensation program, the Compensation Committee granted a mix of stock options, and performance-based RSU awards (PSUs), to focus our Named Executive Officers on the company’s long-term corporate and financial performance. The stock options granted to our Named Executive Officers are subject to service-based vesting. Dr. Morrissey’s stock option is also subject to a market condition and will not be exercisable until the closing market price of our common stock is equal to or greater than 125% of the exercise price of the stock option over a period of at least 30 days. The PSUs granted to Named Executive Officers only vest upon the achievement of certain product revenue, late-stage clinical development and pipeline expansion performance targets. |
Adopt a Recoupment (or Clawback) Policy | | In February 2019, we adopted a clawback policy, which permits us to recoup variable compensation from senior level employees, including our Named Executive Officers, in the event they commit misconduct that causes material harm to the company (see “—Other Compensation Information—‘Clawback’ Policy” below). |
Enhance Disclosure and Provide More Insight With Respect to Annual and Long-Term Incentive Performance Measures | | As reflected in this CD&A, we believe that we have enhanced our disclosure and provided more insight into the performance measures established by the Compensation Committee for our Annual Cash Bonus Plan and our long-term incentive compensation. |
40 Exelixis, Inc.
Salaries for NamedCompensation of Executive Officers. | Compensation Discussion and Analysis
In February 2015, in part to encourage retentioncontemplation of our keycontinued growth and evolving complexities of our business following the commercial success of CABOMETYX, we undertook a comprehensive review of our executive officers during a critical timecompensation program, including through the evaluation of each component of our program relative to our 2018 peer group. That review process resulted in the company’s evolution, the Compensation Committee increased base salaries offollowing additional actions with respect to 2018 compensation for our Named Executive Officers for 2015 by between 3%Officers:
| | |
Priorities Following Evaluation of
Executive Compensation Program | | How We Responded |
Develop a More Objective Framework for Bonus Determinations | | In February 2019, subject to the terms of our Annual Cash Bonus Compensation Plan for Executives (Annual Cash Bonus Plan) adopted by the Compensation Committee in February 2018, the Compensation Committee approved the payment of cash bonuses in amounts between 105% and 139% of each Named Executive Officer’s 2018 target cash bonuses resulting from the Compensation Committee’s assessment of the overall achievement of ourpre-determined corporate goals for 2018, and the contribution of each Named Executive Officer toward such achievements. |
Enhance Alignment Between the Financial Interests of Our Named Executive Officers with Those of Our Stockholders | | In February 2018, the Board adopted Stock Ownership Guidelines for our Named Executive Officers to further align their financial interests with those of our stockholders, as well as to promote sound corporate governance. When determining the amount of Dr. Morrissey’s 2018 cash bonus, the Compensation Committee considered our meaningful accomplishments in 2018 and level of achievement of pre-determined corporate goals. However, the Compensation Committee also considered that it is Dr. Morrissey’s responsibility to create stockholder value, and because our stock price declined during 2018, the Compensation Committee used its discretion, as provided for in the Annual Cash Bonus Plan, to reduce Dr. Morrissey’s 2018 cash bonus below the level of the overall corporate performance. |
We intend to continue to engage with our stockholders throughout the year, and 5% over salaries for 2014.
Equity Incentive Compensation. In February 2015, taking considerationwe invite you to reach out with any comments or questions at any time. Please see the Investor section of the decision not to pay cash bonuses for 2014 and conscious of need to focus all employees, including our Named Executive Officers, on the achievement of critical milestones in our development of cabozantinib, the Compensation Committee recommended, and the Board approved, a grant of performance-based stock options with vesting milestones tied to our METEOR trial related to cabozantinibwebsite for the treatment of advanced RCC. The grants were designed to align employee incentives with those of our investorsappropriate contact information.Compensation Practices and only deliver rewards for positive results that drive stockholder value. Then consistent with our prior practice, in September 2015, as part of our ongoing equity incentive compensation program and following positive clinical results from our METEOR trial, the Board granted time-based stock options in order to focus our Named Executive Officers on the company's long-term performance. In February 2016, the Compensation Committee approved a special one-time grant of stock options to certain of our Named Executive Officers in consideration of their exceptional service to our company during 2015 and the desire to retain the executive talent necessary to drive toward the achievement of our company's future commercial and research and development goals.Governance Highlights
| | |
Pay for Performance | | Link the compensation of our Named Executive Officers to the success of our corporate goals and stock price appreciation. |
Stockholder Alignment | | Align the interests of our Named Executive Officers with those of our stockholders through the use of long-term equity incentives. |
Compensation Governance | | Compensation Committee made up entirely of independent directors. Compensation Committee engages an independent compensation consultant to advise on executive compensation matters. Conduct an annual stockholder vote to approve our say-on-pay proposal. |
Recoupment (or Clawback) Policy | | Ability to recoup variable compensation from senior level employees, including our Named Executive Officers, for their misconduct that causes material harm to the company. |
Annual Cash Bonus Amounts Subject to Payment Maximums | | Achieved corporate performance percentage and/or individual performance percentage may not exceed 200%. |
Equity Plan Features | | Apply a maximum7-year term for stock options. No repricing of underwater stock options without prior stockholder approval. 2017 Plan includes minimum vesting requirements of no less than one year for all types of awards, subject to limited exceptions. |
Stock Ownership Guidelines | | Apply stock ownership guidelines to directors and executive officers to further align their interests with those of our stockholders. |
Change in Control Provisions | | No excessive change in control or severance payments. Provide “double-trigger” change in control benefits. No taxgross-ups on severance or change in control benefits. |
Annual Cash Bonus (Equity in Lieu of Cash).2019 Proxy Statement In February 2016, as part of our continued cash conservation efforts and to align the interests of the Named Executive Officers with those of our stockholders, in lieu of cash bonus payments, the Compensation Committee paid our Named Executive Officers their annual bonuses for 2015 in the form of fully-vested restricted stock units, or RSUs, in amounts ranging from 100% to 111% of annual bonus target.41
| | |
Perquisites, Retirement and Pension Benefits | | Named Executive Officers do not receive excessive perquisites or post-termination retirement or pension benefits that are not available to all employees generally. |
Prohibition on Hedging and Margin Loans | | Prohibit hedging and purchases on margin by executive officers and directors. |
Meaningful Limits on Pledging | | Limit pledging of our common stock by executive officers and directors to circumstances where the individual can clearly demonstrate the financial capacity to repay the loan without resorting to the pledged securities. No executive officers or directors pledged our common stock during 2018. |
Compensation Risk Assessment | | Compensation Committee conducts an annual risk assessment of our compensation policies and practices to ensure that our programs are not reasonably likely to have a material adverse effect on us. |
Objectives of the Compensation Program
The primary goals of our executive compensation program are to:
| ›› | Provide market-competitive compensation that attracts, retains and motivates our executive officers to achieve our short- and long-term business objectives; |
| ›› | Align our executive officers’ compensation with the interests of our stockholders; and |
| ›› | Reward our executive officers for success in achieving our corporate goals. |
The success of biopharmaceutical companiesany biotechnology company is significantly influenced by the quality of theirits work forces.force. We believe it is critical to our business that we retain our core team of highly qualified employees, including our Named Executive Officers. As a testament to their high value in the marketplace, for human resources, large pharmaceutical and biopharmaceuticalbiotechnology companies and strong local competitors have aggressively recruited our executives and other skilled employees, with the most critical positions at our company among those that are the most in demand.employees. In light of these circumstances, each year we have designeddesign our executive compensation program to help attract and retain highly qualified individuals with relevant experience in the biopharmaceuticalbiotechnology industry to manage the varied aspects of our evolving business. The primary goals of our executive compensation program are to: provide market-competitive compensation that motivates our executive officers to achieve our short- and long-termgrowing business objectives, align our executive officers’ compensation with the interests of our stockholders and reward our executive officers for their exceptional performance and the success of our business.
To this end, our 2015 compensation program for Named Executive Officers aimed to strike a balance among, the following three principal components, which are discussed in further detail under “Elements of Compensation” below:
Base Salary. We pay a base salary to each ofoperations. In fact, our Named Executive Officers to providehave an appropriate and competitive base levelaverage tenure of current cash income.
Annual Cash Bonus. We establish target annual cash bonuses for our Named Executive Officers based on the seniorityover eleven years with Exelixis, which we believe is evidence of the applicable position, as an incentive to encourage superior performance. Actual cash bonuses are discretionary, but the amounts generally are determined following pre-established guidelines that take into account achievement against pre-established company, department and, in some cases, personal objectives.
Equity Incentive Compensation. We grant equity incentive awards designed to ensure that incentive compensation is linked to our long-term performance. These awards align our executives’ performance objectives with the interestseffectiveness of our stockholders, and provide strong incentives for our key executivescompensation program with respect to remain with our company.
In addition, we have a Change in Control and Severance Benefit Plan in which all of our Namedretention.How We Determine Executive Officers participate, as well as an employee stock purchase plan, a 401(k) Retirement Plan, and healthcare, dental and vision plans in which all of our employees are entitled to participate, including our Named Executive Officers.
The Compensation Committee has not established any formal policies or guidelines for allocating compensation between current and long-term incentive compensation, or between cash and non-cash compensation. The Compensation Committee uses its judgment in determining an appropriate mix of current and long-term incentive compensation, and cash and non-cash compensation for our Named Executive Officers, that it believes appropriate to achieve the compensation and corporate objectives described above.
Role of the Compensation Committee, Compensation Consultants and Executive Officers in Compensation Decisions
Role of the Compensation Committee
The Compensation Committee is responsible for determiningevaluating and approving the compensation packages offered to our Named Executive Officers. When appropriate, the Compensation Committee will solicit the input of, or present its recommendations on compensation matters for consideration and approval to, the full Board. For example, the Compensation Committee recommended to the Board for approval the 2015 salary and annual bonus determinations, and determined that it was appropriate to discuss with, and obtain input from, the Board on the decision to grant fully-vested RSUs in lieu of cash compensation for the 2015 annual bonus, as well as the related share amounts. The Compensation Committee acts on behalf of the Board in discharging the Board’s responsibilities with respect to overseeing our compensation policies, plans, and programs, and establishing and reviewing general policies relating to compensation and benefits of our employees. The Compensation Committee also administers our equity and other incentive plans. The Compensation Committee does not delegate any of its functions to others in determining executive compensation.
Role of Compensation Consultants
Under its charter, the Compensation Committee has the authority to obtain the advice and assistance from external advisors to assist it in the performance of its duties. In accordance with this authority, the Compensation Committee engages external advisors to advise on executive officer compensation, including base salaries, bonus targets and equity compensation, as well as director compensation. These advisors also prepare industry compensation data, assist with the development of our peer group, and advise the Compensation Committee on best industry practices and relevant changes to regulations that may impact the Compensation Committee’s decision-making process with regard to executive officer and director compensation determinations. In particular, in November 2017, the Compensation Committee retained the consulting firm Compensia, to compile biotechnology industry compensation of our executive officers and directors against that peer group. The Compensation Committee also received documentary support, including peer group and industry data from Compensia with
42 Exelixis, Inc.
Compensation of Executive Officers | Compensation Discussion and Analysis
respect to base salaries, target annual cash bonuses, and equity compensation. In January 2018, the Compensation Committee retained Compensia to provide a market analysis of executive compensation of our executive officers compared to our peer group. This market analysis was reviewed with the Compensation Committee and used to guide 2018 base salary and bonus target decisions for our Named Executive Officers. In August 2018, at the request of the Compensation Committee, Compensia also prepared a market analysis of long-term incentive compensation of our executive officers compared to our peer group. This market analysis was reviewed with the Compensation Committee and used to guide annual long-term equity compensation determinations in September 2018.
The Compensation Committee assessed the independence of Compensia pursuant to SEC rules and concluded that the work performed by Compensia for the Compensation Committee had not raised any conflict of interest. The Compensation Committee did not engage any other consultants in 2018 with respect to executive and/or director compensation matters, other than Compensia.
Role of Executive Officers
Dr. Morrissey, our President and Chief Executive Officer, also participates in the Compensation Committee’s deliberations with respect to Named Executive Officer compensation other than his own, and is not present during deliberation and voting on his compensation. Each year, Dr. Morrissey and other senior management also develop annual corporate business goals for the company, which are reviewed and, subject to their input, approved by the Compensation Committee and the Board. In determining Named Executive Officer compensation recommendations for 2015,2018, our Chief Executive Officer solicited the input of and received documentary support from our senior human resources personnel. The Compensation Committee also received documentary support, including benchmark and industry data, from third-party salary survey sources, including the Radford Global Life Sciences Survey, with respect to base salaries, target annual cash bonuses, and equity compensation. In December 2015, the Compensation Committee also retained the consulting firm Compensia, Inc., or Compensia, to compile benchmark biopharmaceutical industry compensation data and help us to evaluate of the compensation of our executive officers in comparison with that benchmark. This information was discussed with the Compensation Committee in January 2016 for use in making 2016 executive compensation decisions. The Compensation Committee does not delegate any of its functions to others in determining executive compensation, and we do not currently engage any other consultants with respect to executive and/or director compensation matters, other than Compensia, and Radford, retained to provide compiled industry compensation data for our senior management and directors.
Compensation Committee Process
In setting the level of salary, annual cash bonus and equitylong-term incentive compensation for our Named Executive Officers, the Compensation Committee typically considers various factors, including the performance of each Named Executive Officer and of the company itself during the prior year; the criticality of each such officer’s skill set; the officer’s performance and expected future contributions; market data for our industry and specific peer group; the officer’s tenure; the percentage of vested versus unvested equity awards held by the officer; and the value of the equity awards he or she holds. including:
| ›› | the performance of the company during the prior year; |
| ›› | the performance of each Named Executive Officer during the prior year; |
| ›› | the criticality of each Named Executive Officer’s skill set and expected future contributions to our business; |
| ›› | the growing complexity of our business resulting in increased workloads and responsibilities of our Named Executive Officers; |
| ›› | the appropriate mix of compensation for each Named Executive Officer; |
| ›› | the historical salary, cash bonus and percentage of vested versus unvested equity awards held by each Named Executive Officer; and |
| ›› | market data, which include competitive information relating to compensation levels for comparable positions in the biotechnology and life sciences sector and for our specific peer group. |
The Compensation Committee balances each of these factors against the company’sour cash resources and equity award burn rate guidelines established by proxy advisory firms. When establishing each element of a Named Executive Officer’s compensation, the Compensation Committee also typically takes into consideration the officer’s historical salary, cash bonuscritical need to prioritize clinical development and equity compensation,pipeline expansion investments over other expenditures, as well as his or her total current and potential compensation.our aggregate equity burn rate. Using this process, our Compensation Committee strives to ensure that our executive
compensation program as a whole is competitive. ConsistentCompetitive Assessment
A key objective of our executive compensation program is to ensure that the overall compensation package we provide to our executive officers is competitive relative to the companies with the Compensation Committee’s philosophy of maintaining compensation levels that attract and retain the highest caliber executives, thewhich we compete for executive talent. The Compensation Committee generally targets total cash (including salaryconsults with its independent compensation consultant, Compensia, to develop a peer group of companies to serve as the basis for comparing our executive compensation program to the market.
Peer Group Development Process and cash bonus)How We Used the Data
The Compensation Committee reviews and equity compensation abovemakes adjustments to the 50
th percentilecomposition of the peer company marketgroup as often as deemed necessary, to account for changes in both our business and the positions for which data is available. In determiningbusinesses of the amount and mix of compensation elements and whether each element providescompanies in the correct incentives and rewards for performance consistent with our short and long-term goals and objectives, the Compensation Committee relies on its judgment about each individual rather than adopting a formulaic approach to compensatory decisions. peer group. The Compensation Committee believes that it is important when making itsdoes not have a specific target compensation decisions to be informed as tolevel for the currentNamed Executive Officers. Instead, we review the practices of comparable publicly-held companies. To this end,our peer group and the Compensation Committee reviewsgeneral biotechnology market data, which include competitive information relating to compensation levels for comparable positions in the biotechnology and life sciences sector.
Peer Companies. In February 2015, the Compensation Committee, in consultation with management, developed a peer companies list to be used as a reference point to assist us in assistingdeveloping programs designed to attract and retain exceptional talent and drive company performance.2019 Proxy Statement 43
In developing the 2018 peer group, the Compensation Committee in setting base salaries, bonus targets and annual equity incentive compensation for 2015. The list was developed by modifying the peer companies list originally developed in 2013 in consultation with Frederick W. Cook & Co., Inc. The Compensation Committee used the same peer companies list to assist it with making its executive officer compensation decisions for 2014. Given the company's financial position and cash conservation efforts following the restructuring in late 2014, the Compensation Committee determined that utilizing the resources from prior years was the most appropriate and economical approach for the development of a peer companies list for 2015, and therefore was guided by the prior year's peer companies list to make its compensation decisions for 2015.
In developing the 2015 peer companies list, the Compensation Committee first considered the significant
changes toevolution of our business
and meaningful developments
atin comparison to the
2017 peer group companies. It also considered what other companies
included on the prior list, as well as companies that might
constitutemake suitable additions to the
2018 list. The key qualitative and quantitative considerations that influenced the development of the
2018 peer
companies listgroup were:
(1) comparability of science and therapeutic focus; (2) company size, as reflected generally by headcount and market capitalization; (3) company complexity, as reflected by the number of marketed or investigational products and their respective phase of development; and (4) recent company performance or the proximity of near-term material events. | ›› | industry—including, biotechnology and pharmaceutical companies; |
| ›› | therapeutic area, including companies with an oncology product focus, while avoiding the inclusion of companies marketing solely orphan and rare disease drugs; |
| ›› | stage of business, as reflected by the existence of a marketed product; |
| ›› | location of headquarters and whether the company is traded on a major U.S. exchange; |
| ›› | market capitalization between 0.25x—4.0x of our then-current market capitalization (approximately $1.9 billion—approximately $30.0 billion); and |
| ›› | revenue between 0.3x—5.0x of our then revenue (approximately $96 million—approximately $1.6 billion). Due to the limited number of companies that had comparable revenue growth expectations after taking into consideration the possibility that our revenue could grow appreciably due to increased CABOMETYX sales during 2018, the Compensation Committee expanded the top end of the revenue range from 2.0x of current revenue to 5.0x. |
Following this analysis, the Compensation Committee, determined that eleven ofin consultation with management and Compensia identified the following 19 publicly-traded, U.S—based biotechnology/pharmaceutical companies that were on the list originally developed in in 2013 were no longer appropriateas our peer companies and removed them from the list, and added nine new companies that it determined appropriate based on the criteria set forth above.
The peer companiesgroup to be used in reviewing compensation for 2015 comprised the following 16 publicly-traded, U.S- based biotechnology/pharmaceutical companies:
2018: |
| | | |
AegerionFiscal 2018 Peer Group |
Alkermes Public Limited Company | | Ionis Pharmaceuticals, Inc. | Incyte Corporation | Pharmacyclics LLC |
Array BioPharma, Inc. | Infinity Pharmaceuticals, Inc. | Rigel Pharmaceuticals,OPKO Health, Inc. |
Arena Pharmaceuticals,BioMarin Pharmaceutical Inc. | | Ironwood Pharmaceuticals, Inc. | Sunesis Pharmaceuticals, inc. | Seattle Genetics, Inc. |
AriadBioverativ Inc. | | Jazz Pharmaceuticals Public Limited Company | | Supernus Pharmaceuticals, Inc. | Lexicon Pharmaceuticals, Inc. | Synta Pharmaceuticals Corp. |
CelldexClovis Oncology, Inc. | | Juno Therapeutics, Inc. | Merrimack Pharmaceuticals, | TESARO, Inc. |
Corcept Therapeutics Incorporated | | Ligand Pharmaceuticals Incorporated | | The Medicines Company |
Clovis Oncology,Halozyme Therapeutics, Inc. | | Nektar Therapeutics | | United Therapeutics Corporation |
Incyte Corporation | | | | |
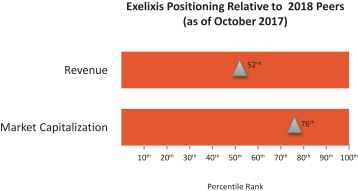
Of these companies, ten companies (Alkermes Public Limited Company, Corcept Therapeutics Incorporated, Halozyme Therapeutics, Inc., Ionis Pharmaceuticals, Inc., Ironwood Pharmaceuticals, Inc., Nektar Therapeutics, Seattle Genetics, Inc., Supernus Pharmaceuticals, Inc. TESARO, Inc. and United Therapeutics Corporation) were in our 2017 peer group. The Compensation Committee did not include in our 2018 peer group nine companies (ACADIA Pharmaceuticals Inc., Intercept
44 Exelixis, Inc.
Stockholder Advisory Vote onCompensation of Executive Compensation.Officers We provide| Compensation Discussion and Analysis
Pharmaceuticals, Inc. and Neurocrine Biosciences, Inc., Acorda Therapeutics, Inc., Eagle Pharmaceuticals, Inc., Lexicon Pharmaceuticals, Inc., Momenta Pharmaceuticals, Inc. and Vanda Pharmaceuticals, Inc. and ARIAD Pharmaceuticals, Inc.) that were in our stockholders with2017 peer group, because they were either acquired or had a market capitalization or revenue that fell below the opportunityselection criteria.
Reference Peer Group
In addition to cast an annual advisory vote onthe analysis of the compensation data from the peer group, due to our executive compensation program, which vote,anticipated growth during 2018, the Compensation Committee takes into account when determiningalso reviewed the compensation levels and disclosed program design for executive officers of Biogen Inc., Regeneron Pharmaceuticals, Inc., Shire Plc, Vertex Pharmaceuticals Incorporated and Alexion Pharmaceuticals, Inc. to provide perspective on what executive officer pay would look like at the next stage of growth. We refer to this group of companies as our 2018 reference peer group. The Compensation Committee did not include in our 2018 reference peer group six companies (Endo International plc, United Therapeutics Corporation, Jazz Pharmaceuticals Public Limited Company, Medivation, Inc., BioMarin Pharmaceutical Inc., Incyte Corporation) that were in our 2017 reference peer group, because they were either acquired, already included in our 2018 peer group based on application of the company’sselection criteria or had a market capitalization that fell below the selection criteria for our 2018 reference peer group. The Compensation Committee did not use the 2018 reference peer group to inform any compensation decisions for Named Executive Officers. At our annual meeting of stockholders heldOfficers in May 2014 and May 2015, approximately 90% and 92%, respectively, of the votes cast on the say-on-pay proposal voted in favor of the proposal. Our 2018.
Compensation Committee considered these votes to be an endorsement of the Compensation Committee’s policies and practices and has continued to conduct its review of executive compensation generally consistent with past practice.Elements
Elements of Compensation
As summarized above, ourOur executive compensation program consists of three principal components: base salary,salary; annual discretionary cash bonuses,bonus; and equitylong-term incentive compensation. OurA description and objective(s) for each element of compensation is set forth below:
| | | | | | |
Element | | Description | | Objective(s) |
Annual Base Salary | | Fixed cash compensation to recognize each Named Executive Officer’sday-to-day responsibilities. | | Provide an appropriate and competitive base level of current cash income for Named Executive Officers. |
Annual Cash Bonus | | Variable cash compensation based on performance againstpre-determined corporate goals. | | Align our Named Executive Officer’s compensation with our annual corporate goals. Enable us to reward Named Executive Officers who fulfill or exceed performance expectations. |
Long-Term Incentive Compensation | | RSUs (or PSUs, if performance-based) | | Variable share-based compensation, subject to either time-based vesting or performance-based vesting based on the achievement of key commercial, product development and pipeline expansion goals. | | Align the interests of our Named Executive Officers’ with those of our stockholders. Promote retention, including during periods of stock price volatility common to biotechnology companies. Motivate our Named Executive Officers to achieve long-term corporate performance objectives. |
| Stock Options | | Variable share-based compensation whereby value is derived from appreciation in stock price. | | Align the interests of our Named Executive Officers with those of our stockholders. Motivate our Named Executive Officers to aggressively pursue our critical business objectives, because stock options only have value if the value of our company as reflected by our stock price increases over time. |
2019 Proxy Statement 45
Other Compensation
In addition to the primary elements of compensation described above, our Named Executive Officers are also eligible to participate, on the same basis as other employees, in our 401(k) Retirement Plan, our employee stock purchase plan (ESPP) and other benefit programs generally available to all employees. These programs are intended to providetax-beneficial ways to save toward retirement, promote health and wellness and encourage stock ownership. Our Named Executive Officers currently do not receive any perquisites.
Base Salary. The Compensation Committee reviews annually each Named Executive Officer’s base salary and sets salary generally based on the criticality of such officer’s skill set, past performance and expected future contributions, the market data for our industry and specific peer group, tenure, and market pressures, with each of these factors balanced against consideration of our cash resources and other elements of the officer’s compensation.
Annual Cash Bonus. Our discretionary annual cash bonuses are intended to align the Named Executive Officers’ compensation with our business objectives and performance, as well as the interests of our stockholders, and to enable us to retain and reward executive officers who fulfill or exceed performance expectations. Our Compensation Committee recommends bonus targets (expressed as a percentage of base salary) based on the seniority of the applicable position and takes into account the achievement of company-wide and, other than for our Chief Executive Officer, applicable department or personal performance objectives in determining actual cash bonuses. The bonus targets are reviewed annually by the Compensation Committee. The Compensation Committee does not follow rigid percentage allocations or other guidelines related to the portion of each Named Executive Officer’s annual cash bonus tied to company-wide, department or personal performance components, but instead exercises its discretion based on its assessment of each officer’s individual performance and department and company factors. Whether or not a bonus is paid for any year is solely within the discretion of our Compensation Committee and Board. Additionally, from time to time, the Compensation Committee may decide to grant equity in lieu of cash compensation if deemed appropriate based upon the business requirements and financial position of the company. The actual bonus awarded in any year, if any, may be more or less than the target, depending on individual performance and the achievement of our company-wide objectives, as well as other factors, including our cash resources.
Equity Incentive Compensation. We have granted, and intend to continue to grant, stock options and RSUs, and potentially other types of equity incentive awards, to align our Named Executive Officers’ compensation with achievement of the company’s most important business objectives and its long-term performance, thereby linking their compensation to the interests of our stockholders. The Compensation Committee believes that stock options are an effective equity-based tool to motivate our Named Executive Officers to pursue these critical company interests aggressively because options only have value if the value of our company as reflected by our stock price increases over time. Stock options are also the most common long-term incentive instrumenteligible to participate in use at our peer companies. Another commonly utilized long-term incentive compensation instrument, however, is RSUs. The Compensation Committee believes that RSUs are another effective instrument for employee retention, and that they also have incentive value since the value of RSUs increase as our stock price increases over time; RSUs are also generally less dilutive to our stockholders than stock options. Further, RSUs continue to have incentive value even in the event of a severe stock price decline, unlike stock options that can lose their incentive value in such an event. Because of the overall importance to our success of aggressively pursuing our strategic goals, as well as preserving our cash resources, a significant portion of the Named Executive Officers’ total compensation typically has consisted of, and is expected to continue to consist of, equity-based awards.
Change in Control and Severance Benefit Plan. We have adopted a Change in Control and Severance Benefit Plan, in which all ofa compensation program that incentivizes our Named Executive Officers participate, to maintain the competitiveness of our executive compensation program and to remove or at least reduce an executive’s potential personal bias against a takeover attempt. It also provides incentives to our executive officers to remain with our company, and objectively evaluate and facilitate an acquisition of our company should consideration of such a transaction be determined appropriate by the Board and in the best interests of our stockholders. A description of this plan is set forth below under the heading “Potential Payments Upon Termination or Change-in-Control.” This plan is a double-trigger plan, in which each participant receives benefits under the plan only if the participant is terminated without cause or is constructively terminated in connection with a change in control, rather than a single-trigger plan, in which each participant would receive benefits under the plan if a change in control occurs or the participant resigns for any reason after a change in control. We adopted a double-trigger plan because it protects the participants from post-change in control events that are not related to the participants’ performance, encourages them to stay throughout a transition period in the event of a change in control, and does not provide for benefits for a participant who remains with the surviving company in a comparable position. The Compensation Committee believes that, together, these benefits are also an important elementdriver of the Named Executive Officers’ retention and motivation and are consistent with compensation arrangements provided in a competitive market for executive talent,talent. | | |
Element | | Description |
401(k) Plan | | Named Executive Officers contribute their own funds, as salary deductions, on apre-tax orafter-tax basis, subject to plan and government limits. We matchpre-tax and Roth 401(k) contributionsdollar-for-dollar up to $8,500 in each calendar year, in the form of Exelixis common stock. |
Employee Stock Purchase Plan | | Our ESPP allows for Named Executive Officers to purchase shares of our common stock at a price equal to the lower of 85% of the closing price on the first day of thesix-month offering period or 85% of the closing price on the final day of such offering period, subject to specified limits. |
Health Care, Dental and Vision Benefits | | Subject to applicable laws, these health and welfare benefits are available to all eligible employees, including our Named Executive Officers. |
Change in Control and Severance Benefit Plan (1) | | Double-trigger plan, in which each participant receives plan benefits only if the participant is terminated without cause or is constructively terminated in connection with a change in control. We adopted a double-trigger plan because it protects the participants from post-change in control events that are not related to the participants’ performance, encourages them to stay throughout a transition period in the event of a change in control, and does not provide for benefits for a participant who remains with the surviving company in a comparable position. To ensure our best interests, the plan requires a release of claims against us as a condition to receiving any severance benefits. |
| (1) | A description of the Change in Control and Severance Benefit Plan is set forth below under the heading “Potential Payments Upon Termination or Change in Control.” |
46 Exelixis, Inc.
Compensation of Executive Officers | Compensation Discussion and Analysis
Compensation Mix
The Compensation Committee has not established any formal policies or guidelines for allocating compensation between annual and long-term incentive compensation, or between cash andnon-cash compensation. Instead, through our compensation program, the Compensation Committee seeks to align pay and performance. To that end, a significant portion of our Named Executive Officers’ compensation is “at risk” because it is variable, performance-based and in large part dependent on the success of our company.At-risk compensation for 2018 included stock option awards, the value of which depends on increases in the price of our common stock, PSUs and annual incentive cash bonuses, which relate to the overall level of achievement of our company performance goals and, other than for Dr. Morrissey, each Named Executive Officer’s individual contributions toward the achievement of such performance goals. The following charts highlight the 2018 pay mix for our Chief Executive Officer and all of our other Named Executive Officers as a group.
| | |
Chief Executive Officer Pay Mix1 88% of CEO 2018 Compensation is ConsideredAt-Risk | | All Other Named Executive Officers (as a group) Pay Mix1 82% of All Other Named Executive Officers (as a group) 2018 Compensation is ConsideredAt-Risk |
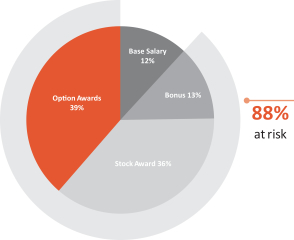 | | 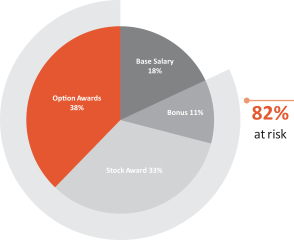 |
| 1 | Percentages calculated from values reported in the 2018 Summary Compensation Table |
2018 Compensation Decisions
In 2018, we achieved significant corporate and financial milestones that helped fuel the growth of our organization. The Compensation Committee believes that the benefits of such severance rights agreements, including generally requiring a release of claims against us as a condition to receiving any severance benefits are in our best interests.
Other Benefits. We have a 401(k) Retirement Plan in which substantially all2018 compensation of our employees, including our Named Executive Officers, are entitled to participate. Employees contribute their own funds, as salary deductions, on a pre-tax basis. Contributions may be made up to plan limits, subject to government limitations. We match 100% ofreflects not only those achievements, but also encourages appropriate efforts towards the first 3% of employee contributions, in the form of Exelixis stock. Our employee stock purchase plan allows
for eligible employees to purchase sharesachievement of our
common stock at a price equal tofuture commercial, product development and pipeline expansion goals.2018 Base Salaries
In considering the lowerappropriate level of 85% of the closing price at the beginning of the offering period or 85% of the closing price at the end of each six month purchase period, subject to specified limits. We provide health care, dental and vision benefits to all full-time employees, includingbase salaries for our Named Executive Officers. These and other standard heath and welfare benefits are available to all eligible employees, subject to applicable laws. We provide these benefits because we believe they are consistent and competitive with the market practices of our peers.
2015 Compensation Decisions
2015 Base Salaries
In February 2015, the Compensation Committee reviewed the base salaries of our Named Executive Officers, and determined that, in light of their performance in 2014 and their importance to the future of the company, the Compensation Committee would recommend to the Board that the base salary for each of the Named Executive Officers be increased by the following percentages over 2014, with base salary increases approved by the Board as follows, except for Mr. Senner's due to the fact that he joined the company in July 2015. In reaching these decisions, the Compensation Committee did not apply a formula, but rather employed a holistic analysis emphasizing the factors described above.
|
| | | | | | |
| Name | | 2014 Base Salary | | 2015 Base Salary | | Percentage Increase |
| Michael M. Morrissey, Ph.D. | | $770,000 | | $800,000 | | 4% |
| Christopher J. Senner | | -- | | $500,000 | | -- |
| Deborah Burke | | $315,000(1) | | $327,600 | | 4% |
| Jeffrey J. Hessekiel, J.D. | | $450,000 | | $465,750 | | 3% |
| Peter Lamb, Ph.D. | | $411,184 | | $423,519 | | 3% |
| Gisela M. Schwab, M.D. | | $523,982 | | $550,181 | | 5% |
emphasizing: | |
(1)›› | Ms. Burke’s annual salary from January 1 through February 28, 2014, was $273,114 when she was serving as Vice President, Finance and Controller. Her salary increased to $284,039 effective March 1, 2014 as partthe individual contribution of the company’s annual reviewNamed Executive Officer to our exceptional 2017 commercial, regulatory and financial achievements; |
| ›› | the criticality of compensation,each Named Executive Officer’s skill set and relative expected future contributions to $315,000 effective September 21, 2014, when she was appointed Senior Vice President and Chief Financial Officer after serving as Vice President and interim Chief Financial Officer following the departure of Frank Karbe, our former Executive Vice President and Chief Financial Officer, on June 2, 2014.business; |
| ›› | the growing complexity of our business resulting in increased workloads and responsibilities of each of our Named Executive Officers; and |
| ›› | other competing business priorities for our company that require substantial investment. |
2019 Proxy Statement 47
2015 Annual Cash Bonuses (EquityThe Compensation Committee also considered the base salaries of similarly situated executives at our 2018 peer group companies for an understanding of whether our compensation program is competitively positioned to retain our highly qualified Named Executive Officers. Following such analysis, in Lieu of Cash)
In February
2015,2018, the Compensation Committee
recommended,determined that each Named Executive Officer should receive an increase in his or her base salary for 2018 as follows: | | | | | | | | | | | | |
Name | | 2017 Base
Salary | | | 2018 Base
Salary | | | Percentage
Increase | |
Michael M. Morrissey, Ph.D. (1) | | $ | 901,000 | | | $ | 955,060 | | | | 6.00 | % |
Gisela M. Schwab, M.D. (2) | | $ | 636,000 | | | $ | 680,520 | | | | 7.00 | % |
Christopher J. Senner (3) | | $ | 567,000 | | | $ | 601,020 | | | | 6.00 | % |
Jeffrey J. Hessekiel, J.D. (4) | | $ | 511,045 | | | $ | 536,597 | | | | 5.00 | % |
Peter Lamb, Ph.D. (5) | | $ | 476,100 | | | $ | 497,525 | | | | 4.50 | % |
| (1) | Dr. Morrissey’s base salary increase was the result of the strength of our overall corporate performance during 2017, including our clinical and regulatory achievements and commercial success of CABOMETYX. Dr. Morrissey was also viewed as critical to our advancement toward our goal to become a fully integrated biotechnology company focused on the discovery, development and commercialization of oncology products. |
| (2) | Dr. Schwab’s base salary increase was the result of her strong leadership of our product development and medical affairs organizations and her performance with respect to the execution of our product development goals in 2017, particularly with respect to the FDA’s approval of CABOMETYX as a treatment for patients with previously untreated advanced RCC, positivetop-line results from CELESTIAL, support of cabozantinib worldwide regulatory filings by collaboration partners, and the initiation of multiple clinical trials of cabozantinib in combination with immunotherapy agents. Dr. Schwab’s leadership was also viewed as critical to our ability to execute on our 2018 product development goals, including with respect to regulatory filings, late-stage clinical trial initiation, the continued broad evaluation of cabozantinib and anticipated organizational growth. |
| (3) | Mr. Senner’s base salary increase was the result of his leadership of our finance organization and his performance with respect to the execution of our financial goals in 2017, particularly with respect to the repayment of all of our outstanding debt. Mr. Senner’s leadership was also viewed as critical to our ability to execute on our 2018 fiscal management objectives. |
| (4) | Mr. Hessekiel’s base salary increase was the result of his leadership of our legal and compliance organizations and his performance with respect to our business and legal goals in 2017, particularly with respect to the resolution of our arbitration with Genentech. Mr. Hessekiel’s leadership was also viewed as critical to our ability to ensure the continued strength of our compliance program during a period of anticipated growth. |
| (5) | Dr. Lamb’s base salary increase was the result of his leadership of our research and discovery organization and his performance with respect to our pipeline development goals, particularly with respect to the progress on the license and collaboration agreement with StemSynergy and the rebuilding of our discovery organization. Dr. Lamb’s leadership was also viewed as critical to our efforts to expand our product pipeline beyond cabozantinib. |
2018 Annual Cash Bonuses
In February 2018, the Compensation Committee approved the Annual Cash Bonus Plan, an annual incentive program designed to provide our senior management team, including our Named Executive Officers, with incentives and rewards for working to achieve the Board approved,strongest possible performance againstpre-determined corporate goals, while also enhancing our ability to attract and retain highly talented individuals. Under our Annual Cash Bonus Plan, Named Executive Officers are eligible to receive an annual performance-based cash bonus award, the amount of which is based on apre-set target percentage of the Named Executive Officer’s annual base salary earned during the year. The Compensation Committee is responsible for establishing the bonus target percentages, as well as the relative percentage contributions of corporate performance and individual performance. For each Participant, the amount of the cash bonus award for each fiscal year depends upon our achievement of applicable corporate performance goals established by the Compensation Committee for that 2015year, and, as applicable, an assessment of each Named Executive Officer’s individual performance. The corporate performance goals under the Annual Cash Bonus Plan may be based on criteria such as the following: sales or commercial goals; research, development and clinical activities; financial metrics, including revenue, cash flow and net income, cash balance, operating expenses and stock price performance; hiring, retention, development of plans and other operational goals; commercial, clinical and strategic collaborations and alliance management; acquisitions and licensing or partnering
48 Exelixis, Inc.
Compensation of Executive Officers | Compensation Discussion and Analysis
transactions; manufacturing and supply goals; quality goals; regulatory goals; and government affairs and public policy goals. Individual performance may be assessed by the Compensation Committee based on the individual participant’s contributions toward the achievement of our corporate performance goals, department goals for the participant’s area of responsibility, or other individual goals derived from or related to our corporate performance goals. For any year, the achieved corporate performance percentage and/or individual performance percentage may exceed 100% in the event the company or the Named Executive Officer exceeds the targeted level of achievement of the applicable goals, provided that neither percentage may exceed 200%.
Bonus Targets
Bonus targets (expressed as a percentage of base salary) are based on the seniority of the applicable position. They are reviewed annually by the Compensation Committee, taking into consideration competitive market data. In February 2018, the Compensation Committee determined 2018 target discretionary annual cash bonus as percentages of base salarybonuses should remain at the same levels as 20142017 for all Named Executive Officers other than Dr. Morrissey (i.e., 60%50% for Dr. Morrissey,Schwab and 45% for each of the other Named Executive Officers, other than Ms. Burke whose 2015 target discretionary bonus remained at 35%)Messrs. Senner and Hessekiel and Dr. Lamb). The Compensation Committee’s decision regarding 20152018 target bonuses was based on its subjective assessment that the percentages of base salaries previously established were appropriate and continuecontinued to align us competitively with our 2018 peer group. TheA review of a market analysis prepared by Compensia indicated that with a bonus target bonus amounts forof 75%, Dr. Morrissey’s target total cash compensation fell below the 50th percentile of market peers. In order to better align his total cash compensation with those of chief executive officers of the companies included in our 2018 peer group and in recognition of his greater role in determining the course of, and ability to influence the future of the company, as well as the critical importance of his leadership to the company’s discretionary cash bonuses are intendedachievement of its 2018 business and financial objectives, the Compensation Committee approved an increase to serve as general guidelines for awarding actual bonusesDr. Morrissey’s 2018 target annual bonus to 100%.
Corporate Goal Development and are not designed to set formulaic payout levels.
WeightingIn connection with establishing the bonus program for 2015,2018, the Compensation Committee reviewed the company’smanagement’s proposed commercial, researchproduct development, pipeline development, finance, legal and development and general businessfacilities goals as presented by management and recommended them to the Board for approval. In February 2015,2018, the Board then reviewed and approved the proposed company goals for 2018, as identified in the table below. In selecting these goals, withthe Compensation Committee and the Board believed that they were appropriate drivers for our business, as they provided a balance between the efforts necessary to continue to execute on a successful commercial launch for CABOMETYX in advanced RCC, further our cabozantinib development program and expand our product pipeline, all while maintaining a solid financial position, which together, would enhance stockholder value. At the time the 2018 corporate performance goals were set, the Compensation Committee and management and determinedbelieved that thesuch goals were appropriate for 2015. These goals were many-faceted, withchallenging and achieving them would require not only continued strong commercial performance, research and product development success, and prudent fiscal and legal management, but also a high level of effort and execution on the primary goals consistingpart of our Named Executive Officers.
The Compensation Committee also applied a performance weighting to each goal relative to the overall performance of the following:
Deliver top-line resultscompany to reflect the prioritization of key business objectives. Additionally, a weighting between corporate performance and individual performance was also applied for each Named Executive Officer to reflect the level of impact such individual would be able to make on the primary endpoint of progression-free survivaloverall corporate performance. The relative weighting for our phase 3 pivotal trial of cabozantinib in advanced RCC (METEOR);
Execute on our NDA filing for cabozantinib in advanced RCCeach corporate goal and launch plan if METEOR top-line results were positive;
Execute on a partnership to support the commercialization of cabozantinib in approved indications outside of the United States;
Support a successful launch of COTELLIC, in combination with vemurafenib,corporate versus individual performance is as
a treatment for patients with BRAF V600E or V600K mutation-positive advanced melanoma by our collaboration partner Genentech;follows: | | | | | | | | | | | | | | | | | | |
Corporate Performance Goals | | Weighting (%) | | | | | | Named Executive Officer | | Weighting of Corporate Performance Goals | | | Weighting of Individual Performance Assessment | |
Commercial | | | 40 | % | | | | | | Michael M. Morrissey, Ph.D. | | | 100 | % | | | 0 | % |
Product Development | | | 30 | % | | | | | | Gisela M. Schwab, M.D. | | | 60 | % | | | 40 | % |
Pipeline Development | | | 20 | % | | | | | | Christopher J. Senner | | | 60 | % | | | 40 | % |
Finance, Legal and Facilities | | | 10 | % | | | | | | Jeffrey J. Hessekiel, J.D. | | | 50 | % | | | 50 | % |
Total | | | 100 | % | | | | | | Peter Lamb, Ph.D. | | | 60 | % | | | 40 | % |
2019 Proxy Statement 49
Exceed specified fiscal 2015 revenue targets for sales of COMETRIQ for MTC in the United States; and
End fiscal 2015 with a specified cash balance, taking into account either an equity financing or a partnership for ex-U.S. rights to develop and commercialize cabozantinib.
Performance Evaluation
During 2015,2018, management reported regularly to the Compensation Committee and the Board on the status of the company’s performance against these goals, including in formal meetings in February, May, September and December. In February 2016,2019, the Compensation Committee and the Board evaluated the company’s performance in relation to the 20152018 goals, as well as, the department and personal objectives applicable toother than for Dr. Morrissey, each of the respective Named Executive Officers (other than Dr. Morrissey, whose annual bonus is based solely on company performance goals). TheOfficer’s individual contribution to the achievement of those goals. As further described below, the Compensation Committee concluded that 20152018 was a year of strong and meaningful accomplishments and noted, the following achievements, in particular:
Positive top-line results from our METEOR phase 3 pivotal trial of cabozantinib in advanced RCC;
Breakthrough Therapy and Fast Track designations for cabozantinib in advanced RCC and submission of our NDA to the FDA in December 2015 forduring which the FDA granted priority review;
Continued execution on our broad development program to explorecompany partially met, met or exceeded each of the clinical potential of cabozantinib in other tumor types;
Regulatory approval of COTELLIC in combination with vemurafenib,goals, as
a treatment for patients with BRAF V600E or V600K mutation-positive advanced melanomahighlighted by the corporate achievements identified in the
United States, European Uniontable below. | | | | | | | | | | |
| | | PERFORMANCE OBJECTIVES | | ACHIEVEMENTS | | EVALUATION | | TARGET % | | PAYOUT % |
Commercial | | 40% | | 50% |
| | | Exceed cabozantinib franchise net product revenue target established by the Board | | ›› Generated cabozantinib franchise net product revenue of $619.3 million during 2018. | | Exceeded | | | | |
| | Implement a comprehensive and rigorous launch strategy for CABOMETYX in the first-line (1L) RCC indication | | ›› Implemented 1L RCC tactical plan immediately upon FDA approval of CABOMETX as a treatment for 1L RCC. ›› Improved CABOMETYX position in marketplace relative to competitors, despite launch of competing products, according to brand marketplace metrics. ›› As of September 2018, CABOMETYX became the VEGFR-targeting tyrosine kinase inhibitor (TKI) of choice in RCC, as reflected by IQVIA National Prescription AuditTM data. | | Exceeded | | | | |
| | Implement launch readiness plan for potential second-line (2L) HCC indication | | ›› Overall launch readiness was completed on November 5, 2018, including HCC staffing and the completion of an HCC commercial strategy plan. | | Met | | | | |
50 Exelixis, Inc.
Compensation of Executive Officers | Compensation Discussion and Switzerland;Analysis
| | | | | | | | | | |
| | | PERFORMANCE OBJECTIVES | | ACHIEVEMENTS | | EVALUATION | | TARGET % | | PAYOUT % |
Product Development and Medical Affairs | | 30% | | 40% |
| | File sNDA for CABOMETYX as a treatment for 2L HCC based on CELESTIAL clinical trial results in the first quarter of 2018 | | ›› Filed sNDA with the FDA on March 15, 2018; the FDA subsequently approved CABOMETX as a treatment for patients with previously treated HCC on January 14, 2019. | | Met | | | | |
| | Initiate at least 2 late stage studies of cabozantinib alone or in combination with immune checkpoint inhibitors (ICI) in renal, bladder, liver, or thyroid cancer indications | | ›› InitiatedCOSMIC-311, evaluating cabozantinib as a monotherapy (thyroid) in October 2018 andCOSMIC-312 (liver) in December 2018. ›› Supported The Alliance for Clinical Trials in Oncology’s (The Alliance) initiation of a phase 3 trial evaluating cabozantinib in neuroendocrine tumors. The Alliance is conducting the trial through our Cooperative Research and Development Agreement with the National Cancer Institute’s Cancer Therapy Evaluation Program. ›› Extensive planning for the initiation of additional phase 3 trials in renal and bladder cancer planned for 2019. ›› Finalized launch preparedness efforts in line with regulatory timelines for a potential approval of CABOMETYX as a treatment for patients with previously treated advanced HCC. | | Exceeded | | | | |
| | Continue the broad evaluation of cabozantinib in other tumor types through internally and externally-sponsored studies | | ›› Managed or supported 77 active or planned studies, including those administered through our internal and clinical partner sponsored development program, our Cooperative Research and Development Agreement with National Cancer Institute’s Cancer Therapy Evaluation Program and our investigator sponsored trial program. ›› Studies are evaluating cabozantinib as a single agent and in combination with other therapies, including ICIs, in a variety of tumor types such asnon-small cell lung cancer, endometrial cancer, head and neck cancer, RCC, HCC and urothelial carcinoma. | | Met | | | | |
| | Add additional full-time employees across the product development team in accordance with the 2018 budget approved by the Board | | ›› 100% of planned hires for 2018 had either started, accepted offers or were in the offer process. | | Met | | | | |
| | | Support cabozantinib worldwide regulatory filings by cabozantinib partners | | ›› Supported Ipsen’s receipt of regulatory approvals in the EU and throughout the world for CABOMETYX as a 1L treatment for advanced RCC and 2L treatment for HCC by providing filing documents and assistance in responding to regulatory inquiries. ›› Assisted Takeda Pharmaceutical Company Limited’s with its clinical development activities in Japan in support of potential future regulatory filings for CABOMETYX in both RCC and HCC. | | Exceeded | | | | |
2019 Proxy Statement 51
The hiring of key leadership and expansion of our commercial and medical affairs capabilities;
Continued execution on our COMETRIQ commercialization plan for MTC, including achievement of specified fiscal 2015 revenue targets for sales of COMETRIQ for MTC in the United States;
Final fiscal 2015 cash balance of $253.3 million; and
Progress on business development activities towards a partnership to cabozantinib commercialization and development outside of the United States.
| | | | | | | | | | |
| | | PERFORMANCE OBJECTIVES | | ACHIEVEMENTS | | EVALUATION | | TARGET % | | PAYOUT % |
Pipeline Development | | 20% | | 20% |
| | Complete two early-stage oncologyin-licensing deals (not including StemSynergy) | | ›› Conducted a worldwide landscape review of oncology opportunities, followed by due diligence efforts, site visits and early stage partnering discussions for multiple preclinical and clinical stage oncology assets, resulting in the execution of an exclusive license and collaboration agreement with Invenra on May 2, 2018. | | Partially met | | | | |
| | Complete buildout of internal discovery team per 2018 budget | | ›› Hired the necessary FTEs to enable substantial internal discovery work, conducted multipleproof-of-concept experiments and advanced programs to preclinical development. | | Exceeded | | | | |
| | | Advance two discovery programs to development candidate (DC) status and initiate good laboratory practice (GLP) toxicology studies for one DC | | ›› Completed GLP toxicology study and filed an IND with the FDA for XL092 in December 2018. ›› Profiled additional late stage analogues that did not reach DC status byyear-end. | | Partially met | | | | |
Finance, Legal and Facilities | | 10% | | 15% |
| | Manage cash balance to a target established by the Board and manage operating expenses within the agreed budget and subsequent forecasts | | ›› Managed operating expenses to $415.0 million and ended fiscal 2018 with a cash balance of $851.6 million. | | Exceeded | | | | |
| | Integrate and implement compliance into all aspects of organizational activities | | ›› Appropriately managed business risks and operated in compliance with applicable laws, regulations, and guidance. | | Met | | | | |
| | Complete buildout and move into Alameda facility by end of June 2018 | | ›› Completed the renovation and buildout of new corporate headquarters in Alameda, California in June 2018 and successfully relocated our employee base in the same month. | | Met | | | | |
| | | | | | |
| | | | | | | TOTAL | | 100% | | 125% |
The Compensation Committee did not assign any weighting of specific company, departmental or personal goals in making its assessment of overall performance, but rather made its assessment of performance againstalso recognized the goals on an overall basis. Viewing the performance and resultsindividual contributions, other than for Dr. Morrissey, toward achievement of the year,corporate goals and the combined contribution of our Named Executive Officers and cohesive management effort that led to these 2018 corporate achievements. Specifically, the Compensation Committee considered the following contributions from each Named Executive Officer, other than Dr. Morrissey:
| ›› | Dr. Schwab’s overall leadership of our product development and medical affairs organizations and her performance with respect to the execution of our product development goals in 2018, particularly regarding the filing and acceptance of our sNDA with the FDA for cabozantinib as a treatment for patients with previously treated HCC, the initiation ofCOSMIC-311 andCOSMIC-312, support of cabozantinib worldwide regulatory filings by collaboration partners, and the oversight of a rapidly growing Product Development organization; |
| ›› | Mr. Senner’s overall leadership of our finance organization and his performance with respect to the execution of our financial goals in 2018, particularly regarding our disciplined expense management; |
| ›› | Mr. Hessekiel’s overall leadership of our legal and compliance organizations and his performance with respect to fulfillment of our business and legal goals in 2018, particularly regarding the integration and implementation of compliance throughout a rapidly expanding organization; and |
| ›› | Dr. Lamb’s overall leadership of our research and discovery organization and his performance with respect to our pipeline development goals in 2018, particularly regarding the rebuilding of our discovery organization, research progress made with respect to potential development candidates, the filing of an IND with the FDA for XL092 and the execution of our license and collaboration agreements with StemSynergy and Invenra. |
52 Exelixis, Inc.
Compensation of Executive Officers | Compensation Discussion and Analysis
In consideration of our overall performance during 2018 and the contributions of our Named Executive Officers to such performance, in February 2019, pursuant to the terms and conditions of the Annual Cash Bonus Plan, the Compensation Committee approved annual cash bonus payments for each Named Executive Officer as reflected in the table below.
When determining the annual cash bonus payment for Dr. Morrissey in particular, the Compensation Committee considered our meaningful accomplishments in 2018 and level of achievement of pre-determined corporate goals. However, the Compensation Committee also considered that it is Dr. Morrissey’s responsibility to create stockholder value, and because our stock price declined during 2018, the Compensation Committee used its discretion, as provided for in the Annual Cash Bonus Plan, to reduce Dr. Morrissey’s 2018 cash bonus below the level of the overall corporate performance. Dr. Morrissey’s annual cash bonus payment is also reflected in the table below.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Name | | 2018 Base Salary | | | 2018 Target Award (%) | | | 2018 Corporate Performance Weighting (%) | | | 2018 Approved Corporate Performance (%) | | | 2018 Individual Performance Weighting (%) | | | 2018 Individual Performance (%) | | | 2018 Annual Cash Bonus Payout (% of Target Award) | | | 2018 Annual Cash Bonus Payout ($) | |
| Michael M. Morrissey, Ph.D. | | $ | 955,060 | | | | 100 | % | | | 100 | % | | | 125 | % | | | N/A | | | | N/A | | | | 105 | % | | $ | 1,000,000 | |
| Gisela M. Schwab, M.D. | | $ | 680,520 | | | | 50 | % | | | 60 | % | | | 125 | % | | | 40 | % | | | 155 | % | | | 137 | % | | $ | 466,156 | |
| Christopher J. Senner | | $ | 601,020 | | | | 45 | % | | | 60 | % | | | 125 | % | | | 40 | % | | | 125 | % | | | 125 | % | | $ | 338,074 | |
| Jeffrey J. Hessekiel, J.D. | | $ | 536,597 | | | | 45 | % | | | 50 | % | | | 125 | % | | | 50 | % | | | 125 | % | | | 125 | % | | $ | 301,836 | |
| Peter Lamb, Ph.D. | | $ | 497,525 | | | | 45 | % | | | 60 | % | | | 125 | % | | | 40 | % | | | 160 | % | | | 139 | % | | $ | 311,202 | |
2018 Long-Term Incentive Awards
Because of our goal to align executive compensation and performance that advances our critical business objectives, a significant portion of the Named Executive Officers’ total compensation typically has consisted of, and is expected to continue to consist of, equity-based awards. In evaluating the mix of equity awards for 2018, the Compensation Committee considered market trends, as well as feedback from stockholders and proxy advisory firms, and determined that a combination of stock options and PSUs would be the companymost appropriate incentive structure for our Named Executive Officers to reward performance over time and achieve our retention objectives.
Allocation of Long-Term Incentive Awards
For 2018, the Compensation Committee intended to deliver approximately 50% of the value of each Named Executive Officer’s equity award in the form of stock options and 50% of the value in the form of PSUs (except for Mr. Hessekiel who received a mix of 65% stock options and 35% PSUs) and applied a 2 to 1 ratio of stock option grants to PSUs, in order to mitigate dilution and to reflect the increased value of receiving shares at full value. The mix for each Named Executive Officer took into consideration both peer practices and market data, as a wholewell as, in the case of Mr. Hessekiel, his ability to impact the achievement of key corporate objectives in his position as Executive Vice President and General Counsel relative to the rest of the Named Executive Officers.
2019 Proxy Statement 53
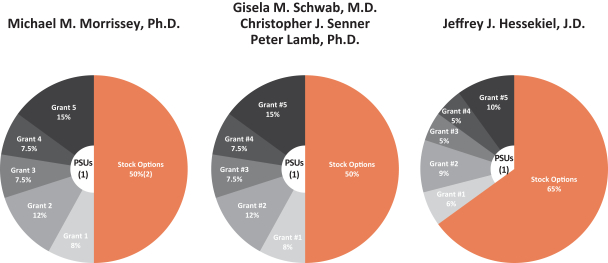
| (1) | Performance criteria for the PSU awards are described in the table below. |
| (2) | Dr. Morrissey’s stock option is subject to a market performance condition as further described below. |
The stock options for each of the Named Executive Officers had performed well. However, given the company’s continued cash conservation efforts and the Compensation Committee's goal to further align the interests of the Named Executive Officer's with our stockholders, the Compensation Committee determined that 2015 annual cash bonuses would be paid in the form of equity in lieu of cash. On February 11, 2016, the Compensation Committee approved the grant of fully-vested RSUs in the following amounts:
|
| | |
Name | | Number of Shares
Subject to RSUs
|
Michael M. Morrissey, Ph.D. | | 114,286 |
Christopher J. Senner | | 53,572 |
Deborah Burke | | 27,301 |
Jeffrey J. Hessekiel, J.D. | | 49,902 |
Peter Lamb, Ph.D. | | 45,378 |
Gisela M. Schwab, M.D. | | 65,498 |
In determining the number of RSUs for each Named Executive Officer, the Compensation Committee assumed a bonus payment of 100% of the Named Executive Officer's cash bonus target for 2015 (with the exception of Dr. Schwab for whom a bonus payment of 111% of her cash bonus target for 2015 was assumed) divided by $4.20, the closing market price of our common stock on February 11, 2016. The Compensation Committee determined that Dr. Schwab’s exceptional efforts in leading the development team through major milestones, including obtaining positive clinical results from our phase 3 pivotal trial METEOR, receipt of Breakthrough Therapy and Fast Track designation from the FDA for cabozantinib in RCC, and swiftly completing our NDA submission to the FDA, warranted a bonus award at 50%, which is 111% of her 2015 target of 45%.
2015 Equity Incentive Awards
2015 Performance-Based Stock Options Grants. In February 2015, in light of the fact that no cash bonuses were paid for 2014 due in part to cash conservation efforts and the need for all employees, including our executives, to remain focused on the achievement of critical milestones in our development of cabozantinib, the Compensation Committee recommended, and the Board approved, a grant of performance-based stock options to the Named Executive Officers. As part of this process, management engaged Radford to assist in recommending the appropriate number of stock options for each executive, with equity values for each executive targeted at approximately 100% of the executive’s cash bonus target. Following its review, the Compensation Committee granted the following number of performance-based stock options to the Named Executive Officers:
|
| | |
Name | | Number of Shares Subject to Performance-Based
Stock Options
|
Michael M. Morrissey, Ph.D. | | 450,000 |
Deborah Burke | | 175,000 |
Jeffrey J. Hessekiel, J.D. | | 200,000 |
Peter Lamb, Ph.D. | | 175,000 |
Gisela M. Schwab, M.D. | | 250,000 |
The stock options have an exercise price of $1.90$18.80 per share, the fair market value of our common stock on the date of grant (September 10, 2018), and expire seven years from the date of grant or earlier upon termination of service with us. VestingAll of the stock options is subject to the achievement of critical clinical, regulatory, and commercialization goals set by the Compensation Committee and approved by the Board, as follows: (i) 50% of the options would vest if the Compensation Committee determined that top-line efficacy data received from the METEOR phase 3 pivotal trial of cabozantinib in advanced RCC met its primary endpoint at a specified level, with such result to occur no later than a specified date; (ii) 25% of the options would vest if the Compensation Committee confirmed that a new drug application for cabozantinib for the treatment of advanced RCC is accepted for review by the FDA by a specified date; and (iii) 25% of the options will vest if the Compensation Committee confirms that the FDA has approved cabozantinib for the treatment of advanced RCC by a specified date. On July 20, 2015, the Compensation Committee convened to determine that top-line efficacy data received from METEOR met its primary endpoint at the level specified and within the time period permitted by the performance goals, resulting in the vesting of 50% of the option. On March 7, 2016, the Compensation Committee convened to determine that the NDA for cabozantinib for the treatment of advanced RCC was accepted for review by the FDA within the time period permitted by the performance goals, resulting in the vesting of 25% of the option. If the Compensation Committee has not concluded that FDA has approved cabozantinib for the treatment of advanced RCC by the specified date, the performance stock award with respect to that performance goal will be forfeited. We are not disclosing certain additional details with respect to the goals or deadlines because we believe that their disclosure might cause us competitive harm.The Compensation Committee believes the remaining target deadlines it has established are achievable with an appropriate amount of diligent effort and expertise. However, the Compensation Committee believes that at the time the goals were set, there would be a substantial degree of difficulty in achieving the goals, particularly due to the inherent unpredictability of clinical trial outcomes and of regulatory approval processes.
2015 Ongoing Equity Grants. In September 2015, the Compensation Committee reviewed the annual equity compensation awards for our Named Executive Officers for 2015. In determining the form of equity incentive grants for the Named Executive Officers, the Compensation Committee:
evaluated the status of equity incentive awards held by the Named Executive Officers to assess the retention and incentive values of those awards since prior awards made to the Named Executive Officers were either (i) not in the money at the time the Compensation Committee was considering grants for 2015 or (ii) the majority of performance targets tied to the vesting of outstanding performance-based stock options had been met or were highly likely to be achieved;
determined the appropriate size and value of new equity incentive awards for our Named Executive Officers during the coming year, taking into consideration the need to be competitive in the market to retain the talent required to navigate the company through a potential commercial launch in advanced RCC; and
recommended the grant of stock options since options only have value if the value of our company, as reflected by our stock price, increases over time and thus incentivizes our executives to perform for the benefit of our stockholders.
When determining the appropriate size and value of new equity incentive awards, management asked Radford to provide guidance with respect to implementing a program that would incentivize our Named Executive Officers to drive toward the achievement of key company priorities and increase stockholder value over the long-term, while maintaining acceptable overhang and burn rates with respect to our equity use. The Compensation Committee reviewed materials provided by Radford and determined that, to be both impactful and within acceptable share use limits, grants should be sized between the 50th and 75th percentile of our peer group for each of our Named Executive Officers. In September 2015, the Board accepted the Compensation Committee’s recommendations and approved the following grants to the Named Executive Officers:
|
| | |
Name | | Number of Shares Subject to Time-Based Stock Options |
Michael M. Morrissey, Ph.D. | | 500,000 |
Christopher J. Senner | | 225,000 |
Deborah Burke | | 64,000 |
Jeffrey J. Hessekiel, J.D. | | 190,000 |
Peter Lamb, Ph.D. | | 190,000 |
Gisela M. Schwab, M.D. | | 245,000 |
The stock options have an exercise price of $6.21 per share, the fair market value of our common stock on the date of grant, and expire seven years from the date of grant or earlier upon termination of service with us. The stock options vest over a
four yearfour-year period following the grant date.
Dr. Morrissey’s stock option is also subject to a market performance condition and will not be exercisable until the closing market price of our common stock is equal to or greater than 125% of the exercise price of the option over a period of at least 30 consecutive calendar days.The PSUs granted to Named Executive Officers were allocated among five separate awards and only vest upon the achievement of certain commercial, clinical development and pipeline expansion performance targets, as described below. The Compensation Committee selected these performance targets because they represent the critical business objectives for which the Named Executive Officers are responsible, thereby linking executive compensation with continued long-term success for the company. Additional detail about each performance target will be disclosed at the end of the relevant performance period, as disclosing the specific, numerical target before that time could be competitively harmful to the company.
| | | | | | |
PSU Grant # | | Business Focus | | Performance Target | | Factors Considered to Establish Rigorous
Performance Targets |
1 | | Commercial | | Cabozantinib global net product revenue target in a single quarter | | Requires successful execution of our cabozantinib commercial strategy, such that we achieve or exceed internal benchmarks for performance derived from product sales forecasts and predictions of competitive market dynamics |
2 | | Commercial | | Cabozantinib global net product revenue target over four consecutive quarters | | Requires successful execution of our cabozantinib commercial strategy, such that we achieve or exceed internal benchmarks for performance derived from product sales forecasts and predictions of competitive market dynamics |
3 | | Development | | Initiation of a target number of potentially label-enabling clinical trials for cabozantinib (both alone or in combination with another therapy) | | Requires successful execution of the cabozantinib clinical development and regulatory program by the company and its clinical collaboration partners |
54 Exelixis, Inc.
Compensation of Executive Officers | Compensation Discussion and Analysis
| | | | | | |
PSU Grant # | | Business Focus | | Performance Target | | Factors Considered to Establish Rigorous
Performance Targets |
4 | | Development | | Achievement of either (I) full target enrollment, as specified in the applicable clinical trial protocol, or (II) positivetop-line results, in each case with respect to a target number of potentially label-enabling clinical trials for cabozantinib (both alone or in combination with another therapy) | | Requires significant operational progress on the cabozantinib clinical development program by the company and its clinical collaboration partners, along with positive results from ongoing and planned cabozantinib clinical trials |
5 | | Pipeline | | Approval or acceptance by the FDA of a target number of IND applications (or equivalent filings with foreign governmental authorities) with respect to product candidates that have either (x) been discovered by us or(y) in-licensed or acquired by us | | Requires successful execution of our internal drug discovery research program, as well as successful execution of our strategy for the identification, in-licensing or acquisition of promising early-stage product candidates and their advancement to IND status |
Each PSU will vest as to 50% of the original number of shares subject to the award upon the Compensation Committee’s certification that we have achieved the performance target and 50% of the original number of shares subject to the award on the first quarterly vesting date (i.e. February 15th, May 15th, August 15th and November 15th) following theone-year anniversary of the Compensation Committee’s certification. Failure to achieve a performance target by December 31, 2021 will result in forfeiture of 100% of the applicable PSU award.
Vesting of these stock options and PSUs will cease upon termination of service as an employee for any reason. The Named Executive Officers are party to our Change in Control and Severance Benefit Plan, which provides for acceleration of vesting of the award in the event of certain specified change in control events involving us, for the reasons discussed above.
As an inducement In addition, following certain change in control events wherein the PSU awards are assumed by the surviving entity, the PSU awards will convert to
Mr. Senner's appointment as Executive Vice Presidenttime-based vesting and
Chief Financial Officer,vest annually over a three-year period following the grant date.Value of Long-Term Incentive Awards
When determining the appropriate value of equity incentive awards, the Compensation Committee approved the grant of (i)asked Compensia to provide guidance with respect to implementing a time-based stock option award in the amount of 350,000 shares, with an exercise price of $3.66 per share, the fair market value of Exelixis' common stock on the date of grant, which was July 15, 2015, the date Mr. Senner commenced employment with the company, which option will vest over a four year period following the date of grant and (ii) a restricted stock unit award in the amount of 100,000 shares, which will vest in full on the one-year anniversary of the date of grant. The option award will expire seven years from the date of grant or earlier upon termination of service with us. When approving his compensation at the time he was hired, the Compensation Committee solicited the input of and received documentary support fromprogram that would incentivize our senior human resources personnel. Using this information and considering the compensation Mr. Senner would forfeit to join our company, the Compensation Committee approved the forms and levels of Mr. Senner's equity compensation, within which his ultimate compensation was established during arms-length negotiations.
2016 Special Equity Incentive Compensation. In February 2016, the Compensation Committee approved a special one-time grant of stock options to Drs. Morrissey, Lamb and Schwab, in consideration of their exceptional service to our company during 2015 and the desire to retain the executive talent necessaryNamed Executive Officers to drive toward the achievement of our company's future commercialkey company priorities and researchincrease stockholder value over the long-term, while maintaining competitive market practices and development goals, including the potential commercial launch of cabozantinib in advanced RCC. In determining the sizebeing mindful of the awardcompany’s equity burn rate.As part of the decision-making process, the Compensation Committee considered the challenges of managing a rapidly expanding organization, as well as our need to retain a cohesive management team to facilitate the continued commercial success of CABOMETYX, and the achievement of product development and pipeline expansion objectives that would position the company for future success. The Compensation Committee also believed it was important that the value of the equity awards continue to reflect the individual performance of each Named Executive Officer during the 2018 fiscal year to date and criticality of each Named Executive Officer’s skill set and expected future contributions to our business. Taking these factors into consideration and applying Compensia’s market analysis of long-term incentive compensation of our Named Executive Officers compared to our 2018 peer group, the Compensation Committee considered a number of factors, including an assessment of individual performance,determined that the individual’s overall contributions and the company's current equity reserve. However, the Compensation Committee did not use a formula or assign a particular weight to any one factor when it determined the sizevalue of the award. Rather, the Compensation Committee used a subjective approach, and awarded the specialaggregate equity grants at levels it believes in its judgment are appropriate and helpawards granted to further align the interestseach of these particularour Named Executive Officers with thoseshould be between the 50th and 75th percentile of our stockholders. On February 11, 2016, the Compensation Committee,market peers and in consultation with the Board,September 2018, approved the following grants to the Named Executive Officers identified below:
|
| | |
Name | | Number of Shares Subject to Time-Based Stock Options |
Michael M. Morrissey, Ph.D. | | 150,000 |
Peter Lamb, Ph.D. | | 40,000 |
Gisela M. Schwab, M.D. | | 75,000 |
The stock options have an exercise price of $4.20 per share, the fair market value of our common stock on the date of grant, and expire seven years from the date of grant or earlier upon termination of service with us. The stock
options vest over a four year period following the date of grant. Vesting of these stock options will cease upon termination of service as an employee for any reason. The Named Executive Officers identifiedsummarized in the table above are partybelow. The actual share amounts granted to our Change in Control and Severance Benefit Plan, which provides for acceleration of vesting ofeach executive officer were determined by dividing the award in the event of certain specified change in control events involving us, for the reasons discussed above.
Compensation of the Chief Executive Officer for 2015
The Compensation Committee and the Board considered the factors and criteria described under the heading “2015 Compensation Decisions” above in determining Dr. Morrissey’s total compensation for 2015. The Board increased Dr. Morrissey’s base salary for 2015 by 4% over his 2014 base salary. Consistent with the Compensation Committee’s recommendation, the Board has kept Dr. Morrissey’s target cash bonus at 60% of base salary, which is higher than the 45% target for the other Named Executive Officers (other than Ms. Burke’s whose target is 35% of base salary) due to his position as Chief Executive Officer and the critical role he plays at the company. This percentage has not changed since 2011, is consistent with industry practice and reflects Dr. Morrissey’s greater role in determining the course, and ability to influence the future, of the company. In line with the Compensation Committee’s recommendation to issue RSUs in lieu of paying cash bonuses for 2015, Dr. Morrissey received a grant of 114,286 RSUs which vested in full upon grant. In determining the number of RSUs for Dr. Morrisseyvalue the Compensation Committee
assumed a bonus payment of 100% of Dr. Morrissey's cash bonus target for 2015 dividedintended to deliver by
$4.20, the
closing market30-day average trailing stock price
of the Company's common stock on February 11, 2016. In February 2015, upon recommendation of the Compensation Committee, in lieu(as of a
cash bonus paid to Dr. Morrissey for 2014,date shortly before the
Board granted 450,000 performance-based stock options subject to specified vesting conditions tied to our METEOR trial related to cabozantinib for the treatment of advanced RCC. In determining the equity value for Dr. Morrissey, the Compensation Committee targeted approximately 100% of Dr. Morrissey's cash bonus target. In September 2015, upon recommendation of the Compensation Committee, the Board granted Dr. Morrissey a time-based stock option to purchase 500,000 shares of our common stock which was substantially larger than those made to our other Named Executive Officers, given his positiongrant date) in the
companycase of PSUs and
applying a Black-Scholes option pricing model calculation using the
other factors described above. The option grant is subjectaverage share price, in the case of stock options.A 30-day average share price was used, rather than a single day share price, in order to
our standard vesting schedule overprovide a
four year period followingmore stabilized share value less susceptible to possible swings in the
date of grant. In February 2016, in consideration for his exceptional service to our company and the desire to retain Dr. Morrissey to lead our company through the potential commercial launch of cabozantinib in advanced RCC, the Compensation Committee, in consultation with the Board, granted to Dr. Morrissey 150,000 additional time-based stock options, subject to the same standard vesting schedule.market.2019 Proxy Statement 55
| | | | | | | | | | | | |
Name | | Number of Shares
Subject to Time-
Based Stock
Options | | | Number of Shares
Subject to Time- and
Performance- Based Stock Options | | | Number of Shares
Subject to PSUs | |
Michael M. Morrissey, Ph.D. | | | — | | | | 308,365 | | | | 154,923 | |
Gisela M. Schwab, M.D. | | | 129,314 | | | | — | | | | 64,968 | |
Christopher J. Senner | | | 129,314 | | | | — | | | | 64,968 | |
Jeffrey J. Hessekiel, J.D. | | | 129,314 | | | | — | | | | 34,983 | |
Peter Lamb, Ph.D. | | | 124,341 | | | | — | | | | 62,469 | |
Other Compensation Information
GrantsAnnual grants of equity awards to our executive officers, including our Named Executive Officers, are generally determined and approved at our pre-scheduled Compensation Committee meetings, whenever practicable.with such meeting date typically serving as the grant date. However, the Compensation Committee may otherwisesometimes approve the grant of equity awards to executive officers and other employees in advance of its next scheduled meeting, either at a special meeting or by unanimous written consent, in connection with acertain new hire, promotion,hires, promotions and other circumstances where the Compensation Committee deems it appropriate to make such grants. The grant dates for these equity awards are typically the same date that a newly hired officer begins employment or the effective date of an officer’s promotion, as applicable. Grants made to new hires below the level of Vice President, are approved on a monthly basis by our Equity Award Committee, comprised solely of Dr. Morrissey, acting in his capacity as a director pursuant to authority delegated to him by the Compensation Committee. All stock options are granted with an exercise price that is not less than the closing price of our common stock on The NASDAQNasdaq Global Select Market on the grant date. It is our policy not to purposely accelerateWe have no plan or delay the public release of material information in consideration of a pending equity grant to allow the grantee to benefit from a more favorable stock price. We recognize that a release of information in close proximity to an equity grant may appear to be an effortpractice to time option grants in coordination with the announcementrelease ofnon-public information, and we do not time the release ofnon-public information to a grantee’s benefit (even if no such benefit was intended). Accordingly, it isaffect the value of executive compensation.
Stock Ownership Guidelines
In February 2018, the Board adopted Stock Ownership Guidelines for our policy thatNamed Executive Officers in order to further align their financial interests with those of our management team makes a good faith effortstockholders, as well as to advisepromote sound corporate governance. The Stock Ownership Guidelines for our Named Executive Officers provide for ownership targets as follows:
| | |
Position | | Ownership Level |
Chief Executive Officer | | Lesser of 160,000 shares or a value equivalent to 5 times annual base salary |
Other Named Executive Officers | | Lesser of 25,000 shares or a value equivalent to 1.5 times annual base salary |
In considering appropriate guidelines for our executive officers and directors, the Compensation Committee whenever itreviewed a market analysis of stock ownership guidelines of our 2018 peer group. The approved guidelines provide for a “lesser of” approach to address stock price volatility often experienced by biotechnology companies.
All Named Executive Officers are expected to achieve their stock ownership targets within five years of becoming subject to these guidelines. The policy includes procedures for granting exemptions in the case of severe financial hardship. In determining ownership levels for each Named Executive Officer under our Stock Ownership Guidelines, credit is aware that material non-public informationprovided for shares held outright (including shares owned through trusts, our 401(k) Plan, or by a spouse), as well as 50% of the number of vested, but unexercised, stock options. No credit is plannedprovided for RSUs until they vest. The values for all shares determined to be releasedheld by Named Executive Officers are based on the200-day average stock price as of the measurement date. As of February 12, 2019, all of our Named Executive Officers had met the required ownership targets.
“Clawback” Policy
We are dedicated to maintaining a culture of high integrity and accountability, and to discourage conduct harmful to our business and the interests of our various stakeholders. On February 28, 2019, upon recommendation from the Compensation Committee, the Board approved aPolicy for Recoupment of Variable Compensation (Clawback Policy) that applies to all forms of compensation, except base salary, granted after the adoption of the Clawback Policy. A triggering event under the
56 Exelixis, Inc.
Compensation of Executive Officers | Compensation Discussion and Analysis
Clawback Policy shall occur when a covered employee (which includes all Named Executive Officers):
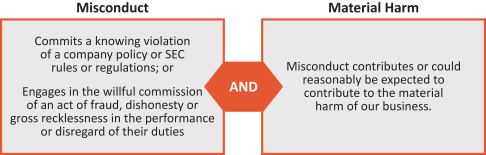
For clarity, “material harm” includes, but is not limited to, the public in close proximityrequirement to prepare an accounting restatement for any fiscal quarter or year commencing after adoption of the grant of equity awards.
“Clawback”Clawback Policy
The Compensation Committee has not yet established a policy to recover bonuses from our executive officers if the performance objectives that led to a bonus determination were to be restated, or found not to have been met to the extent originally believed by the Compensation Committee. As a public company, if we are required to restate our financial results due to our material noncompliance with any financial reporting requirements underrequirement. If triggered, then to the federal securities laws as a result of misconduct, our Chief Executive Officer and Chief Financial Officerfullest extent permitted by law, we may be legally requiredrecoup all variable compensation granted or paid to reimburse us for any bonus or other incentive-based or equity-based compensation they receive,the covered employee during each fiscal year in accordance withwhich the provisions of Section 304 of the Sarbanes-Oxley Act of 2002. Further, the Dodd-Frank Wall Street Reform and Consumer Protection Act, or Dodd-Frank Act, requires that the SEC promulgate rules which would require that, in the event we are required to restate our financial statements, we “claw back” any bonuses paid
based on financial performance that would not have been paid if based on the restated financial performance. The SEC has not yet finalized its “claw back” rules and we expect that the Compensation Committee and Board will re-evaluate the potential adoption of a “claw back” policy once such rules have been promulgated.
covered employee’s “misconduct” occurred.Accounting and Tax Considerations
Under Financial Accounting Standard Board ASC Topic 718, orFASB ASC 718, we are required to estimate and record an expense for each award of equity compensation (including stock options, RSUs and RSUs)PSUs) over the vesting period of the award. As long as stock options, RSUs and RSUsPSUs remain the sole components of our long-term compensation program, we expect to record stock-based compensation expense on an ongoing basis according to ASC 718. Compensation expense relating to awards subject to performance conditions is recognized if it is probable that the performance goals will be achieved. The probability of achievement of such goals is assessed on a quarterly basis. The Compensation Committee has considered, and may in the future consider, the grant of restricted stock to our executive officers in lieu of stock option grants, RSU and/or RSUPSU awards.
Section 162(m) of the Code, as amended by the Tax Cuts and Jobs Act, limits our deductionthe deductibility for federal income tax purposes to not more than $1 million of compensation paid to certain executive officers“covered employees” in a calendar year. Compensation abovePrior to the recent enactment of the Tax Cuts and Jobs Act, compensation that qualified as “performance-based compensation” under Section 162(m) of the Code was not subject to this deduction limitation. Pursuant to the Tax Cuts and Jobs Act, this exception for “performance-based compensation” under Section 162(m) of the Code was repealed with respect to taxable years beginning after December 31, 2017, except that certain transition relief is provided for compensation paid pursuant to a written binding contract which was in effect on November 2, 2017, and which was not modified in any material respect on or after such date. As a result, compensation paid to any of our “covered employees” in excess of $1 million mayper taxable year generally will not be deducted ifdeductible unless, among other requirements, it is “performance-based compensation.” Our Compensation Committee has not yet established a policy for determining which forms of incentive compensation awardedintended to our executive officers should be designatedqualify, and is eligible to qualify, as “performance-based compensation.” To maintain flexibility in compensating our executive officers in a mannercompensation” under Section 162(m) of the Code pursuant to the transition relief provided by the Tax Cuts and Jobs Act. Certain equity awards issued prior to the transition date were designed to promote our objectives,permit us to qualify for the performance-based exception, but because of certain ambiguities and uncertainties as to the application and interpretation of Section 162(m) of the Code and the regulations issued thereunder, as well as other factors beyond the control of the Compensation Committee, has not adopted a policyno assurance can be given that requires allany compensation topaid by the company will be deductible. However,eligible for such transition relief and be deductible by the company in the future. Although the Compensation Committee intendswill continue to evaluate the effects of theconsider tax implications as one factor in determining executive compensation, limits of Section 162(m) on any compensation it proposes to grant, and the Compensation Committee intendsalso looks at other factors in making its decisions and retains the flexibility to provide future compensation for the Named Executive Officers in a manner consistent with ourthe goals of the company’s executive compensation program and the best interests of the company and those of our stockholders.
its stockholders, which may include providing for compensation that is not deductible by the company due to the deduction limit under Section 162(m). The Compensation Committee also retains the flexibility to modify compensation that was initially intended to be exempt from the deduction limit under Section 162(m) if it determines that such modifications are consistent with the company’s business needs.Compensation Policies and Practices as They Relate to Risk Management
In 2015,2018, the Compensation Committee reviewed our compensation policies and practices and concluded that the mix and design of these policies and practices are not reasonably likely to encourage our employees to take excessive risks. In connection with its evaluation, the Compensation Committee considered, among other things, the structure, philosophy and
2019 Proxy Statement 57
design characteristics of our primary incentive compensation plans and programs in light of our risk management and governance procedures, as well as other factors that may calibrate or balance potential risk-taking incentives. Based on this assessment, the Compensation Committee concluded that risks arising from our compensation policies and practices for all employees, including executive officers, are not reasonably likely to have a material adverse effect on us.
Conclusion
It isForward-Looking Statements
This CD&A contains forward-looking statements, including, without limitation, statements related to: Exelixis’ expectation that MINNEBRO will soon enter the Compensation Committee’s opinion thatmarketplace in Japan; Exelixis’ goal to become a fully integrated biotechnology company; the compensation policies and elements discussed above providepotential for the necessary incentivestreatment market for HCC to align our performance and the interests of our stockholders properly while maintaining balanced and competitive executive compensation practices that enable us to attract and retain the highest caliber of executives. The year 2015 was a significant inflection point for our company: we obtained positive clinical results from our phase 3 pivotal trial METEOR, received Breakthrough Therapy and Fast Track designation from the FDA for cabozantinib in RCC, and swiftly completed our NDA submission to the FDA. These events constituted major milestones for our company, and moved us closer to our commercial launch of cabozantinib in advanced RCC. The compensation of our employees, including our Named Executive Officers, was reflective of not only of those achievements, but also the efforts of our employees to execute on the company's proposed goals, and to ensure appropriate focus on our future business objectives. Our immediate focus now is to prepare for a potential commercial launch of cabozantinibgrow significantly in the United Statescoming years; the potential of cabozantinb as a treatment option for patients with advanced RCC. The Compensation Committee believesradioactive iodine-refractory DTC who have progressed after prior VEGFR-targeted therapy; the potential for Ipsen to make future regulatory submissions outside of the U.S. and Japan in HCC, based on the results of COSMIC-312; the anticipated organizational growth of Exelixis; Exelixis’ plans to initiate additional phase 3 trials in renal and bladder cancer in 2019; the potential for Takeda Pharmaceutical Company Limited to make future regulatory filings for CABOMETYX in Japan in both RCC and HCC indications; and other statements that are not historical fact. Any statements that refer to expectations, projections or other characterizations of future events or circumstances are forward-looking statements and are based upon Exelixis’ current plans, assumptions, beliefs, expectations, estimates and projections. Forward-looking statements involve risks and uncertainties. Actual results and the compensation policiestiming of events could differ materially from those anticipated in the forward-looking statements as a result of these risks and elements described above,uncertainties, which include, without limitation: the degree of market acceptance of CABOMETYX, COMETRIQ, COTELLIC and MINNEBRO in the territories where they are approved, and the availability of sufficient coverage and adequate reimbursement for these products; the effectiveness of CABOMETYX, COMETRIQ, COTELLIC and MINNEBRO in comparison to competing products; the level of costs associated with Exelixis’ commercialization, research and development, in-licensing or acquisition of product candidates, and other activities; the potential failure of cabozantinib, cobimetinib, esaxerenone and other Exelixis product candidates, both alone and in combination with other therapies, to demonstrate safety and/or efficacy in clinical testing; uncertainties inherent in the drug discovery and product development process; Exelixis’ dependence on its relationships with its collaboration partners, including their pursuit of regulatory approvals for partnered compounds in new indications, their adherence to their obligations under relevant collaboration agreements and the emphasis upon long-term equity incentive compensation, will encouragelevel of their investment in the company’s current employees, including its executive officers, to strive for the successful achievement of our immediate commercialization objectives and both short-term and long-term clinical development goals. The Compensation Committee further believes that these policies and elements support our objective of closely managing our expenses to conserve the cash resources necessary to secure long-term value appreciation.complete clinical trials or successfully commercialize partnered compounds in the territories where they are approved; risks and uncertainties related to regulatory review and approval processes; Exelixis’ continuing compliance with applicable legal and regulatory requirements; unexpected concerns that may arise as a result of the occurrence of adverse safety events or additional data analyses of clinical trials evaluating cabozantinib, cobimetinib or esaxerenone; Exelixis’ dependence on third-party vendors for the manufacture and supply of its products; Exelixis’ ability to protect its intellectual property rights; market competition, including the potential for competitors to obtain approval for generic versions of Exelixis’ marketed products; changes in economic and business conditions; and other factors discussed under the caption “Risk Factors” in Exelixis’ Annual Report on Form 10-K filed with the SEC on February 22, 2019, and in Exelixis’ future filings with the SEC. All forward-looking statements in this CD&A are based on information available to Exelixis as of the date of this Proxy Statement, and Exelixis undertakes no obligation to update or revise any forward-looking statements contained herein.
58 Exelixis, Inc.
Compensation of Executive Officers |Summary of Compensation
Summary of Compensation
The following table shows for the fiscal years ended January 1, 2016, January 2, 2015December 28, 2018, December 29, 2017 and December 27, 201330, 2016 (referred to as fiscal years 2015, 20142018, 2017 and 2013,2016, respectively), compensation awarded to, paid to or earned by our Chief Executive Officer, each person serving as our Chief Financial Officer, in 2015, and our other three most highly compensated executive officers in 2015,2018, which we refer to as our “Named Executive Officers.”
Summary Compensation Table for Fiscal 2015
|
| | | | | | | | | | | | | | | | | | | | |
| Name and Principal Position | | Year (1) | | Salary ($)(2) | | Bonus ($)(3) | | Stock Awards ($)(4) | | Option Awards ($)(5) | | All Other Compensation ($)(6) | | Total ($) |
| Michael M. Morrissey, Ph.D. | | 2015 | | 824,423 |
| | 480,001 |
| (7) | — |
| | 2,693,750 |
| | 9,326 |
| | 4,007,500 |
|
| President and Chief | | 2014 | | 755,192 |
| | — |
| | — |
| | 1,378,800 |
| | 7,800 |
| | 2,141,792 |
|
| Executive Officer | | 2013 | | 684,003 |
| | 420,000 |
| | — |
| | 3,713,760 |
| | 7,650 |
| | 4,825,413 |
|
| Christopher J. Senner* | | 2015 | | 226,923 |
| (8) | 225,002 |
| (7) | 366,000 |
| | 1,809,593 |
| | — |
| | 2,627,518 |
|
| Executive Vice President and | | | | | | | | | | | | | | |
| Chief Financial Officer | | | | | | | | | | | | | | |
| Deborah Burke**^ | | 2015 | | 337,535 |
| | 115,210 |
| (7) | — |
| | 491,802 |
| | 6,578 |
| | 951,125 |
|
| Senior Vice President, Finance | | 2014 | | 288,873 |
| (9) | 30,000 |
| (10) | — |
| | 459,600 |
| | 7,800 |
| | 786,273 |
|
| and Controller | | | | | | | | | | | | | |
|
| Jeffrey J. Hessekiel, J.D.** | | 2015 | | 480,332 |
| | 209,588 |
| (7) | — |
| | 1,062,021 |
| | — |
| | 1,751,941 |
|
| Executive Vice President, General | | 2014 | | 380,769 |
| (11) | — |
| | — |
| | 1,657,536 |
| | — |
| | 2,038,305 |
|
| Counsel and Secretary | | | | | | | | | | | | | | |
| Peter Lamb, Ph.D.** | | 2015 | | 437,199 |
| | 190,588 |
| (7) | — |
| | 1,028,921 |
| | 9,464 |
| | 1,666,172 |
|
| Executive Vice President, | | 2014 | | 407,042 |
| | — |
| | — |
| | 490,240 |
| | 7,800 |
| | 905,082 |
|
| Scientific Strategy and Chief | | | | | | | | | | | | | | |
| Scientific Officer | | | | | | | | | | | | | | |
| Gisela M. Schwab, M.D. | | 2015 | | 565,800 |
| | 275,092 |
| (7) | — |
| | 1,358,996 |
| | 7,950 |
| | 2,207,838 |
|
| President, Product Development | | 2014 | | 513,906 |
| | — |
| | — |
| | 612,800 |
| | 7,800 |
| | 1,134,506 |
|
| and Medical Affairs and Chief | | 2013 | | 467,353 |
| | 214,356 |
| | — |
| | 1,237,920 |
| | 7,650 |
| | 1,927,279 |
|
| Medical Officer | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Name and Principal Position | | Year
(1) | | | Salary
($)(2) | | | Bonus
($)(3) | | | Non-Equity
Incentive Plan
Compensation
($)(4) | | | Stock
Awards
($)(5) | | | Option
Awards
($)(6) | | | All Other
Compensation
($) | | | Total ($) | |
Michael M. Morrissey, Ph.D. | | | 2018 | | | | 945,080 | | | | — | | | | 1,000,000 | | | | 2,912,552 | | | | 3,111,403 | | | | 8,500 | | | | 7,977,535 | |
President and Chief | | | 2017 | | | | 891,781 | | | | 675,750 | | | | — | | | | 1,952,800 | | | | 5,911,152 | | | | 8,100 | | | | 9,439,583 | |
Executive Officer | | | 2016 | | | | 841,154 | | | | 800,000 | | | | — | | | | 918,600 | | | | 3,700,434 | | | | 7,950 | | | | 6,268,138 | |
Gisela M. Schwab, M.D. | | | 2018 | | | | 672,301 | | | | ��� | | | | 466,156 | | | | 1,221,398 | | | | 1,227,500 | | | | 8,500 | | | | 3,595,855 | |
President, Product Development | | | 2017 | | | | 629,493 | | | | 318,000 | | | | — | | | | 1,830,750 | | | | 1,847,235 | | | | 8,100 | | | | 4,633,578 | |
and Medical Affairs and Chief | | | 2016 | | | | 591,186 | | | | 375,000 | | | | — | | | | 294,800 | | | | 1,257,579 | | | | 8,511 | | | | 2,527,076 | |
Medical Officer | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Christopher J. Senner | | | 2018 | | | | 594,740 | | | | — | | | | 338,074 | | | | 1,221,398 | | | | 1,227,500 | | | | 8,500 | | | | 3,390,212 | |
Executive Vice President and | | | 2017 | | | | 562,120 | | | | 255,150 | | | | — | | | | 1,525,625 | | | | 1,539,363 | | | | 8,100 | | | | 3,890,358 | |
Chief Financial Officer | | | 2016 | | | | 532,923 | | | | 303,750 | | | | — | | | | 294,800 | | | | 1,062,084 | | | | 7,950 | | | | 2,201,507 | |
Jeffrey J. Hessekiel, J.D. | | | 2018 | | | | 531,880 | | | | — | | | | 301,836 | | | | 657,680 | | | | 1,227,500 | | | | 8,500 | | | | 2,727,396 | |
Executive Vice President and | | | 2017 | | | | 507,067 | | | | 229,970 | | | | — | | | | 1,220,500 | | | | 1,231,490 | | | | 7,076 | | | | 3,196,103 | |
General Counsel | | | 2016 | | | | 484,918 | | | | 275,084 | | | | — | | | | 239,525 | | | | 862,943 | | | | — | | | | 1,862,470 | |
Peter Lamb, Ph.D. | | | 2018 | | | | 493,570 | | | | — | | | | 311,202 | | | | 1,174,417 | | | | 1,180,295 | | | | 8,500 | | | | 3,167,984 | |
Executive Vice President, | | | 2017 | | | | 473,190 | | | | 214,245 | | | | — | | | | 1,342,550 | | | | 1,354,639 | | | | 8,100 | | | | 3,392,724 | |
Scientific Strategy and Chief | | | 2016 | | | | 453,546 | | | | 258,750 | | | | — | | | | 239,525 | | | | 967,207 | | | | 7,950 | | | | 1,926,978 | |
Scientific Officer | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| |
* | Mr. Senner joined Exelixis in July 2015. |
| |
** | Was not in our Summary Compensation Table prior to fiscal year 2014 and, according to applicable SEC rules, no information is provided for fiscal year 2013. |
| |
^ | Was our principal financial officer until Mr. Senner commenced employment with the company on July 15, 2015, at which point Ms. Burke assumed the position of Senior Vice President, Finance and Controller. |
| |
| (1) | The compensation reflected in the Summary Compensation Table reflects a52-week period for each of fiscal 2015, a 53-week period for fiscal 2014,2018, 2017 and a 52-week period for fiscal 2013.2016. |
| |
| (2) | The amount in this column represents the amount actually paid to each Named Executive Officer for fiscal 2015.2018. For information regarding 20152018 base salaries, please see “Compensation Discussion and Analysis-2015Analysis—2018 Compensation Decisions-2015Decisions—2018 Base Salaries.” |
| |
| (3) | The amount in this column represents discretionary cash bonuses awarded for services rendered during the indicated fiscal years by the Named Executive Officers (equity was issuedOfficers. |
| (4) | The amount in lieuthis column represents cash bonuses awarded under our Annual Cash Bonus Plan in fiscal 2018 based on the Compensation Committee’s assessment of cash for bonuses for 2015).the achievement ofpre-determined corporate goals. For a description of the company’s cash bonus program,Annual Cash Bonus Plan for fiscal 2018, see “Compensation Arrangements--AnnualDiscussion and Analysis—2018 Compensation Decisions—2018 Annual Cash Bonuses (Equity in Lieu of Cash)” following the Grants of Plan Based Awards table. The company does not maintain a “Non-Equity Incentive Plan” as defined in applicable SEC rules.Bonuses” above. |
| (5) | |
(4) | Amounts shown in this column reflect the aggregate grant date fair value in the indicated fiscal year for the RSU and PSU awards as computed in accordance with FASB ASC 718. The assumptions used to calculate the value of RSU and PSU awards are set forth in Note 107 of the Notes to Consolidated Financial Statements included in our Annual Report on Form10-K for the fiscal year ended January 1, 2016,December 28, 2018, filed with the SEC on February 29, 2016.22, 2019. With respect to PSU awards granted during fiscal 2018, the grant date fair values presented in the table assume achievement of the highest level of performance conditions and exclude estimates of forfeiture. |
| (6) | |
(5) | Amounts shown in this column do not reflect compensation actually received or amounts that may be realized in the future by the Named Executive Officers. The amounts shown reflect the aggregate grant date fair value in the indicated fiscal years for option awards as computed in accordance with FASB ASC 718. The assumptions used to calculate the value of option awards are set forth in Note 107 of the Notes to Consolidated Financial Statements included in our Annual Report on Form10-K for the fiscal year ended January 1, 2016,December 28, 2018, filed with the SEC on February 29, 2016.22, 2019. The grant |
2019 Proxy Statement 59
| date fair values presented in the table for the performance-based option awards assume achievement of the highest level of performance conditions and excludesexclude estimates of forfeiture. There can be no assurance that the stock option awards will ever be exercised (in which case no value will actually be realized by the executive) or that the value on exercise will be equal to the FASB ASC 718 value shown in this column. |
| |
(6) | The amounts in this column consist of (i) the value of matching contributions made by us under our tax-qualified 401(k) Retirement Plan, which provides for broad-based employee participation and (ii) a 10-year service award designed to recognize the extraordinary work and commitment of our employees to our company in the following amounts: Dr. Morrissey - $1,376, Ms. Burke - $530 and Dr. Lamb - $1,514. |
| |
(7) | Represents the aggregate grant date fair value for the RSU award issued to the Named Executive Officer in lieu of a cash bonus for 2015 as computed in accordance with ASC 718. The assumptions used to calculate the value of the RSU award is set forth in Note 10 of the Notes to Consolidated Financial Statements included in our Annual Report on Form 10-K for the fiscal year ended January 1, 2016, filed with the SEC on February 29, 2016. |
| |
(8) | Mr. Senner's base salary for 2015 was $500,000 per year. The amount shown represents the amount of salary he was actually paid in fiscal 2015, taking into account his start date in July 2015. |
| |
(9) | Ms. Burke’s salary from January 1, 2014, through February 28, 2014, was $273,114 annually. Her salary increased to $284,039 effective March 1, 2014, and to $315,000 effective September 21, 2014. The amount shown reflects the amount of salary actually paid to Ms. Burke during 2014. |
| |
(10) | Represents a one-time special cash bonus of $30,000 payable 50% upon Ms. Burke’s transition to the role of interim Chief Financial Officer and 50% upon Ms. Burke becoming the permanent Chief Financial Officer in September 2014. |
| |
(11) | Mr. Hessekiel’s base salary for 2014 was $450,000 per year. The amount shown represents the amount of salary he was actually paid in fiscal 2014, taking into account his start date in February 2014. |
Grants of Plan-Based Awards
The following table shows for the fiscal year ended January 1, 2016,December 28, 2018, certain information regarding grants of plan-based awards to the Named Executive Officers:
Grants of Plan-Based Awards in Fiscal 20152018
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | Estimated Future
Payouts Under
Non-Equity Incentive
Plan Awards (1) | | | Estimated Future
Payouts Under
Equity Incentive
Plan Awards | | | All other
option
awards:
Number of
Securities
Underlying
Options (#)(2) | | | | | | Grant Date
Fair Value of
Stock and
Option
Awards
($)(3) | |
| | Grant Date | | | Threshold
($) | | | Target
($) | | | Maximum
($) | | | Threshold
(#) | | | Target
(#) | | | Maximum
(#) | | | Exercise or
Base Price
of Option
Awards
($/Sh) | |
Michael M. Morrissey, Ph.D. | | | 09/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | 308,365 | (4) | | | 18.80 | | | | 3,111,403 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 12,394 | (5) | | | 24,788 | (5) | | | 24,788 | (5) | | | | | | | | | | | 466,015 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 18,590 | (6) | | | 37,181 | (6) | | | 37,181 | (6) | | | | | | | | | | | 699,003 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 11,619 | (7) | | | 23,238 | (7) | | | 23,238 | (7) | | | | | | | | | | | 436,874 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 11,619 | (8) | | | 23,238 | (8) | | | 23,238 | (8) | | | | | | | | | | | 436,874 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 23,239 | (9) | | | 46,478 | (9) | | | 46,478 | (9) | | | | | | | | | | | 873,786 | |
| | | | N/A | | | | — | | | | 955,060 | | | | 1,910,120 | | | | | | | | | | | | | | | | | | | | | | | | | |
Gisela M. Schwab, M.D. | | | 09/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | 129,314 | | | | 18.80 | | | | 1,227,500 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 5,197 | (5) | | | 10,395 | (5) | | | 10,395 | (5) | | | | | | | | | | | 195,426 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 7,796 | (6) | | | 15,592 | (6) | | | 15,592 | (6) | | | | | | | | | | | 293,129 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 4,872 | (7) | | | 9,745 | (7) | | | 9,745 | (7) | | | | | | | | | | | 183,206 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 4,872 | (8) | | | 9,745 | (8) | | | 9,745 | (8) | | | | | | | | | | | 183,206 | |
| | | 09/10/2018 | | �� | | | | | | | | | | | | | | 9,745 | (9) | | | 19,491 | (9) | | | 19,491 | (9) | | | | | | | | | | | 366,431 | |
| | | | N/A | | | | — | | | | 340,260 | | | | 680,520 | | | | | | | | | | | | | | | | | | | | | | | | | |
Christopher J. Senner | | | 09/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | 129,314 | | | | 18.80 | | | | 1,227,500 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 5,197 | (5) | | | 10,395 | (5) | | | 10,395 | (5) | | | | | | | | | | | 195,426 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 7,796 | (6) | | | 15,592 | (6) | | | 15,592 | (6) | | | | | | | | | | | 293,129 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 4,872 | (7) | | | 9,745 | (7) | | | 9,745 | (7) | | | | | | | | | | | 183,206 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 4,872 | (8) | | | 9,745 | (8) | | | 9,745 | (8) | | | | | | | | | | | 183,206 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 9,745 | (9) | | | 19,491 | (9) | | | 19,491 | (9) | | | | | | | | | | | 366,431 | |
| | | | N/A | | | | — | | | | 270,549 | | | | 540,918 | | | | | | | | | | | | | | | | | | | | | | | | | |
Jeffrey J. Hessekiel, J.D. | | | 09/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | 129,314 | | | | 18.80 | | | | 1,227,500 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 2,998 | (5) | | | 5,997 | (5) | | | 5,997 | (5) | | | | | | | | | | | 112,744 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 4,498 | (6) | | | 8,996 | (6) | | | 8,996 | (6) | | | | | | | | | | | 169,125 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 2,499 | (7) | | | 4,998 | (7) | | | 4,998 | (7) | | | | | | | | | | | 93,962 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 2,499 | (8) | | | 4,998 | (8) | | | 4,998 | (8) | | | | | | | | | | | 93,962 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 4,997 | (9) | | | 9,994 | (9) | | | 9,994 | (9) | | | | | | | | | | | 187,887 | |
| | | | N/A | | | | — | | | | 241,469 | | | | 482,937 | | | | | | | | | | | | | | | | | | | | | | | | | |
60 Exelixis, Inc.
|
| | | | | | | | | | | | | | | | | | | | | | | |
| | Grant Date | | Approval Date of Stock Options Grant(1) | | Estimated Future Payouts Under Equity Incentive Plan Awards (Sh)(2)
| | All other stock awards: Number of Number of Shares of Stock or Units (#)(3) | All other option awards: Number of Securities Underlying Options (#)(4) | | Exercise or Base Price of Option Awards ($/Sh) | | Grant Date Fair Value of Stock and Option Awards ($)(5) |
| | | Threshold | | Target | | Maximum | |
| Michael M. Morrissey, Ph.D. | 2/5/2015 | | | | 225,000 |
| | 450,000 |
| | 450,000 |
| | | | | 1.90 |
| | 595,800 |
|
| | 9/16/2015 | | | | | | | | | | | 500,000 |
| | 6.21 |
| | 2,097,950 |
|
| Christopher J. Senner | 7/15/2015 | | 6/30/2015 | | | | | | | | | 350,000 |
| | 3.66 |
| | 865,515 |
|
| | 7/15/2015 | | 6/30/2015 | | | | | | | | 100,000 |
| | | | | 366,000 |
|
| | 9/16/2015 | | | | | | | | | | | 225,000 |
| | 6.21 |
| | 944,078 |
|
| Deborah Burke | 2/5/2015 | | | | 87,500 |
| | 175,000 |
| | 175,000 |
| | | | | 1.90 |
| | 231,700 |
|
| | 9/16/2015 | | | | | | | | | | | 64,000 |
| | 6.21 |
| | 260,102 |
|
| Jeffrey J. Hessekiel, J.D. | 2/5/2015 | | | | 100,000 |
| | 200,000 |
| | 200,000 |
| | | | | 1.90 |
| | 264,800 |
|
| | 9/16/2015 | | | | | | | | | | | 190,000 |
| | 6.21 |
| | 797,221 |
|
| Peter Lamb, Ph.D. | 2/5/2015 | | | | 87,500 |
| | 175,000 |
| | 175,000 |
| | | | | 1.90 |
| | 231,700 |
|
| | 9/16/2015 | | | | | | | | | | | 190,000 |
| | 6.21 |
| | 797,221 |
|
| Gisela M. Schwab, M.D. | 2/5/2015 | | | | 125,000 |
| | 250,000 |
| | 250,000 |
| | | | | 1.90 |
| | 331,000 |
|
| | 9/16/2015 | | | | | | | | | | | 245,000 |
| | 6.21 |
| | 1,027,996 |
|
Compensation of Executive Officers | Grants of Plan-Based Awards
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | Estimated Future
Payouts Under
Non-Equity Incentive
Plan Awards (1) | | | Estimated Future
Payouts Under
Equity Incentive
Plan Awards | | | All other
option
awards:
Number of
Securities
Underlying
Options (#)(2) | | | | | | Grant Date
Fair Value of
Stock and
Option
Awards
($)(3) | |
| | Grant Date | | | Threshold
($) | | | Target
($) | | | Maximum
($) | | | Threshold
(#) | | | Target
(#) | | | Maximum
(#) | | | Exercise or
Base Price
of Option
Awards
($/Sh) | |
Peter Lamb, Ph.D. | | | 09/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | 124,341 | | | | 18.80 | | | | 1,180,295 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 4,997 | (5) | | | 9,995 | (5) | | | 9,995 | (5) | | | | | | | | | | | 187,906 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 7,496 | (6) | | | 14,993 | (6) | | | 14,993 | (6) | | | | | | | | | | | 281,868 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 4,685 | (7) | | | 9,370 | (7) | | | 9,370 | (7) | | | | | | | | | | | 176,156 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 4,685 | (8) | | | 9,370 | (8) | | | 9,370 | (8) | | | | | | | | | | | 176,156 | |
| | | 09/10/2018 | | | | | | | | | | | | | | | | 9,370 | (9) | | | 18,741 | (9) | | | 18,741 | (9) | | | | | | | | | | | 352,331 | |
| | | | N/A | | | | — | | | | 223,886 | | | | 447,773 | | | | | | | | | | | | | | | | | | | | | | | | | |
| |
| (1) | ReflectsThe dollar amount represents the date the Compensation Committee determined to make the grant, such grant to be effective on the grant date designated in the column to the left, at the fair market value on the grant date. The grant date was designated at the timetarget and maximum amounts of the Compensation Committee’s action. If no date appears in this column for a particular grant, the date of approval is the same as the date of grant, as reflected in the column to the left. |
| |
(2) | Options were granted as performance stock awardseach Named Executive Officer’s potential annual cash bonus award pursuant to Section 6(c)our Annual Cash Bonus Plan for the fiscal year ended December 28, 2018. The amounts shown under “Target” reflects the applicable target payment under the Annual Cash Bonus Plan if (i) of our 2014 Equity Plan. Vestingwe achieved 100% of the performance-based stock options is tied topre-determined corporate performance goals setestablished by the Compensation Committee for fiscal 2018, and (ii) as follows: (i) 50% of the options would vest ifapplicable, each Named Executive Officer’s individual performance percentage was assessed at 100% by the Compensation Committee determined that top-line efficacy data received fromwith respect to his or her contributions to the METEOR phase 3 pivotal trialachievement of cabozantinib in advanced RCC met its primary endpoint at a specified level, with such result to occur no later than a specified date; (ii) 25%these corporate performance goals. The amounts shown under “Maximum” reflects the applicable maximum payment under the Annual Cash Bonus Plan if (i) we achieved 200% of the options would vest ifpre-determined corporate performance goals established by the Compensation Committee confirmed that a new drug application for cabozantinib for the treatment of advanced RCC is accepted for reviewfiscal 2018, and (ii) as applicable, each Named Executive Officer’s individual performance percentage was assessed at 200% by the U.S. Food and Drug Administration, or FDA, by a specified date; and (iii) 25% of the options will vest if the Compensation Committee confirms that the FDA has approved cabozantinib for the treatment of advanced RCC by a specified date. On July 20, 2015, the Compensation Committee convened to determine that top-line efficacy data received from METEOR met its primary endpoint at the level specified and within the time period permitted by the performance goals, resulting in the vesting of 50% of the option. On March 7, 2016, the Compensation Committee convened to determine that the NDA for cabozantinib for the treatment of advanced RCC was accepted for review by the FDA within the time period permitted by the performance goals, resulting in the vesting of 25% of the option. If the Compensation Committee has not concluded that FDA has approved cabozantinib for the treatment of advanced RCC by the specified date, the performance stock award with respect to his or her contributions to the achievement of these corporate performance goals; provided, however, that neither the corporate performance goal will be forfeited. Each stock award expires seven years frompercentage nor the date of grant or earlier upon termination of service andindividual performance percentage may exceed 200% in any given year. Since the Annual Cash Bonus Plan is subject to vesting acceleration as describedentirely performance-based, the minimum possible payment under the caption “Potential Payments Upon Termination or Change-in-Control” below.Annual Cash Bonus Plan is zero. For more information regarding our Annual Cash Bonus Plan, please see “Compensation Discussion and Analysis—2018 Compensation Decisions—2018 Annual Cash Bonuses” above. |
| (2) | |
(3) | RSU award was granted pursuant to our 2014 Equity Plan. The RSU award will vest in full on the one year anniversary of the date of grant, provided that delivery may be delayed pursuant to the terms of the award agreement. Vesting is subject to acceleration as described under the caption “—Potential Payments Upon Termination or Change-in-Control” below. |
| |
(4) | The option award was granted pursuant to our 2014 Equity2017 Plan and expires seven years from the date of grant or earlier upon termination of service. The option will vest as to 1/4th4th of the original number of shares subject to the option on theone-year anniversary of the grant date and will continue to vest thereafter as to 1/48th48th of the original number of shares subject to the option on each monthly anniversary of the grant date. Vesting is subject to acceleration as described under the caption “—Potential Payments Upon Termination or Change-in-Control”Change in Control” below. |
| (3) | |
(5) | Amounts shown in this column do not reflect compensation actually received or amounts that may be realized in the future by the Named Executive Officers. The amounts shown in this column reflect the aggregate grant date fair value in fiscal year 20152018 for the option award or the target PSU award as computed in accordance with FASB ASC 718, excluding estimates of forfeiture.718. The assumptions used to calculate the value of the option awardawards and the PSU awards are set forth in Note 107 of the Notes to Consolidated Financial Statements included in our Annual Report on Form10-K for the fiscal year ended January 1, 2016,December 28, 2018, filed with the SEC on February 29, 2016.22, 2019. There can be no assurance that the stock option award will ever be exercised (in which case no value will actually be realized by the executive) or that the value on exercise will be equal to the FASB ASC 718 value shown in this column. |
| (4) | The option award was granted pursuant to our 2017 Plan and expires seven years from the date of grant or earlier upon termination of service. The option will vest as to 1/4th of the original number of shares subject to the option on theone-year anniversary of the grant date and will continue to vest thereafter as to 1/48th of the original number of shares subject to the option on each monthly anniversary of the grant date. However, the option will not be exercisable until the closing market price of our common stock is equal to or greater than 125% of the exercise price of the option over a period of at least 30 consecutive calendar days. Vesting is subject to acceleration as described under the caption “—Potential Payments Upon Termination or Change in Control” below. |
| (5) | The PSU award was granted pursuant to our 2017 Plan. The PSU award will vest as to (i) 50% of the original number of shares subject to the award upon the Compensation Committee’s certification that Exelixis has achieved a quarterly net product revenue target, and (ii) 50% of the original number of shares subject to the award on the first quarterly vesting date (i.e. February 15th, May 15th, August 15th and November 15th) following theone-year anniversary of the Compensation Committee’s certification. Failure to achieve the aforementioned performance target by December 31, 2021 will result in forfeiture of 100% of the PSU award, and vesting is subject to acceleration as described under the caption “—Potential Payments Upon Termination or Change in Control” below. |
| (6) | The PSU award was granted pursuant to our 2017 Plan. The PSU award will vest as to (i) 50% of the original number of shares subject to the award upon the Compensation Committee’s certification that Exelixis has achieved a net product revenue target over four consecutive fiscal quarters, and (ii) 50% of the original number of shares subject to the award on the first quarterly vesting date (i.e. February 15th, May 15th, August 15th and November 15th) following theone-year anniversary of the Compensation Committee’s certification. Failure to achieve the aforementioned performance target by December 31, 2021 will result in forfeiture of 100% of the PSU award, and vesting is subject to acceleration as described under the caption “—Potential Payments Upon Termination or Change in Control” below. |
| (7) | The PSU award was granted pursuant to our 2017 Plan. The PSU award will vest as to (i) 50% of the original number of shares subject to the award upon the Compensation Committee’s certification that Exelixis has initiated a target number of potentially label-enabling clinical trials for cabozantinib (both alone or in combination with another therapy), and (ii) 50% of the original number of shares subject to the award on the first quarterly vesting date (i.e. February 15th, May 15th, August 15th and November 15th) following theone-year anniversary of the Compensation Committee’s certification. Failure to achieve the aforementioned performance target by December 31, 2021 will result in forfeiture of 100% of the PSU award, and vesting is subject to acceleration as described under the caption “—Potential Payments Upon Termination or Change in Control” below. |
2019 Proxy Statement 61
| (8) | The PSU award was granted pursuant to our 2017 Plan. The PSU award will vest as to (i) 50% of the original number of shares subject to the award upon the Compensation Committee’s certification that Exelixis has achieved either (x) full target enrollment, as specified in the applicable clinical trial protocol, or (y) positivetop-line results, in each case with respect to a target number of potentially label-enabling clinical trials for cabozantinib (both alone or in combination with another therapy), and (ii) 50% of the original number of shares subject to the award on the first quarterly vesting date (i.e. February 15th, May 15th, August 15th and November 15th) following theone-year anniversary of the Compensation Committee’s certification. Failure to achieve the aforementioned performance target by December 31, 2021 will result in forfeiture of 100% of the PSU award, and vesting is subject to acceleration as described under the caption “—Potential Payments Upon Termination or Change in Control” below. |
| (9) | The PSU award was granted pursuant to our 2017 Plan. The PSU award will vest as to (i) 50% of the original number of shares subject to the award upon the Compensation Committee’s certification that a target number of IND applications or equivalent filings with foreign governmental authorities have been approved or otherwise become effective with respect to product candidates that have either (x) been discovered by Exelixis or(y) in-licensed or acquired by Exelixis, and (ii) 50% of the original number of shares subject to the award on the first quarterly vesting date (i.e. February 15th, May 15th, August 15th and November 15th) following theone-year anniversary of the Compensation Committee’s certification. Failure to achieve the aforementioned performance target by December 31, 2021 will result in forfeiture of 100% of the PSU award, and vesting is subject to acceleration as described under the caption “—Potential Payments Upon Termination or Change in Control” below. |
Compensation Arrangements
Base Salaries. For a description of actions taken by the Compensation Committee with respect to base salaries for our Named Executive Officers for fiscal 2015,2018, please see “Compensation Discussion and Analysis-2015Analysis—2018 Compensation Decisions-2015Decisions—2018 Base Salaries” above.
Annual Cash Bonuses (Equity In Lieu of Cash). Each yearBonuses. Prior to 2018 the Compensation Committee considersconsidered and approved the payment of annual cash bonuses to the Named Executive Officers for services rendered in the pastprior year. Whether or not a bonus is paid for any year is solelyHistorically, the payment of bonuses had been entirely within the discretion of the Board or Compensation Committee upon delegation by the Board.Committee. While the Compensation Committee hashad established general guidelines related to bonus target amounts, and the portionpayment of actual cash bonuses was generally aligned with the achievement of our corporate goals and the Compensation Committee’s subjective determination of each Named Executive Officer’s annual cash bonus that is tied to company-wide, department or personal performance components,contribution toward the achievement of such goals, the Compensation Committee exercisesexercised broad discretion in determining the amount of cash bonuses and doesdid not attempt to quantify the level of achievement of corporate goals or the extent to which each Named Executive Officer or his or her department contributed to Exelixis’ overall success. Accordingly, we dodid not consider thesethe bonuses reported in the Summary Compensation Table in fiscal 2017 and fiscal 2016 tobe “Non-Equity Incentive Plan Compensation” within the meaning of applicable SEC rules. In 2018, we adopted our Annual Cash Bonus Plan that provides for annual bonus awards to reward executive officers based on our achievement ofpre-determined
corporate performance goals and their contribution to the that achievement, which we consider to be“Non-Equity Incentive Plan Compensation” within the meaning of applicable SEC rules. For additionalmore information with respect to annual bonuses forregarding our Named Executive Officers for fiscal 2015,Annual Cash Bonus Plan, please see “Compensation Discussion and Analysis-2015Analysis—2018 Compensation Decisions-2015Decisions—2018 Annual Cash Bonuses (Equity in Lieu of Cash)”Bonuses” above.
Stock Awards and Option Awards. Our 2014 Equity2017 Plan provides for the grant of RSUs, PSUs and compensatory stock options to our Named Executive Officers and other employees. In 2015,September 2018, we granted both performance-based and time-based stock options to all of our Named Executive Officers to keep(other than the Chief Executive Officer), and we also granted PSU awards, which will only vest following our management team focused onachievement of certain shorter-term objectives that we believe will furtherperformance targets in line with our longer-term business strategy.strategy, to all of our Named Executive Officers. In 2015,September 2018, we also granted a time-based RSU awardtime- and performance-based stock option to Mr. Senner in connection with his joiningour Chief Executive Officer, which will not be exercisable until the company in July 2015.closing market price of our common stock is equal to or greater than 125% of the exercise price of the option over a period of at least 30 consecutive calendar days.
For information regarding stock option grantsand PSU awards granted to the Named Executive Officers in fiscal 2015,2018, including the number of options granted, theunderlying shares and any exercise price and vesting conditions related thereto, please see “Compensation Discussion and Analysis-2015Analysis—2018 Compensation Decisions-2015 EquityDecisions—2018 Long-Term Incentive Awards” above. As a general matter, the vested portion of options granted to our Named Executive Officers will expire three months after each Named Executive Officer’s termination of continuous service, subject to extension in certain termination situations or events that can accelerate the vesting, as described under “Potential Payments Upon Termination or Change-in-Control”Change in Control” below.
Employment Agreements.We have no employment agreements with our Named Executive Officers.
Change in Control and Severance Benefit Plan. Each of our Named Executive Officers participates in our Change in Control and Severance Benefit Plan, a description of which is included below under the heading “Potential Payments Upon Termination or Change-in-Control.Change in Control.”
Other Compensatory Arrangements. Please see “Compensation Discussion and Analysis-Elements of Compensation-Other Benefits”Analysis—Compensation Elements—Other Compensation” above for a description of other executive compensatory arrangements, including our 401(k) Retirement Plan and other benefits.
62 Exelixis, Inc.
Compensation of Executive Officers |Outstanding Equity Awards at Fiscal Year–End
Outstanding Equity Awards at Fiscal Year–End
The following table shows certain information regarding outstanding equity awards at January 1, 2016,December 28, 2018, for the Named Executive Officers.
Outstanding Equity Awards at January 1, 2016December 28, 2018
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | Option Awards(1) | | | Stock Awards(2) | |
Name | | Grant Date | | | Number of
Securities
Underlying
Unexercised
Options
(#) Exercisable | | | Number of
Securities
Underlying
Unexercised
Options
(#) Unexercisable | | | Equity
Incentive
Plan Awards:
Number of
Securities
Underlying
Unexercised
Unearned
Options
(#) | | | Option
Exercise
Price ($) | | | Option
Expiration Date | | | Number of
Shares or
Units of
Stock That
Have Not
Vested (#) | | | Market
Value of
Shares
or Units
of Stock
That
Have Not
Vested
($)(3) | | | Equity Incentive Plan Awards:
Number of
Unearned Shares,
Units or
Other Rights That
Have Not
Vested (#) | | | Equity Incentive
Plan Awards:
Market or
Payout Value of
Unearned
Shares, Units or
Other Rights
That Have Not
Vested ($)(3) | |
Michael M. Morrissey, Ph.D. | | | 12/9/2009 | | | | 300,000 | | | | | | | | | | | | 7.18 | | | | 12/8/2019 | | | | | | | | | | | | | | | | | |
| | | 9/21/2012 | | | | 402,000 | | | | | | | | | | | | 5.555 | | | | 9/20/2019 | | | | | | | | | | | | | | | | | |
| | | 9/18/2013 | | | | 960,000 | | | | | | | | | | | | 5.51 | | | | 9/17/2020 | | | | | | | | | | | | | | | | | |
| | | 9/19/2014 | | | | 570,284 | | | | | | | | | | | | 1.70 | | | | 9/18/2021 | | | | | | | | | | | | | | | | | |
| | | 9/16/2015 | | | | 406,250 | | | | 93,750 | (5) | | | | | | | 6.21 | | | | 9/15/2022 | | | | | | | | | | | | | | | | | |
| | | 2/11/2016 | | | | 106,250 | | | | 43,750 | (6) | | | | | | | 4.20 | | | | 2/10/2023 | | | | | | | | | | | | | | | | | |
| | | 9/26/2016 | | | | 202,500 | | | | 157,500 | (8) | | | | | | | 15.31 | | | | 9/25/2023 | | | | | | | | | | | | | | | | | |
| | | 9/26/2016 | | | | | | | | | | | | | | | | | | | | | | | | 30,000 | (9) | | | 583,200 | | | | | | | | | |
| | | 10/3/2017 | | | | 140,000 | | | | 340,000 | (10) | | | | | | | 24.41 | | | | 10/2/2024 | | | | | | | | | | | | | | | | | |
| | | 10/3/2017 | | | | | | | | | | | | | | | | | | | | | | | | 60,000 | (11) | | | 1,166,400 | | | | | | | | | |
| | | 9/10/2018 | | | | | | | | | | | | 308,365(12) | | | | 18.80 | | | | 9/9/2025 | | | | | | | | | | | | | | | | | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 24,788 | (13) | | | 481,879 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 37,181 | (14) | | | 722,798 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 23,238 | (15) | | | 451,747 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 23,238 | (16) | | | 451,747 | |
| | | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 46,478 | (17) | | | 903,532 | |
Gisela M. Schwab, M.D. | | | 12/9/2009 | | | | 163,109 | | | | | | | | | | | | 7.18 | | | | 12/8/2019 | | | | | | | | | | | | | | | | | |
| | | 9/21/2012 | | | | 123,000 | | | | | | | | | | | | 5.555 | | | | 9/20/2019 | | | | | | | | | | | | | | | | | |
| | | 9/18/2013 | | | | 270,000 | | | | | | | | | | | | 5.51 | | | | 9/17/2020 | | | | | | | | | | | | | | | | | |
| | | 9/19/2014 | | | | 500,000 | | | | | | | | | | | | 1.70 | | | | 9/18/2021 | | | | | | | | | | | | | | | | | |
| | | 2/5/2015 | | | | 250,000 | | | | | | | | | | | | 1.90 | | | | 2/4/2022 | | | | | | | | | | | | | | | | | |
| | | 9/16/2015 | | | | 199,062 | | | | 45,938 | (5) | | | | | | | 6.21 | | | | 9/15/2022 | | | | | | | | | | | | | | | | | |
| | | 2/11/2016 | | | | 53,125 | | | | 21,875 | (6) | | | | | | | 4.20 | | | | 2/10/2023 | | | | | | | | | | | | | | | | | |
| | | 9/22/2016 | | | | 67,500 | | | | 52,500 | (7) | | | | | | | 14.74 | | | | 9/21/2023 | | | | | | | | | | | | | | | | | |
| | | 9/22/2016 | | | | | | | | | | | | | | | | | | | | | | | | 10,000 | (9) | | | 194,400 | | | | | | | | | |
| | | 10/3/2017 | | | | 43,750 | | | | 106,250 | (10) | | | | | | | 24.41 | | | | 10/2/2024 | | | | | | | | | | | | | | | | | |
| | | 10/3/2017 | | | | | | | | | | | | | | | | | | | | | | | | 56,250 | (11) | | | 1,093,500 | | | | | | | | | |
| | | 9/10/2018 | | | | | | | | 129,314 | (18) | | | | | | | 18.80 | | | | 9/9/2025 | | | | | | | | | | | | | | | | | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 10,395 | (13) | | | 202,079 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 15,592 | (14) | | | 303,108 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 9,745 | (15) | | | 189,443 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 9,745 | (16) | | | 189,443 | |
| | | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 19,491 | (17) | | | 378,905 | |
2019 Proxy Statement 63
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | Option Awards(1) | | | Stock Awards(2) | |
Name | | Grant Date | | | Number of
Securities
Underlying
Unexercised
Options
(#) Exercisable | | | Number of
Securities
Underlying
Unexercised
Options
(#) Unexercisable | | | Equity
Incentive
Plan
Awards:
Number of
Securities
Underlying
Unexercised
Unearned
Options
(#) | | | Option
Exercise
Price ($) | | | Option
Expiration Date | | | Number of
Shares or
Units of
Stock That
Have Not
Vested (#) | | | Market
Value of
Shares
or Units
of Stock
That
Have Not
Vested
($)(3) | | | Equity Incentive Plan Awards:
Number of
Unearned Shares,
Units or
Other Rights That
Have Not
Vested (#) | | | Equity Incentive
Plan Awards:
Market or
Payout Value of
Unearned
Shares, Units or
Other Rights
That Have Not
Vested ($)(3) | |
Christopher J. Senner | | | 7/15/2015 | | | | 198,958 | | | | 51,042 | (4) | | | | | | | 3.66 | | | | 7/14/2022 | | | | | | | | | | | | | | | | | |
| | | 9/16/2015 | | | | 182,812 | | | | 42,188 | (5) | | | | | | | 6.21 | | | | 9/15/2022 | | | | | | | | | | | | | | | | | |
| | | 9/22/2016 | | | | 67,500 | | | | 52,500 | (7) | | | | | | | 14.74 | | | | 9/21/2023 | | | | | | | | | | | | | | | | | |
| | | 9/22/2016 | | | | | | | | | | | | | | | | | | | | | | | | 10,000 | (9) | | | 194,400 | | | | | | | | | |
| | | 10/3/2017 | | | | 36,458 | | | | 88,542 | (10) | | | | | | | 24.41 | | | | 10/2/2024 | | | | | | | | | | | | | | | | | |
| | | 10/3/2017 | | | | | | | | | | | | | | | | | | | | | | | | 46,875 | (11) | | | 911,250 | | | | | | | | | |
| | | 9/10/2018 | | | | | | | | 129,314 | (18) | | | | | | | 18.80 | | | | 9/9/2025 | | | | | | | | | | | | | | | | | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 10,395(13) | | | | 202,079 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 15,592(14) | | | | 303,108 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 9,745(15) | | | | 189,443 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 9,745(16) | | | | 189,443 | |
| | | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 19,491(17) | | | | 378,905 | |
Jeffrey J. Hessekiel, J.D. | | | 2/10/2014 | | | | 130,000 | | | | | | | | | | | | 7.27 | | | | 2/9/2021 | | | | | | | | | | | | | | | | | |
| | | 9/19/2014 | | | | 120,000 | | | | | | | | | | | | 1.70 | | | | 9/18/2021 | | | | | | | | | | | | | | | | | |
| | | 2/5/2015 | | | | 172,698 | | | | | | | | | | | | 1.90 | | | | 2/4/2022 | | | | | | | | | | | | | | | | | |
| | | 9/16/2015 | | | | 154,375 | | | | 35,625 | (5) | | | | | | | 6.21 | | | | 9/15/2022 | | | | | | | | | | | | | | | | | |
| | | 9/22/2016 | | | | 54,843 | | | | 42,657 | (7) | | | | | | | 14.74 | | | | 9/21/2023 | | | | | | | | | | | | | | | | | |
| | | 9/22/2016 | | | | | | | | | | | | | | | | | | | | | | | | 8,124 | (9) | | | 157,931 | | | | | | | | | |
| | | 10/3/2017 | | | | 29,166 | | | | 70,834 | (10) | | | | | | | 24.41 | | | | 10/2/2024 | | | | | | | | | | | | | | | | | |
| | | 10/3/2017 | | | | | | | | | | | | | | | | | | | | | | | | 37,500 | (11) | | | 729,000 | | | | | | | | | |
| | | 9/10/2018 | | | | | | | | 129,314 | (18) | | | | | | | 18.80 | | | | 9/9/2025 | | | | | | | | | | | | | | | | | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 5,997(13) | | | | 116,582 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 8,996(14) | | | | 174,882 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 4,998(15) | | | | 97,161 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 4,998(16) | | | | 97,161 | |
| | | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 9,994(17) | | | | 194,283 | |
Peter Lamb, Ph.D. | | | 9/21/2012 | | | | 123,000 | | | | | | | | | | | | 5.555 | | | | 9/20/2019 | | | | | | | | | | | | | | | | | |
| | | 9/18/2013 | | | | 168,000 | | | | | | | | | | | | 5.51 | | | | 9/17/2020 | | | | | | | | | | | | | | | | | |
| | | 9/19/2014 | | | | 400,000 | | | | | | | | | | | | 1.70 | | | | 9/18/2021 | | | | | | | | | | | | | | | | | |
| | | 2/5/2015 | | | | 175,000 | | | | | | | | | | | | 1.90 | | | | 2/4/2022 | | | | | | | | | | | | | | | | | |
| | | 9/16/2015 | | | | 154,375 | | | | 35,625 | (5) | | | | | | | 6.21 | | | | 9/15/2022 | | | | | | | | | | | | | | | | | |
| | | 2/11/2016 | | | | 28,333 | | | | 11,667 | (6) | | | | | | | 4.20 | | | | 2/10/2023 | | | | | | | | | | | | | | | | | |
| | | 9/22/2016 | | | | 54,843 | | | | 42,657 | (7) | | | | | | | 14.74 | | | | 9/21/2023 | | | | | | | | | | | | | | | | | |
| | | 9/22/2016 | | | | | | | | | | | | | | | | | | | | | | | | 8,124 | (9) | | | 157,931 | | | | | | | | | |
| | | 10/3/2017 | | | | 32,083 | | | | 77,917 | (10) | | | | | | | 24.41 | | | | 10/2/2014 | | | | | | | | | | | | | | | | | |
| | | 10/3/2017 | | | | | | | | | | | | | | | | | | | | | | | | 41,250 | (11) | | | 801,900 | | | | | | | | | |
| | | 9/10/2018 | | | | | | | | 124,341 | (18) | | | | | | | 18.80 | | | | 9/9/2025 | | | | | | | | | | | | | | | | | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 9,995(13) | | | | 194,302 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 14,993(14) | | | | 291,464 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 9,370(15) | | | | 182,153 | |
| | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 9,370(16) | | | | 182,153 | |
| | | | 9/10/2018 | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 18,741(17) | | | | 364,325 | |
64 Exelixis, Inc.
|
| | | | | | | | | | | | | | | | | | | | | | |
| | | | | Option Awards | | Stock Awards |
| | | Grant Date | | Number of Securities Underlying Unexercised Options (#)(1) | | Number of Securities Underlying Unexercised Options (#)(1) | | Equity Incentive Plan Awards: Number of Securities Underlying Unexercised Unearned Options (#) | | Option Exercise Price ($) | | Option Expiration Date | | Number of Shares or Units of Stock That Have Not Vested (#)(2) | | Market Value of Shares or Units of Stock That Have Not Vested ($)(3) |
| Name | | | Exercisable | | Unexercisable | | | | | |
| Michael M. Morrissey, Ph.D. | | 12/8/2006 | | 200,000 |
| | | | | | 8.99 |
| | 12/7/2016 | | | | |
| | | 12/6/2007 | | 200,000 |
| | | | | | 9.91 |
| | 12/5/2017 | | | | |
| | | 12/16/2008 | | 50,000 |
| | | | | | 5.04 |
| | 12/15/2018 | | | | |
| | | 2/26/2009 | | 25,000 |
| | | | | | 4.42 |
| | 2/25/2019 | | | | |
| | | 12/9/2009 | | 300,000 |
| | | | | | 7.18 |
| | 12/8/2019 | | | | |
| | | 9/28/2011 | | 450,000 |
| | | | | | 5.50 |
| | 9/27/2018 | | | | |
| | | 9/21/2012 | | 326,625 |
| | 75,375 |
| (4) | | | 5.555 |
| | 9/20/2019 | | | | |
| | | 9/18/2013 | | 405,000 |
| | 315,000 |
| (5) | | | 5.51 |
| | 9/17/2020 | | | | |
| | | 9/18/2013 | | 240,000 |
| | | | | | 5.51 |
| | 9/17/2020 | | | | |
| | | 9/19/2014 | | 562,500 |
| | | | 562,500 |
| (6) | 1.70 |
| | 9/18/2021 | | | | |
| | | 2/5/2015 | | 225,000 |
| | | | 225,000 |
| (6) | 1.90 |
| | 2/4/2022 | | | | |
| | | 9/16/2015 | | | | 500,000 |
| (7) | | | 6.21 |
| | 9/15/2022 | | | | |
| Christopher J. Senner | | 7/15/2015 | | | | 350,000 |
| (8) | | | 3.66 |
| | 7/14/2022 | | | | |
| | | 7/15/2015 | | | |
|
| | | | | | | | 100,000 |
| (9) | 564,000 |
|
| | | 9/16/2015 | | | | 225,000 |
| (7) | | | 6.21 |
| | 9/15/2022 | | | | |
| Deborah Burke | | 12/16/2008 | | 9,000 |
| | | | | | 5.04 |
| | 12/15/2018 | | | | |
| | | 2/26/2009 | | 12,500 |
| | | | | | 4.42 |
| | 2/25/2019 | | | | |
| | | 9/28/2011 | | 39,188 |
| | | | | | 5.50 |
| | 9/27/2018 | | | | |
| | | 9/21/2012 | | 20,475 |
| | 4,725 |
| (4) | | | 5.555 |
| | 9/20/2019 | | | | |
| | | 9/21/2012 | | | | | | | | | | | | 2,100 |
| (10) | 11,844 |
|
| | | 9/18/2013 | | 21,937 |
| | 17,063 |
| (5) | | | 5.51 |
| | 9/17/2020 | | | | |
| | | 9/18/2013 | | 26,000 |
| | | | | | 5.51 |
| | 9/17/2020 | | | | |
| | | 9/18/2013 | | | | | | | | | | | | 6,500 |
| (11) | 36,660 |
|
| | | 9/19/2014 | | 140,625 |
| | | | 187,500 |
| (6) | 1.70 |
| | 9/18/2021 | | | | |
| | | 2/5/2015 | | 87,500 |
| | | | 87,500 |
| (6) | 1.90 |
| | 2/4/2022 | | | | |
| | | 9/16/2015 | | | | 64,000 |
| (7) | | | 6.21 |
| | 9/15/2022 | | | | |
| Jeffrey J. Hessekiel, J.D. | | 2/10/2014 | | 105,416 |
| | 124,584 |
| (12) | | | 7.27 |
| | 2/9/2021 | | | | |
| | | 9/19/2014 | | 200,000 |
| | | | 200,000 |
| (6) | 1.70 |
| | 9/18/2021 | | | | |
| | | 2/5/2015 | | 100,000 |
| | | | 100,000 |
| (6) | 1.90 |
| | 2/4/2022 | | | | |
| | | 9/16/2015 | | | | 190,000 |
| (7) | | | 6.21 |
| | 9/15/2022 | | | | |
|
| | | | | | | | | | | | | | | | | | | | |
| | | | | Option Awards | | Stock Awards |
| | | Grant Date | | Number of Securities Underlying Unexercised Options (#)(1) | | Number of Securities Underlying Unexercised Options (#)(1) | | Equity Incentive Plan Awards : Number of Securities Underlying Unexercised Unearned Options (#) | | Option Exercise Price ($) | | Option Expiration Date | | Number of Shares or Units of Stock That Have Not Vested (#)(2) | | Market Value of Shares or Units of Stock That Have Not Vested ($)(3) |
| Name | | | Exercisable | | Unexercisable | | | | | |
| Peter Lamb, Ph.D. | | 12/16/2008 | | 50,000 |
| | | | | | 5.04 |
| | 12/15/2018 | | | | |
| | | 2/26/2009 | | 25,000 |
| | | | | | 4.42 |
| | 2/25/2019 | | | | |
| | | 9/1/2009 | | 75,000 |
| | | | | | 5.96 |
| | 8/31/2019 | | | | |
| | | 12/9/2009 | | 75,000 |
| | | | | | 7.18 |
| | 12/8/2019 | | | | |
| | | 12/15/2009 | | 25,000 |
| | | | | | 7.51 |
| | 12/14/2019 | | | | |
| | | 9/28/2011 | | 77,000 |
| | | | | | 5.50 |
| | 9/28/2018 | | | | |
| | | 9/21/2012 | | 99,937 |
| | 23,063 |
| (4) | | | 5.555 |
| | 9/20/2019 | | | | |
| | | 9/18/2013 | | 70,875 |
| | 55,125 |
| (5) | | | 5.51 |
| | 9/17/2020 | | | | |
| | | 9/18/2013 | | 42,000 |
| | | | | | 5.51 |
| | 9/17/2020 | | | | |
| | | 9/19/2014 | | 200,000 |
| | | | 200,000 |
| (6) | 1.70 |
| | 9/18/2021 | | | | |
| | | 2/5/2015 | | 87,500 |
| | | | 87,500 |
| (6) | 1.90 |
| | 2/4/2022 | | | | |
| | | 9/16/2015 | | | | 190,000 |
| (7) | | | 6.21 |
| | 9/15/2022 | | | | |
| Gisela M. Schwab, M.D. | | 9/1/2006 | | 175,000 |
| | | | | | 9.73 |
| | 8/31/2016 | | | | |
| | | 12/8/2006 | | 44,000 |
| | | | | | 8.99 |
| | 12/7/2016 | | | | |
| | | 12/6/2007 | | 200,000 |
| | | | | | 9.91 |
| | 12/5/2017 | | | | |
| | | 12/16/2008 | | 50,000 |
| | | | | | 5.04 |
| | 12/15/2018 | | | | |
| | | 2/26/2009 | | 25,000 |
| | | | | | 4.42 |
| | 2/25/2019 | | | | |
| | | 12/9/2009 | | 210,000 |
| | | | | | 7.18 |
| | 12/8/2019 | | | | |
| | | 9/28/2011 | | 112,500 |
| | | | | | 5.50 |
| | 9/27/2018 | | | | |
| | | 9/21/2012 | | 99,937 |
| | 23,063 |
| (4) | | | 5.555 |
| | 9/20/2019 | | | | |
| | | 9/18/2013 | | 135,000 |
| | 105,000 |
| (5) | | | 5.51 |
| | 9/17/2020 | | | | |
| | | 9/18/2013 | | 80,000 |
| | | | | | 5.51 |
| | 9/17/2020 | | | | |
| | | 9/19/2014 | | 250,000 |
| |
|
| | 250,000 |
| (6) | 1.70 |
| | 9/18/2021 | | | | |
| | | 2/5/2015 | | 125,000 |
| | | | 125,000 |
| (6) | 1.90 |
| | 2/4/2022 | | | | |
| | | 9/16/2015 | | | | 245,000 |
| (7) | | | 6.21 |
| | 9/15/2022 | | | | |
Compensation of Executive Officers | Outstanding Equity Awards at Fiscal Year–End
| |
| (1) | Option awards granted prior to January 26, 2010 were issued under our 2000 Equity Incentive Plan (2000 Plan) and have vested in full and expire ten years from the date of grant or earlier upon termination of continuous service. There were no option awards granted to Named Executive Officers betweenon or after January 26, 2010 and prior to May 18, 2011. Option awards granted betweenon or after May 18, 2011, and prior to May 28, 2014, were issued under our 2011 Equity Incentive Plan or 2011 Equity Plan,(2011 Plan) and are either subject to time-based vesting or were subject to performance-based vesting and expire seven years from the date of grant or earlier upon termination of continuous service. Vesting of awards granted under the 2011 Equity Plan still subject to vesting is set forth in the applicable footnote accompanying the entry. All option awards granted pursuant to our 2011 Equity Plan subject to time-based vesting have vested in part, and the remaining unvested portion will vest as to 1/48th48th of the original number of shares subject to the option on each monthly anniversary of the vesting commencement date. All option awards granted under our 2011 Equity Plan subject to performance-based vesting have either been cancelled or are vested in full. Option awards granted on or after May 28, 2014, and prior to May 24, 2017, were issued under our 2014 Equity Incentive Plan (2014 Plan) and are either subject to time-based vesting or were subject to performance-based vesting and and expire seven years from the date of grant or earlier upon termination of continuous service. Vesting of awards granted under the 2014 Equity Plan is set forth in the applicable footnote accompanying the entry. Option awards granted pursuant to our 2014 Equity Plan subject to time-based vesting vest as to 1/4th of the original number of shares subject to the option on theone-year anniversary of the vesting commencement date and thereafter as to 1/48th of the original number of shares subject to the option on each monthly anniversary of the vesting commencement date. Option awards granted pursuant to our 2014 Equity Plan subject to performance-based vesting are vested in full. Option awards granted on or after May 24, 2017 were issued under our 2017 Plan and are subject to time-based vesting and expire seven years from the date of grant or earlier upon termination of continuous service. Vesting of awards granted under the 2017 Plan is set forth in the applicable footnote accompanying the entry. Option awards granted pursuant to our 2017 Plan subject to time-based vesting vest as to 1/4th of the original number of shares subject to the option on theone-year anniversary of the vesting commencement date and thereafter as to 1/48th of the original number of shares subject to the option on each monthly anniversary of the vesting commencement date. One option award granted pursuant to our 2017 Plan subject to performance-based vesting, in full, in part oraddition to time-based vesting, will not at all based onbe exercisable until the achievement of certain goalsthe stock price target set by the Compensation Committee, as described below in Footnote 6.12. Vesting of all options issued to our Named Executive Officers are subject to acceleration as described under the caption “Potential Payments Upon Termination or Change-in-Control”Change in Control” below. |
| |
| (2) | RSU awards granted prior to May 28, 2014 were issued under our 2011 EquityPlan, RSU awards granted on or after May 28, 2014 and prior to May 24, 2017 were issued under our 2014 Plan, and RSU awards granted on or after May 28, 2014,24, 2017 were issued under our 2014 Equity2017 Plan. Except as described in Footnote 9, RSU awards generally vest as to 1/4th of the original number of shares subject to the RSU award on the first established RSU vesting date following the one year anniversary of the grant date and thereafter as to 1/4th of the original number of shares subject to the RSU award on each anniversary of the first established RSU vesting date following the one year anniversary of the grant date, until fully-vested.fully vested. We have established February 15th, May15May 15th, August 15th and November 15th as restricted stock unitquarterly vesting dates. Vested shares will be delivered to the Named Executive Officer on the applicable quarterly vesting date, provided that delivery may be delayed pursuant to the terms of the award agreement. In addition, PSU awards granted pursuant to our 2017 Plan vest based on the achievement of certain performance targets set by the Compensation Committee as described below in Footnotes 13, 14, 15, 16 and 17. Vesting of all RSU awards and PSU awards issued to our Named Executive Officers is subject to acceleration as described under the caption “Potential Payments Upon Termination or Change-in-Control”Change in Control” below. |
| |
| (3) | For purposes of determining market value, we assumed a stock price of $5.64,$19.44, the closing sale price per share of our common stock on December 31, 2015,28, 2018, the last business day of our last fiscal year. |
| |
| (4) | Options vestOption vests as to 1/48th of the original number of shares subject to the option on each monthly anniversary of the vesting commencementgrant date with a final vesting date of September 21, 2016July 15, 2019 (assuming that such options are not accelerated).
|
| |
| (5) | Options vest as to to 1/48th of the original number of shares subject to the option on each monthly anniversary of the vesting commencement date with a final vesting date of September 18, 2017 (assuming that such options are not accelerated).
|
| |
(6) | Options were granted as performance stock awards pursuant to Section 6(c)(i) of our 2014 Equity Plan. Vesting of the performance-based stock options is tied to performance goals set by the Compensation Committee as follows: (i) 50% of the options would vest if the Compensation Committee determined that top-line efficacy data received from the METEOR phase 3 pivotal trial of cabozantinib in advanced RCC met its primary endpoint at a specified level, with such result to occur no later than a specified date; (ii) 25% of the options would vest if the Compensation Committee confirmed that a new drug application for cabozantinib for the treatment of advanced RCC is accepted for review by the U.S. Food and Drug Administration, or FDA, by a specified date; and (iii) 25% of the options will vest if the Compensation Committee confirms that the FDA has approved cabozantinib for the treatment of advanced RCC by a specified date. On July 20, 2015, the Committee convened to determine that top-line efficacy data received from METEOR met its primary endpoint at the level specified and within the time period permitted by the performance goals, resulting in the vesting of 50% of the option. On March 7, 2016, the Committee convened to determine that the NDA for cabozantinib for the treatment of advanced RCC was accepted for review by the FDA within the time period permitted by the performance goals, resulting in the vesting of 25% of the option. If the Compensation Committee has not concluded that FDA has approved cabozantinib for the treatment of advanced RCC by the specified date, the performance stock award with respect to that performance goal will be forfeited. |
| |
(7) | Option vests as to 1/4th of the original number of shares subject to the option on the one-year anniversary of the grant date and thereafter as to 1/48th48th of the original number of shares subject to the option on each monthly anniversary of the grant date with a final vesting date of September 16, 2019 (assuming that such options are not accelerated). |
| (6) | |
(8) | Option vests as to 1/4th4th of the original number of shares subject to the option on theone-year anniversary of the grant date and thereafter as to 1/48th of the original number of shares subject to the option on each monthly anniversary of the grant date with a final vesting date of July 15, 2019 (assuming that such options are not accelerated). |
| |
(9) | RSU vests in full on July 15, 2016, the one-year anniversary of the grant date (assuming that such RSUs are not accelerated). |
| |
(10) | RSUs vest as to 1/448th of the original number of shares subject to the RSU award on each November 15th with a final vesting date of November 15, 2016 (assuming that such RSUs are not accelerated).
|
| |
(11) | RSUs vest as to 1/4th of the original number of shares subject to the RSU award on each November 15th with a final vesting date of November 15, 2017 (assuming that such RSUs are not accelerated).
|
| |
(12) | Option vests as to 1/48th of the original number of shares subject to the option on each monthly anniversary of the grant date with a final vesting date of February 10, 201811, 2020 (assuming that such options are not accelerated). |
| (7) | Option vests as to 1/4th of the original number of shares subject to the option on theone-year anniversary of the grant date and thereafter as to 1/48th of the original number of shares subject to the option on each monthly anniversary of the grant date with a final vesting date of September 22, 2020 (assuming that such options are not accelerated). |
| (8) | Option vests as to 1/4th of the original number of shares subject to the option on theone-year anniversary of the grant date and thereafter as to 1/48th of the original number of shares subject to the option on each monthly anniversary of the grant date with a final vesting date of September 26, 2020 (assuming that such options are not accelerated). |
| (9) | RSUs vest as to 1/4th of the original number of shares subject to the RSU award on each November 15th with a final vesting date of November 15, 2020 (assuming that such RSUs are not accelerated). |
| (10) | Option vests as to 1/4th of the original number of shares subject to the option on theone-year anniversary of the grant date and thereafter as to 1/48th of the original number of shares subject to the option on each monthly anniversary of the grant date with a final vesting date of October 3, 2021 (assuming that such options are not accelerated). |
| (11) | RSUs vest as to 1/4th of the original number of shares subject to the RSU award on each November 15th with a final vesting date of November 15, 2021 (assuming that such RSUs are not accelerated). |
| (12) | Option vests as to 1/4th of the original number of shares subject to the option on theone-year anniversary of the grant date and thereafter as to 1/48th of the original number of shares subject to the option on each monthly anniversary of the grant date with a final vesting date of September 10, 2022 (assuming that such options are not accelerated). However, the option will not be exercisable until the closing market price of our common stock is equal to or greater than 125% of the exercise price of the option over a period of at least 30 consecutive calendar days. |
| (13) | PSUs vest as to (i) 50% of the original number of shares subject to the award upon the Compensation Committee’s certification that Exelixis has achieved a quarterly net product revenue target, and (ii) 50% of the original number of shares subject to the award on the first quarterly vesting date (i.e. February 15th, May 15th, August 15th and November 15th) following theone-year anniversary of the Compensation Committee’s certification |
2019 Proxy Statement 65
| (assuming that such PSUs are not accelerated). Failure to achieve the aforementioned performance target by December 31, 2021 will result in forfeiture of 100% of the PSU award. |
| (14) | PSUs vest as to (i) 50% of the original number of shares subject to the award upon the Compensation Committee’s certification that Exelixis has achieved a net product revenue target over four consecutive fiscal quarters, and (ii) 50% of the original number of shares subject to the award on the first quarterly vesting date (i.e. February 15th, May 15th, August 15th and November 15th) following theone-year anniversary of the Compensation Committee’s certification (assuming that such PSUs are not accelerated). Failure to achieve the aforementioned performance target by December 31, 2021 will result in forfeiture of 100% of the PSU award. |
| (15) | PSUs vest as to (i) 50% of the original number of shares subject to the award upon the Compensation Committee’s certification that Exelixis has initiated a target number of potentially label-enabling clinical trials for cabozantinib (both alone or in combination with another therapy), and (ii) 50% of the original number of shares subject to the award on the first quarterly vesting date (i.e. February 15th, May 15th, August 15th and November 15th) following theone-year anniversary of the Compensation Committee’s certification (assuming that such PSUs are not accelerated). Failure to achieve the aforementioned performance target by December 31, 2021 will result in forfeiture of 100% of the PSU award. |
| (16) | PSUs vest as to (i) 50% of the original number of shares subject to the award upon the Compensation Committee’s certification that Exelixis has achieved either (x) full target enrollment, as specified in the applicable clinical trial protocol, or (y) positivetop-line results, in each case with respect to a target number of potentially label-enabling clinical trials for cabozantinib (both alone or in combination with another therapy), and (ii) 50% of the original number of shares subject to the award on the first quarterly vesting date (i.e. February 15th, May 15th, August 15th and November 15th) following theone-year anniversary of the Compensation Committee’s certification (assuming that such PSUs are not accelerated). Failure to achieve the aforementioned performance target by December 31, 2021 will result in forfeiture of 100% of the PSU award. |
| (17) | PSUs vest as to (i) 50% of the original number of shares subject to the award upon the Compensation Committee’s certification that a target number of IND applications or equivalent filings with foreign governmental authorities have been approved or otherwise become effective with respect to product candidates that have either (x) been discovered by Exelixis or(y) in-licensed or acquired by Exelixis, and (ii) 50% of the original number of shares subject to the award on the first quarterly vesting date (i.e. February 15th, May 15th, August 15th and November 15th) following theone-year anniversary of the Compensation Committee’s certification (assuming that such PSUs are not accelerated). Failure to achieve the aforementioned performance target by December 31, 2021 will result in forfeiture of 100% of the PSU award. |
| (18) | Option vests as to 1/4th of the original number of shares subject to the option on theone-year anniversary of the grant date and thereafter as to 1/48th of the original number of shares subject to the option on each monthly anniversary of the grant date with a final vesting date of September 10, 2022 (assuming that such options are not accelerated). |
Option Exercises and Stock Vested
The following table includes certain information with respect to stock options exercised and stock awards that vested during the fiscal year ended January 1, 2016.
December 28, 2018.Options Exercised and Stock Vested in Fiscal 2015
|
| | | | | | | | | | | |
| | | Option Awards | Stock Awards |
| Name | | Number of Shares Acquired on Exercise(#) | | Value Realized on Exercise($)(1) | Number of Shares Acquired on Vesting(#) | | Value Realized on Vesting($)(2) |
| Michael M. Morrissey, Ph.D. | | — |
| | — |
| — |
| | — |
|
| Christopher J. Senner | | — |
| | — |
| — |
| | — |
|
| Deborah Burke | | 118,875 |
| | 227,660 |
| 6,438 |
| | 34,765 |
|
| Jeffrey J. Hessekiel | | — |
| | — |
| — |
| | — |
|
| Peter Lamb, Ph.D. | | 352,425 |
| | 71,614 |
| 2,038 |
| | 11,248 |
|
| Gisela M. Schwab, M.D. | | — |
| | — |
| 3,125 |
| | 16,875 |
|
2018
| | | | | | | | | | | | | | | | |
| | | Option Awards | | | Stock Awards | |
Name | | Number of
Shares
Acquired on
Exercise(#) | | | Value Realized on
Exercise($)(1) | | | Number of Shares Acquired on Vesting(#) | | | Value Realized on Vesting($)(2) | |
Michael M. Morrissey, Ph.D. | | | 759,267 | | | | 11,992,460 | | | | 35,000 | | | | 610,750 | |
Gisela M. Schwab, M.D. | | | 221,883 | | | | 3,235,255 | | | | 23,750 | | | | 414,438 | |
Christopher J. Senner | | | 0 | | | | 0 | | | | 20,625 | | | | 359,906 | |
Jeffrey J. Hessekiel | | | 25,000 | | | | 491,000 | | | | 16,563 | | | | 289,024 | |
Peter Lamb, Ph.D. | | | 0 | | | | 0 | | | | 17,813 | | | | 310,837 | |
| |
| (1) | "“Value Realized on Exercise"Exercise” reflects the closing market price at whichof our common stock on the applicable exercise date, net of the applicable exercise price, multiplied by the number of shares acquired upon exercise of the stock options were sold, net of the exercise price for acquiring the shares.options. |
| |
| (2) | “Value Realized on Vesting” reflects the product of the fairclosing market valueprice of our common stock on the applicable quarterly vesting date, multiplied by the number of units vested and does not necessarily reflect proceeds actually received by the Named Executive Officers.vested. |
Potential Payments Upon Termination or Change-in-Control
Change in ControlChange in Control and Severance Benefit Plan
In December 2005, theThe Board, upon recommendation of the Compensation Committee, has adopted a Change in Control and Severance Benefit Plan that provides for certain severance benefits to our officers in connection with specified termination events. Eligible plan
66 Exelixis, Inc.
Compensation of Executive Officers | Potential Payments Upon Termination or Change in Control
participants may include any employee having a rank of vice president or above, which includes the Named Executive Officers. We amended our Change in Control and Severance Benefit Plan in December 2008 and again in December 2010 to bring the plan into compliance with Section 409A of the Code and other rules governing such plans.
If a Named Executive Officer’s employment terminates due to an involuntary termination without cause or a constructive termination or Covered Termination,(Covered Termination) during a period starting one month prior to and ending 13 months following a change in control or a Change(Change in Control Termination,Termination), then the Named Executive Officer would be entitled to the following benefits under the plan:
a cash payment paid in installments pursuant to our regularly scheduled payroll periods equal to the sum of the Named Executive Officer’s base salary and target bonus for (i) 18 months for Named Executive Officers (other than the Chief Executive Officer) and (ii) 24 months for the Chief Executive Officer;
the vesting of up to all of the Named Executive Officer’s options and RSUs will accelerate in full and the exercise period of such options will be extended to the later of (i) 12 months after the change in control and (ii) the post-termination exercise period provided for in the applicable option agreement; the plan also provides that any reacquisition or repurchase rights held by us in respect of common stock issued or issuable pursuant to any stock awards granted under our 2000 Equity Plan, 2011 Equity Plan and 2014 Equity Plan shall lapse;
payment of COBRA premiums, or the cash equivalent thereof, for any health, dental or vision plan sponsored by Exelixis for a period of up to (i) 18 months for Named Executive Officers (other than the Chief Executive Officer) and (ii) 24 months for the Chief Executive Officer; and
payment of outplacement services for (i) 18 months for Named Executive Officers (other than the Chief Executive Officer), subject to a $30,000 limit and (ii) 24 months for the Chief Executive Officer, subject to a $50,000 limit.
| ›› | a cash payment paid in installments pursuant to our regularly scheduled payroll periods equal to the sum of the Named Executive Officer’s base salary and target bonus for (i) 18 months for Named Executive Officers (other than the Chief Executive Officer) and (ii) 24 months for the Chief Executive Officer; |
| ›› | the vesting of up to all of the Named Executive Officer’s options, RSUs and PSUs will accelerate in full, and the exercise period of such options will be extended to the later of: (i) 12 months after the later of (x) the participant’s termination date, or (y) the change in control; and (ii) the post-termination exercise period provided for in the applicable option agreement; the plan also provides that any reacquisition or repurchase rights held by us in respect of common stock issued or issuable pursuant to any stock awards granted under any of our equity incentive plans shall lapse; |
| ›› | payment of COBRA premiums, or the cash equivalent thereof, for any health, dental or vision plan sponsored by Exelixis for a period of up to (i) 18 months for Named Executive Officers (other than the Chief Executive Officer) and (ii) 24 months for the Chief Executive Officer; and |
| ›› | payment of outplacement services for (i) 18 months for Named Executive Officers (other than the Chief Executive Officer), subject to a $30,000 limit and (ii) 24 months for the Chief Executive Officer, subject to a $50,000 limit. |
In the event of a Covered Termination of a Named Executive Officer that is not also a Change in Control Termination, such Named Executive Officer would be entitled to receive a cash severance benefit under the plan equal to six12 months of base salary paid in installments pursuant to our regularly scheduled payroll periods. In such
circumstances, we would also pay for a period of up to six12 months of such Named Executive Officer’s COBRA premiums, or the cash equivalent thereof, for any health, dental or vision plan that we sponsored and that the Named Executive Officer is enrolled in. However, such Named Executive Officer would not be entitled to any vesting acceleration benefits by virtue of such termination.
The payments and benefits described above are subject to certain reductions and offsets if, for example, the Named Executive Officer received other severance benefits from us pursuant to a written employment agreement. In addition, if any of the severance benefits payable under the plan would constitute a “parachute payment” subject to the excise tax imposed by Section 4999 of the Code, a Named Executive Officer may receive a reduced amount of the affected severance benefits,benefits. The plan does not provide for the gross up of any excise taxes imposed by Section 4999 of the Code. No Named Executive Officer would receive benefits under the plan if (i) the Named Executive Officer has entered into an individually negotiated employment agreement that provides for severance or change in control benefits, (ii) the Named Executive Officer voluntarily terminates employment with us to accept employment with another entity that is controlled by us or is otherwise affiliated with us or (iii) the Named Executive Officer does not confirm in writing that he or she is subject to agreements with us relating to proprietary and confidential information. In addition, as a general matter, to be eligible to receive benefits under the plan and if requested by Exelixis, a Named Executive Officer must execute a general waiver and release of claims, and such release must become effective in accordance with its terms.
A Named Executive Officer’s right to receive payment of benefits under the plan will immediately terminate if, at any time prior to or during the period the Named Executive Officer is receiving such benefits, the Named Executive Officer, without the prior written approval of Exelixis, (i) willfully breaches a material provision of a proprietary and confidential information agreement with Exelixis or (ii) willfully encourages or solicits any of Exelixis’ then current employees to leave Exelixis’ employ.Treatment of Equity Awards
Pursuant to our 2000 Equity Plan, 2011 EquityPlan, 2014 Plan and 2014 Equity2017 Plan, in the event of an asset sale, merger or consolidation in which we are not the surviving corporation, or a reverse merger in which we are the surviving corporation but our common stock is converted by virtue of the merger into other property, then any surviving or acquiring corporation may assume outstanding stock awards or substitute similar stock awards for those under the plan. If any surviving or acquiring corporation refuses to assume such outstanding stock awards or substitute similar stock awards, stock awards held by participants whose service has not terminated will be accelerated in full. In addition, if any person, entity or group acquires beneficial ownership of more than 50% of our combined voting power, then stock awards held by participants whose service has not terminated will be accelerated in full.
2019 Proxy Statement 67
The following table sets forth the potential severance payments and benefits under our Change in Control and Severance Benefit Plan to which a Named Executive Officer would be entitled in connection with specified termination events, as if such Named Executive Officers’ employment terminated as of January 1, 2016,December 28, 2018, the last day of our last fiscal year. In addition, the table sets forth the amounts to which such Named Executive Officers would be entitled under our equity plans either (i) in connection with a change in control transaction in which the successor corporation did not assume or substitute outstanding stock awards, or (ii) an entity or group acquired more than 50% of our combined voting power, in each case, as of January 1, 2016.December 28, 2018. There are no other agreements, arrangements or plans that entitle any of the above-mentioned Named Executive Officers to severance, perquisites or other enhanced benefits upon termination of employment, other than certain extensions of the termination date to avoid violation of registration requirements under the Securities Act of 1933, as amended, or for such Named Executive Officer’s death or disability.
The following table shows the potential payments upon termination of employment or a change in control for the Named Executive Officers. The table assumes that the triggering event took place on January 1, 2016,December 28, 2018, the last day of our 20152018 fiscal year.
Potential Payments Upon Termination or Change in Control Table
| | | | | | | | | | | | | | |
Name | | Benefit | | Change in Control and Severance
Benefit Plan | | | Equity Plans | |
| | Involuntary Termination Without Cause or Constructive Termination in Connection with a Change of Control ($)(1) | | | Involuntary Termination Without Cause or Constructive Termination Not in Connection with a Change in Control ($)(2) | | | Certain Change of Control Transactions without Termination ($)(3) | |
Michael M. Morrissey, Ph.D. | | Base Salary | | | 1,910,120 | | | | 955,060 | | | | — | |
| | Bonus | | | 1,910,120 | | | | — | | | | — | |
| | Vesting Acceleration (4) | | | 7,516,194 | | | | — | | | | 7,516,194 | |
| | COBRA Payments | | | 69,440 | | | | 34,720 | | | | — | |
| | | Outplacement Services | | | 50,000 | | | | — | | | | — | |
Benefit Total | | | | | 11,455,874 | | | | 989,780 | | | | 7,516,194 | |
Gisela M. Schwab, M.D. | | Base Salary | | | 1,020,780 | | | | 680,520 | | | | — | |
| | Bonus | | | 510,390 | | | | — | | | | — | |
| | Vesting Acceleration (4) | | | 3,821,524 | | | | — | | | | 3,821,524 | |
| | COBRA Payments | | | 58,588 | | | | 39,059 | | | | — | |
| | | Outplacement Services | | | 30,000 | | | | — | | | | — | |
Benefit Total | | | | | 5,441,282 | | | | 719,579 | | | | 3,821,524 | |
Christopher J. Senner | | Base Salary | | | 901,530 | | | | 601,020 | | | | — | |
| | Bonus | | | 405,689 | | | | — | | | | — | |
| | Vesting Acceleration (4) | | | 4,061,729 | | | | — | | | | 4,061,729 | |
| | COBRA Payments | | | 52,080 | | | | 34,720 | | | | — | |
| | | Outplacement Services | | | 30,000 | | | | — | | | | — | |
Benefit Total | | | | | 5,451,028 | | | | 635,740 | | | | 4,061,729 | |
Jeffrey J. Hessekiel, J.D. | | Base Salary | | | 804,896 | | | | 536,597 | | | | — | |
| | Bonus | | | 362,203 | | | | — | | | | — | |
| | Vesting Acceleration (4) | | | 2,321,568 | | | | — | | | | 2,321,568 | |
| | COBRA Payments | | | 32,762 | | | | 21,841 | | | | — | |
| | | Outplacement Services | | | 30,000 | | | | — | | | | — | |
Benefit Total | | | | | 3,551,429 | | | | 558,438 | | | | 2,321,568 | |
68 Exelixis, Inc.
|
| | | | | | | | | | | |
| Potential Payments Upon Termination or Change-in-Control Table |
| | | | | | | |
| Name | | Benefit | | Change in Control and Severance Benefit Plan | | Equity Plans |
Involuntary Termination Without Cause or Constructive Termination in Connection with a Change of Control ($)(1) | | Involuntary Termination Without Cause or Constructive Termination Not in Connection with a Change in Control ($)(2) | | Certain Change of Control Transactions without Termination ($)(3) |
| Michael M. Morrissey, Ph.D. | | Base Salary | | 1,600,000 |
| | 400,000 |
| | — |
|
| | | Bonus | | 960,000 |
| | — |
| | — |
|
| | | Vesting Acceleration(4) | | 3,105,107 |
| | — |
| | 3,105,107 |
|
| | | COBRA Payments | | 48,752 |
| | 12,188 |
| | — |
|
| | | Outplacement Services | | 50,000 |
| | — |
| | — |
|
| Benefit Total | | | | 5,763,859 |
| | 412,188 |
| | 3,105,107 |
|
| Christopher J. Senner | | Base Salary | | 750,000 |
| | 250,000 |
| | — |
|
| | | Bonus | | 337,500 |
| | — |
| | — |
|
| | | Vesting Acceleration(4) | | 1,257,000 |
| | — |
| | 1,257,000 |
|
| | | COBRA Payments | | 36,564 |
| | 12,188 |
| | — |
|
| | | Outplacement Services | | 30,000 |
| | — |
| | — |
|
| Benefit Total | | | | 2,411,064 |
| | 262,188 |
| | 1,257,000 |
|
| Deborah Burke | | Base Salary | | 491,400 |
| | 163,800 |
| | — |
|
| | | Bonus | | 171,990 |
| | — |
| | — |
|
| | | Vesting Acceleration(4) | | 1,117,124 |
| | — |
| | 1,117,124 |
|
| | | COBRA Payments | | 36,564 |
| | 12,188 |
| | — |
|
| | | Outplacement Services | | 30,000 |
| | — |
| | — |
|
| Benefit Total | | | | 1,847,078 |
| | 175,988 |
| | 1,117,124 |
|
| Jeffrey J. Hessekiel, J.D. | | Base Salary | | 698,625 |
| | 232,875 |
| | — |
|
| | | Bonus | | 314,381 |
| | — |
| | — |
|
| | | Vesting Acceleration(4) | | 1,162,000 |
| | — |
| | 1,162,000 |
|
| | | COBRA Payments | | 22,789 |
| | 7,596 |
| | — |
|
| | | Outplacement Services | | 30,000 |
| | — |
| | — |
|
| Benefit Total | | | | 2,227,795 |
| | 240,471 |
| | 1,162,000 |
|
| Peter Lamb, Ph.D. | | Base Salary | | 635,279 |
| | 211,760 |
| | — |
|
| | | Bonus | | 285,875 |
| | — |
| | — |
|
| | | Vesting Acceleration(4) | | 1,124,377 |
| | — |
| | 1,124,377 |
|
| | | COBRA Payments | | 43,599 |
| | 14,533 |
| | — |
|
| | | Outplacement Services | | 30,000 |
| | — |
| | — |
|
| Benefit Total | | | | 2,119,130 |
| | 226,293 |
| | 1,124,377 |
|
| Gisela M. Schwab, M.D. | | Base Salary | | 825,272 |
| | 275,091 |
| | — |
|
| | | Bonus | | 371,372 |
| | — |
| | — |
|
| | | Vesting Acceleration(4) | | 1,468,110 |
| | — |
| | 1,468,110 |
|
| | | COBRA Payments | | 43,599 |
| | 14,533 |
| | — |
|
| | | Outplacement Services | | 30,000 |
| | — |
| | — |
|
| Benefit Total | | | | 2,738,353 |
| | 289,624 |
| | 1,468,110 |
|
Compensation of Executive Officers | CEO Pay Ratio
| | | | | | | | | | | | | | |
Name | | Benefit | | Change in Control and Severance
Benefit Plan | | | Equity Plans | |
| | Involuntary Termination Without Cause or Constructive Termination in Connection with a Change of Control ($)(1) | | | Involuntary Termination Without Cause or Constructive Termination Not in Connection with a Change in Control ($)(2) | | | Certain Change of Control Transactions without Termination ($)(3) | |
Peter Lamb, Ph.D. | | Base Salary | | | 746,288 | | | | 497,525 | | | | — | |
| | Bonus | | | 335,829 | | | | — | | | | — | |
| | Vesting Acceleration (4) | | | 3,103,418 | | | | — | | | | 3,103,418 | |
| | COBRA Payments | | | 19,213 | | | | 12,809 | | | | — | |
| | | Outplacement Services | | | 30,000 | | | | — | | | | — | |
Benefit Total | | | | | 4,234,748 | | | | 510,334 | | | | 3,103,418 | |
| |
| (1) | These benefits would be payable under the Change in Control and Severance Benefit Plan if the involuntary termination without cause or constructive termination occurred during a period starting one month prior to and ending 13 months following the change in control. |
| |
| (2) | These benefits would be payable under the Change in Control and Severance Benefit Plan if the involuntary termination without cause occurred more than one month before the change in control or if the involuntary termination without cause or a constructive termination occurred more than 13 months following the change in control. |
| |
| (3) | These benefits would be payable under the 2000 Equity Plan and/or the 2011 Equity Plan and/or the 2014 EquityPlan and/or the 2017 Plan if either (i) a successor corporation does not assume outstanding stock awards in a change of control transaction or (ii) a person, entity or group acquires beneficial ownership of more than 50% of our combined voting power, and, in each case, the Named Executive Officers do not terminate employment in connection with such a transaction or event. |
| |
| (4) | Assumes that the triggering event occurred on January 1, 2016,December 28, 2018, the last day of our last fiscal year. However, due to the federal holiday on January 1, 2016, for purposes of this table, we assumed a stock price of $5.64,year, when the closing sale price per share of our common stock on December 31, 2015, the last business day of our last fiscal year.was $19.44. The amount of the vesting acceleration is determined by: (i) aggregating for all accelerated options, the amount equal to (A) the excess, if any, of $5.64$19.44 over the relevant exercise price of the option, multiplied by (B) the number of shares underlying unvested options at such exercise price as of December 31, 2015,28, 2018; and (ii) aggregating for all accelerated RSUs, the amount equal to (X) $5.64$19.44 multiplied by (Y) the number of shares underlying the unvested RSUs. There can be no assurance that a similar triggering event would produce the same or similar results as those estimated if such event occurs on any other date or at a time when our closing sale price is different. |
CEO Pay RatioCWe believe that we provide fair and equitable compensation to our employees through a combination of competitive base pay, incentives, retirement plans and other benefits. In accordance with Item 402(u) of RegulationS-K, promulgated by the Dodd Frank Act, we determined the ratio of: (a) the annual total compensation of our Chief Executive Officer; to (b) the median of the annual total compensation of all of our employees, except for our Chief Executive Officer, both calculated in accordance with the requirements of Item 402(c)(2)(x) of RegulationS-K (CEO Pay Ratio).
In order to determine the median of the annual total compensation of all our employees, we were required to identify the “median employee” of our workforce, without regard to his or her location or employment status (full-time or part-time). The applicable SEC rules require us to identify a median employee only once every three years, as long as there have been no material changes in our employee population or employee compensation arrangements that we believe would significantly impact the calculation of our CEO Pay Ratio. However, because there was a significant increase in our employee population between October 2017, when we identified our median employee to calculate our CEO Pay Ratio for fiscal 2017, and October 2018, we decided to identify a new median employee for purposes of calculating our CEO Pay Ratio for fiscal 2018.
Consistent with the process we adopted for fiscal 2017, to determine the median of the annual total compensation of all of our employees, except for our Chief Executive Officer, we used the following methodology:
| ›› | To identify our employee population, we used tax and payroll records to determine all full-time and part-time employees, excluding our Chief Executive Officer, who were employed as of October 31, 2018. |
2019 Proxy Statement OMPENSATION69
| ›› | To identify the median employee with respect to annual total compensation of all of our employees, we calculated each employee’s “target total direct compensation,” which consists of: (i) fiscal 2018 base salary (using a reasonable estimate of the hours worked and overtime actually paid during fiscal 2018 for hourly employees); (ii) target cash bonus; and (iii) the grant date fair value of any equity awards granted during fiscal 2018 (using the same methodology that we use for estimating the value of the equity awards granted to our Named Executive Officers and reported in our Summary Compensation Table). |
| ›› | In making this determination, we annualized the base salary and target cash bonus for employees who were employed by us for less than the entirety of fiscal 2018. |
Once our representative median employee was identified in the manner described above, we calculated the annual total compensation of the representative median employee using the same methodology that we used to determine the annual total compensation of our Chief Executive Officer, as reported in the Summary Compensation Table included in this Proxy Statement. For fiscal 2018, the median of the annual total compensation of our employees (other than our Chief Executive Officer) was $248,850 and the annual total compensation of our Chief Executive Officer, as reported in the Summary Compensation Table included in this Proxy Statement, was $7,977,535. Based on this information, our CEO Pay Ratio for fiscal 2018 was 32 to 1.
This CEO Pay Ratio represents our reasonable estimate calculated in a manner consistent with Item 402(u) of RegulationS-K and applicable guidance, which provide significant flexibility in how companies identify the median employee. Each company may use a different methodology and make different assumptions particular to that company. As a result, and as explained by the SEC when it adopted these rules, in considering this CEO Pay Ratio disclosure, stockholders should keep in mind that the rule was not designed to facilitate comparisons of CEO Pay Ratios among different companies, even companies within the same industry, but rather to allow stockholders to better understand and assess each particular company’s compensation practices and CEO Pay Ratio disclosures. Neither the Compensation Committee nor our management used our fiscal 2018 CEO Pay Ratio in making compensation decisions.
70 CExelixis, Inc.
Compensation Committee Interlocks and Insider Participation
OMMITTEECOMPENSATION COMMITTEE REPORT
REPORT
The material in this report is not “soliciting material,” is not deemed “filed” with the Securities and Exchange Commission and is not deemed to be incorporated by reference in any filing of Exelixis under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing.
The Compensation Committee of the Board of Directors of Exelixis, Inc., consisting solely of independent directors, has reviewed and discussed with management the Compensation Discussion and Analysis contained in this Proxy Statement and, based on this review and discussion, the Compensation Committee recommended to the Board of Directors that the Compensation Discussion and Analysis be included in this Proxy Statement and incorporated into our Annual Report on Form10-K for the year ended January 1, 2016.
December 28, 2018.Compensation Committee:
Carl B. Feldbaum
Vincent T. Marchesi
Julie A. Smith
Lance Willsey
CCOMPENSATION COMMITTEE INTERLOCKS AND INSIDER PARTICIPATIONOMPENSATION COMMITTEE INTERLOCKSAND INSIDER PARTICIPATION
During 2015,2018, the Compensation Committee comprised Drs. Cohen, Marchesi and Willsey.Willsey, Mr. Feldbaum and Ms. Smith. None of the members of the Compensation Committee during 20152018 has at any time been an officer or employee of Exelixis, except that Dr. Cohen served as our acting Chief Scientific Officer from December 1995 to April 1997, and was named an officer of one of our former subsidiaries from 2001 through March 2005, for which he did not receive any compensation. No interlocking relationship exists between the Board or Compensation Committee and the board of directors or compensation committee of any other company, nor has any interlocking relationship existed in the past.
CCERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONSERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
Indemnification Agreements
As permitted by Delaware law, our Certificate of Incorporation provides that no director will be personally liable to us or our stockholders for monetary damages for breach of fiduciary duty as a director, except for liability for:
any breach of duty of loyalty to us or our stockholders;
acts or omissions not in good faith or that involve intentional misconduct or a knowing violation of law;
unlawful payment of dividends or unlawful stock repurchases or redemptions; or
any transaction from which the director derived an improper personal benefit.
Our Bylaws provide that we will indemnify our directors and executive officers and may indemnify our other officers and employees and other agents to the fullest extent permitted by law. We believe that indemnification under our Bylaws covers at least negligence and gross negligence on the part of indemnified parties. Our Bylaws also permit us to secure insurance on behalf of any officer, director, employee or other agent for any liability arising out of his or her actions in such capacity, regardless of whether the Bylaws would permit indemnification.
We have entered into agreements to indemnify our directors and executive officers, in addition to the indemnification provided for in our Bylaws. These agreements, among other things, indemnify our directors and executive officers for certain expenses, including attorneys’ fees, judgments, fines and settlement amounts incurred by any such person in any action or proceeding, including any action by Exelixis, arising out of such person’s services as a director or executive officer with respect to Exelixis, any of our subsidiaries or any other company or enterprise to which the person provides services at our request. We believe that these provisions and agreements are necessary to attract and retain qualified persons as directors and executive officers.
Policies and Procedures with Respect to Related Party Transactions
The Board recognizes that related party transactions can present a heightened risk of potential or actual conflicts of interests. The Board adopted a written Statement of Policy with respect to transactionsRelated Person Transactions entered into with related parties. Under this policy, the Audit Committee has been tasked with responsibility to review and approve related party transactions. The policy provides that management shall present related party transactions to the Audit Committee for approval. The policy does not prevent management from entering into any related party transaction without prior approval of the Audit Committee, so long as such related party transaction is thereafter presented to the Audit Committee for ratification. If ratification is not forthcoming, then management shall make all reasonable efforts to cancel or annul such transaction.
Under the policy, a “related party” includes: any senior officer (including each executive officer or officer subject to Section 16 of the Securities Exchange Act of 1934, as amended)Act) or director of Exelixis; a person who is an immediate family member of a senior officer, director or director nominee; a security holder who is known to own of record or beneficially more than 5% percent of any class of our securities; a person who is an immediate family member of such security holder; or an entity which is owned or controlled by one of the aforementioned persons, or an entity in which one of the aforementioned persons has a substantial ownership interest in or control over such entity.
All related party transactions shall be disclosed in our applicable filings with the SEC as required under SEC rules. There were no related party transactions reportable under the SEC rules during fiscal 2018, other than as follows:
During 2018, BlackRock, Inc. (BlackRock), a global provider of investment, advisory and risk management solutions and a greater than 5% holder of our common stock, managed a portion of our cash and investments portfolio. As of December 28, 2018 and December 29, 2017, respectively, the fair value of cash and investments managed by BlackRock was $298.5 million and $141.0 million, which included $3.0 million and $1.0 million invested in the BlackRock Liquidity Money Market Fund. We incurred $0.2 million in fees for BlackRock advisory services performed during the year ended December 28, 2018.
2019 Proxy Statement 71
SECTIONSECTION 16(a) BBENEFICIAL OWNERSHIP REPORTING COMPLIANCEENEFICIAL OWNERSHIP REPORTING COMPLIANCE
Section 16(a) of the Securities Exchange Act of 1934, as amended, requires our directors and executive officers and persons who beneficially own more than ten percent of a registered class of our equity securities to file with the SEC initial reports of ownership and reports of changes in ownership of our common stock and other equity securities of Exelixis. Officers, directors and greater than ten percent stockholders are required by SEC regulation to furnish us with copies of all Section 16(a) forms they file.
To our knowledge, based solely on a review of the copies of such reports furnished to us and written representations that no other reports were required, during the fiscal year ended January 1, 2016,December 28, 2018, all Section 16(a) filing requirements applicable to our officers, directors and greater than ten percent beneficial owners were complied with.
HHOUSEHOLDING OF PROXY MATERIALSOUSEHOLDINGOF PROXY MATERIALS
The SEC has adopted rules that permit companies and intermediaries (e.g., brokers, banks and other fiduciaries) to satisfy the delivery requirements for Notices of Internet Availability of Proxy Materials or other Annual Meeting materials with respect to two or more stockholders sharing the same address by delivering a single Notice or other Annual Meeting materials addressed to those stockholders. This process, which is commonly referred to as “householding,” potentially means extra convenience for stockholders and cost savings for companies.
This year, a number of brokers with account holders who are our stockholders will be householding proxy materials. A single Notice will be delivered to multiple stockholders sharing an address unless contrary instructions have been received from the affected stockholders. Once you have received notice from your broker that they will be householding communications to your address, householding will continue until you are notified otherwise or until you revoke your consent. If, at any time, you no longer wish to participate in householding and would prefer to receive a separate Notice, please notify your broker or direct your written request to Investor Relations, Exelixis, Inc., 210 East Grand Avenue, South San Francisco,1851 Harbor Bay Parkway, Alameda, California 9408094502 or contact Exelixis, Inc., Investor Relations at(650) 837-7000. Stockholders who currently receive multiple copies of the Notice at their address and would like to request householding of their communications should contact their broker.
ANNUAL REPORT ON FORMANNUAL REPORTON FORM10-K
A copy of our Annual Report on Form10-K for the fiscal year ended January 1, 2016,December 28, 2018, including the consolidated financial statements, schedules and list of exhibits, and any particular exhibit specifically requested, is available without charge upon written request to: Investor Relations, Exelixis, Inc., 210 East Grand Avenue, South San Francisco,1851 Harbor Bay Parkway, Alameda, California 94080.
OOTHER MATTERSTHER MATTERS
The Board knows of no other matters that will be presented for consideration at the Annual Meeting. If any other matters are properly brought before the meeting, it is the intention of the persons named in the accompanying proxy to vote on such matters in accordance with their best judgment.
|
|
By Order of the Board of Directors |
|
Jeffrey J. Hessekiel |
Executive Vice President, General Counsel and Secretary |
South San Francisco, California
April 13, 2016
APPENDIX A
Exelixis, Inc.
2000 Employee Stock Purchase Plan
Adopted by Board of
Directors: January 27, 2000DirectorsJEFFREY J. HESSEKIEL
Executive Vice President and General Counsel
Alameda, California
April , 2019
72 Exelixis, Inc.
Approved by Stockholders: March 15, 2000
Amended byPRELIMINARY COPY

EXELIXIS OTE 01—Carl B. Feldbaum, Esq. 04—Vincent T. Marchesi, M.D., Ph.D. 02—Maria C. Freire, Ph.D. 05—Julie Anne Smith 03—Alan M. Garber, M.D., Ph.D. For Against Abstain For Against Abstain For Against Abstain 1 P C F Using a black ink pen, mark your votes with an X as shown in this example. Please do not write outside the designated areas. 0312EB Proposals — The Board of Directors: January 28, 2005
Amendment ApprovedDirectors recommend a vote FOR all the nominees listed and A FOR Proposals 2, 3 and 4. 2. To ratify the selection by Stockholders: April 22, 2005
Amended by Boardthe Audit Committee of Directors: February 26, 2009
Amendment Approved by Stockholders: May 13, 2009
Amended by Compensation Committee: February 11, 2016
Amendment Approved by Stockholders: [ ], 2016
Termination Date: None
(a)The purpose of the Plan is to provide a means by which Eligible Employees of the Company and certain designated Affiliates may be given an opportunity to purchase Shares of the Company.
(b)The Company, by means of the Plan, seeks to retain the services of such Employees, to secure and retain the services of new Employees and to provide incentives for such persons to exert maximum efforts for the success of the Company and its Affiliates.
(c)The Company intends that the Rights to purchase Shares granted under the Plan be considered options issued under an “employee stock purchase plan,” as that term is defined in Section 423(b) of the Code.
(a)“Affiliate”means any “parent corporation” or “subsidiary corporation” of the Company, whether now or hereafter existing, as those terms are defined in Sections 424(e) and (f), respectively, of the Code.
(b)“Board”means the Board of Directors of Ernst & Young LLP as Exelixis’ independent registered public accounting firm for the Company.
(c)“Code”meansfiscal year ending January 3, 2020. 3. To approve the United States Internal Revenue Codeproposal of 1986, as amended.
(d)“Committee”means a Committee appointedExelixis’ Board of Directors to amend Exelixis’ Amended and Restated Certificate of Incorporation to declassify the Board of Directors to provide for annual elections by the 2020 Annual Meeting of Stockholders. 1. To elect the five Class II nominees for director named in the accompanying Proxy Statement to hold office until the 2022 Annual Meeting of Stockholders For Against Abstain NOTE: Please sign exactly as name appears hereon. Joint owners should each sign. When signing as attorney, executor, administrator, corporate officer, trustee, guardian or custodian, please give full title as such. Date (mm/dd/yyyy) — Please print date below. Signature 1 — Please keep signature within the box. Signature 2 — Please keep signature within the box. B Authorized Signatures — This section must be completed for your vote to count. Please date and sign below. 4. To approve, on an advisory basis, the compensation of Exelixis’ named executive officers, as disclosed in the accompanying Proxy Statement. qIF VOTING BY MAIL, SIGN, DETACH AND RETURN THE BOTTOM PORTION IN THE ENCLOSED ENVELOPE.q 2019 Annual Meeting Proxy Card For Against Abstain 000004 MR A SAMPLE DESIGNATION (IF ANY) ADD 1 ADD 2 ADD 3 ADD 4 ADD 5 ADD 6 ENDORSEMENT_LINE SACKPACK 1234 5678 9012 345 MMMMMMMMM MMMMMMMMMMMMMMM 4 1 3 6 9 0 MR A SAMPLE (THIS AREA IS SET UP TO ACCOMMODATE 140 CHARACTERS) MR A SAMPLE AND MR A SAMPLE AND MR A SAMPLE AND MR A SAMPLE AND MR A SAMPLE AND MR A SAMPLE AND MR A SAMPLE AND MR A SAMPLE AND C 1234567890 J N T C123456789 MMMMMMMMMMMM MMMMMMM 000000000.000000 ext 000000000.000000 ext 000000000.000000 ext 000000000.000000 ext 000000000.000000 ext 000000000.000000 ext If no electronic voting, delete QR code and control # You may vote online or by phone instead of mailing this card. Online Go to www.investorvote.com/EXEL or scan the QR code — login details are located in the shaded bar below. Phone Call toll free1-800-652-VOTE (8683) within the USA, US territories and Canada Votes submitted electronically must be received by 11:59 p.m., Eastern Time, on May 21, 2019. Your vote matters – here’s how to vote! in Proposals 1

Notice of 2019 Annual Meeting of Stockholders Proxy Solicited by Board of Directors for Annual Meeting — May 22, 2019 The undersigned hereby appoints Michael M. Morrissey, Christopher J. Senner and Jeffrey J. Hessekiel, and each of them, as attorneys and proxies of the undersigned, with full power of substitution, to vote all of the shares of stock of Exelixis, Inc. that the undersigned may be entitled to vote at the Annual Meeting of Stockholders of Exelixis, Inc. to be held at the offices of Exelixis, Inc. at 1851 Harbor Bay Parkway, Alameda, CA 94502 on Wednesday, May 22, 2019, at 8:00 a.m. (local time), and at any and all postponements, continuations and adjournments thereof, with all powers that the undersigned would possess if personally present, upon and in respect of the following matters and in accordance with Section 3(c) of the Plan.
(e)“Company” means Exelixis, Inc., a Delaware corporation.
(f)“Contributions” meansfollowing instructions, with discretionary authority as to any and all other matters that may properly come before the payroll deductionsmeeting. UNLESS A CONTRARY DIRECTION IS INDICATED, THIS PROXY WILL BE VOTED “FOR” ALL NOMINEES LISTED IN PROPOSAL 1 AND “FOR” PROPOSALS 2, 3 AND 4, AS MORE SPECIFICALLY DESCRIBED IN THE PROXY STATEMENT. IF SPECIFIC INSTRUCTIONS ARE INDICATED, THIS PROXY WILL BE VOTED IN ACCORDANCE THEREWITH. (Continued and other additional payments specifically provided for in the Offering that a Participant contributes to fund the exercise of a Right. A Participant may make additional payments into his or her account only if specifically provided for in the Offering and only if the Participant has not already had the maximum permitted amount withheld during the Offering through payroll deductions.
(g)“Director”means a member of the Board.
(h)“Eligible Employee”means an Employee who meets the requirements set forth in the Offering for eligibility to participate in the Offering, provided that such Employee also meets the requirements for eligibility to participate set forth in the Plan.
(i)“Employee”means any person, including Officers and Directors, employed by the Company or an Affiliate of the Company. Neither service as a Director nor payment of a director’s fee shall be sufficient to constitute “employment” by the Company or the Affiliate.
(j)“Employee Stock Purchase Plan”means a plan that grants rights intended to be options issued under an “employee stock purchase plan,” as that term is defined in Section 423(b) of the Code.
(k)“Entity” means a corporation, partnership, limited liability company or other entity.
(l)“Exchange Act”means the United States Securities Exchange Act of 1934, as amended.
(m)“Exchange Act Person” means any natural person, Entity or “group” (within the meaning of Section 13(d) or 14(d) of the Exchange Act), except that “Exchange Act Person” will not include (i) the Company or any Subsidiary of the Company, (ii) any employee benefit plan of the Company or any Subsidiary of the Company or any trustee or other fiduciary holding securities under an employee benefit plan of the Company or any Subsidiary of the Company, (iii) an underwriter temporarily holding securities pursuant to an offering of such securities, (iv) an Entity Owned, directly or indirectly, by the stockholders of the Company in substantially the same proportions as their Ownership of stock of the Company, or (v) any natural person, Entity or “group” (within the meaning of Section 13(d) or 14(d) of the Exchange Act) that, as of May 25, 2016, is the Owner, directly or indirectly, of securities of the Company representing more than 50% of the combined voting power of the Company’s then outstanding securities.
(n)“Fair Market Value”means the value of a security, as of any date, determined as follows:
(i)If the security is listed on any established stock exchange or traded on any established market, then except as otherwise provided by the Board, the Fair Market Value of the security shall be the closing sales price for such security (or the closing bid, if no sales were reported) as quoted on such exchange or market (or the exchange or market with the greatest volume of trading in the security)marked, dated and signed, on the trading day immediately prior toother side) Exelixis, Inc. qIF VOTING BY MAIL, SIGN, DETACH AND RETURN THE BOTTOM PORTION IN THE ENCLOSED ENVELOPE.q Change of Address — Please print new address below. Comments — Please print your comments below.Non-Voting Items Important notice regarding the relevant determination date, as reported in a source the Board deems reliable.
(ii)In the absenceInternet availability of such exchange or marketproxy materials for the security, the Fair Market Value shall be determined by the Board in good faith in compliance with applicable laws and in a manner that complies with Section 409A2019 Annual Meeting of the Code.
(o)“Non-Employee Director”means a Director who either (i) is not a current Employee or Officer of the Company or its parent or subsidiary, does not receive compensation (directly or indirectly) from the Company or its parent or subsidiary for services rendered as a consultant or in any capacity other than as a Director (except for an amount as to which disclosure would not be required under Item 404(a) of Regulation S‑K promulgated pursuant to the Securities Act (“Regulation S-K”)), does not possess an interest in any other transaction as to which disclosure would be required under Item 404(a) of Regulation S-K, and is not engaged in a business relationship as to which disclosure would be required under Item 404(b) of Regulation S-K; or (ii) is otherwise considered a “non-employee director” for purposes of Rule 16b-3.
(p)“Offering”means the grant of Rights to purchase Shares under the Plan to Eligible Employees.
(q)“Offering Date” means a date selected by the Board for an Offering to commence.
(r)“Officer” meansa person who is an officer of the Company or an Affiliate within the meaning of Section 16 of the Exchange Act.
(s)“Outside Director”means a Director who either (i) is not a current employee of the Company or an “affiliated corporation” (within the meaning of the Treasury regulations promulgated under Section 162(m) of the Code), is not a former employee of the Company or an “affiliated corporation” receiving compensation for prior services (other than benefits under a tax-qualified retirement plan) during the taxable year, was not an officer of the Company or an “affiliated corporation” at any time, and is not currently receiving direct or indirect remuneration from the Company or an “affiliated corporation” for services in any capacity other than as a Director, or (ii) is otherwise considered an “outside director” for purposes of Section 162(m) of the Code.
(t)“Own,” “Owned,” “Owner,” “Ownership” A person or Entity will be deemed to “Own,” to have “Owned,” to be the “Owner” of, or to have acquired “Ownership” of securities if such person or Entity, directly or indirectly, through any contract, arrangement, understanding, relationship or otherwise, has or shares voting power, which includes the power to vote or to direct the voting, with respect to such securities.
(u)“Participant”means an Eligible Employee who holds an outstanding Right granted pursuant to the Plan or, if applicable, such other person who holds an outstanding Right granted under the Plan.
(v)“Plan”means this Exelixis, Inc. 2000 Employee Stock Purchase Plan.
(w)“Purchase Date”means one or more dates established by the Board during an Offering on which Rights granted under the Plan shall be exercised and purchases of Shares carried out in accordance with such Offering.
(x)“Right”means an option to purchase Shares granted pursuant to the Plan.
(y)“Rule 16b-3”means Rule 16b-3 of the Exchange Act or any successor to Rule 16b-3 as in effect with respect to the Company at the time discretion is being exercised regarding the Plan.
(z)“Securities Act”means the United States Securities Act of 1933, as amended.
(aa)“Share”means a share of the common stock of the Company.
(bb) “Subsidiary” means, with respect to the Company, (i) any corporation of which more than fifty percent (50%) of the outstanding capital stock having ordinary voting power to elect a majority of the board of directors of such corporation (irrespective of whether, at the time, stock of any other class or classes of such corporation will have or might have voting power by reason of the happening of any contingency) is at the time, directly or indirectly, Owned by the Company, and (ii) any partnership, limited liability company or other entity in which the Company has a direct or indirect interest (whether in the form of voting or participation in profits or capital contribution) of more than fifty percent (50%).
(a)Stockholders. The Board shall administer the Plan unless and until the Board delegates administration to a Committee, as provided in Section 3(c). Whether or not the Board has delegated administration, the Board shall have the final power to determine all questions of policy and expediency that may arise in the administration of the Plan.
(b)The Board (or the Committee) shall have the power, subject to, and within the limitations of, the express provisions of the Plan:
(i)To determine when and how Rights to purchase Shares shall be grantedProxy Statement and the provisions of each Offering of such Rights (which need not be identical).
(ii)To designate from time2018 Annual Report to time which Affiliates of the Company shall be eligible to participate in the Plan.
(iii)To construe and interpret the Plan and Rights granted under it, and to establish, amend and revoke rules and regulations for its administration. The Board, in the exercise of this power, may correct any defect, omission or inconsistency in the Plan, in a manner and to the extent it shall deem necessary or expedient to make the Plan fully effective.
(iv)To amend the Plan as provided in Section 14.
(v)Generally, to exercise such powers and to perform such acts as it deems necessary or expedient to promote the best interests of the Company and its Affiliates and to carry out the intent that the Plan be treated as an Employee Stock Purchase Plan.
(c)The Board may delegate administration of the Plan to a Committee of the Board composed of two (2) or more members, all of the members of which Committee may be, in the discretion of the Board, Non-Employee Directors and/or Outside Directors. If administration is delegated to a Committee, the Committee shall have, in connection with the administration of the Plan, the powers theretofore possessed by the Board, including the power to delegate to a subcommittee of two (2) or more Non-Employee Directors and/or Outside Directors any of the administrative powers the Committee is authorized to exercise (and references in this Plan to the Board shall thereafter be to the Committee or such a subcommittee), subject, however, to such resolutions, not inconsistent with the provisions of the Plan, as may be adopted from time to time by the Board. The Board may abolish the Committee at any time and revest in the Board the administration of the Plan.
(d)All determinations, interpretations and constructions made by the Board in good faith will not be subject to review by any person and will be final, binding and conclusive on all persons.
| |
4. | Shares Subject to the Plan. |
(a)Subject to the provisions of Section 13(a) relating to Capitalization Adjustments, the Shares that may be sold pursuant to Rights granted under the Plan shall not exceed in the aggregate 13,650,000 Shares. If any Right granted under the Plan shall for any reason terminate without having been exercised in full, the Shares not purchased under such Right shall again becomeStockholders are available for issuance under the Plan.at: http://exel-annualstockholdermeeting.com
(b)The Shares subject to the Plan may be unissued Shares or Shares that have been bought on the open market at prevailing market prices or otherwise.
| |
5. | Grant of Rights; Offering. |
(a)The Board may from time to time grant or provide for the grant of Rights to purchase Shares of the Company under the Plan to Eligible Employees in an Offering on an Offering Date or Dates selected by the Board. Each Offering shall be in such form and shall contain such terms and conditions as the Board shall deem appropriate, which shall comply with the requirements of Section 423(b)(5) of the Code that all Employees granted Rights to purchase Shares under the Plan shall have the same rights and privileges. The terms and conditions of an Offering shall be incorporated by reference into the Plan and treated as part of the Plan. The provisions of separate Offerings need not be identical, but each Offering shall include (through incorporation of the provisions of this Plan by reference in the document comprising the Offering or otherwise) the period during which the Offering shall be effective, which period shall not exceed twenty-seven (27) months beginning with the Offering Date, and the substance of the provisions contained in Sections 6 through 9, inclusive.
(b)If a Participant has more than one Right outstanding under the Plan, unless he or she otherwise indicates in agreements or notices delivered hereunder: (i) each agreement or notice delivered by that Participant will be deemed to apply to all of his or her Rights under the Plan, and (ii) an earlier-granted Right (or a Right with a lower exercise price, if two Rights have identical grant dates) will be exercised to the fullest possible extent before a later-granted Right (or a Right with a higher exercise price if two Rights have identical grant dates) will be exercised.
(c)The Board shall have the discretion to structure an Offering so that if the Fair Market Value of a Share on any Purchase Date during an Offering is less than or equal to the Fair Market Value of a Share on the Offering Date for that Offering, then (i) that Offering shall terminate immediately following the purchase of Shares on such Purchase Date, and (ii) the Participants in such terminated Offering shall be automatically enrolled in a new Offering that begins immediately after such Purchase Date.
(a)Rights may be granted only to Employees of the Company or, as the Board may designate as provided in Section 3(b), to Employees of an Affiliate.
(i)Except as provided in Section 6(b), an Employee shall not be eligible to be granted Rights under the Plan unless, on the Offering Date, such Employee has been in the employ of the Company or the Affiliate, as the case may be, for such continuous period preceding such grant as the Board may require in the Offering, but in no event shall the required period of continuous employment be equal to or greater than two (2) years.
(ii)The Board may provide in an Offering that Employees whose customary employment is twenty (20) hours or less per week shall not be eligible to participate.
(iii)The Board may provide in an Offering that Employees whose customary employment is for not more than five (5) months in any calendar year shall not be eligible to participate.
(iv)The Board may provide in an Offering that Employees who are highly compensated Employees within the meaning of Section 423(b)(4)(D) of the Code shall not be eligible to participate.
(b)The Board may provide that each person who, during the course of an Offering, first becomes an Eligible Employee will, on a date or dates specified in the Offering which coincides with the day on which such person becomes an Eligible Employee or which occurs thereafter, receive a Right under that Offering, which Right shall thereafter be deemed to be a part of that Offering. Such Right shall have the same characteristics as any Rights originally granted under that Offering, as described herein, except that:
(i)the date on which such Right is granted shall be the “Offering Date” of such Right for all purposes, including determination of the exercise price of such Right;
(ii)the period of the Offering with respect to such Right shall begin on its Offering Date and end coincident with the end of such Offering; and
(iii)the Board may provide that if such person first becomes an Eligible Employee within a specified period of time before the end of the Offering, he or she will not receive any Right under that Offering.
(c)No Employee shall be eligible for the grant of any Rights under the Plan if, immediately after any such Rights are granted, such Employee owns stock possessing five percent (5%) or more of the total combined voting power or value of all classes of stock of the Company or of any Affiliate. For purposes of this Section 6(c), the rules of Section 424(d) of the Code shall apply in determining the stock ownership of any Employee, and stock which such Employee may purchase under all outstanding rights and options shall be treated as stock owned by such Employee.
(d)An Eligible Employee may be granted Rights under the Plan only if such Rights, together with any other rights granted under all Employee Stock Purchase Plans of the Company and any Affiliates, as specified by Section 423(b)(8) of the Code, do not permit such Eligible Employee’s rights to purchase stock of the Company or any Affiliate to accrue at a rate which exceeds twenty five thousand dollars ($25,000) of the Fair Market Value of such stock (determined at the time such rights are granted, and which, with respect to the Plan, will be determined as of their respective Offering Dates) for each calendar year in which such rights are outstanding at any time.
| |
7. | Rights; Purchase Price. |
(a)On each Offering Date, each Eligible Employee, pursuant to an Offering made under the Plan, shall be granted the Right to purchase up to the number of Shares purchasable either:
(i)with a percentage designated by the Board not exceeding fifteen percent (15%) of such Employee’s Earnings (as defined by the Board in each Offering) during the period which begins on the Offering Date (or such later date as the Board determines for a particular Offering) and ends on the date stated in the Offering, which date shall be no later than the end of the Offering; or
(ii)with a maximum dollar amount designated by the Board that, as the Board determines for a particular Offering, (1) shall be withheld, in whole or in part, from such Employee’s Earnings (as defined by the Board in each Offering) during the period which begins on the Offering Date (or such later date as the Board determines for a particular Offering) and ends on the date stated in the Offering, which date shall be no later than the end of the Offering and/or (2) shall be contributed, in whole or in part, by such Employee during such period.
(b)The Board shall establish one or more Purchase Dates during an Offering on which Rights granted under the Plan shall be exercised and purchases of Shares carried out in accordance with such Offering.
(c)In connection with each Offering made under the Plan, the Board may specify (i) a maximum amount of Shares that may be purchased by any Participant pursuant to such Offering, (ii) a maximum amount of Shares that may be purchased by any Participant on any Purchase Date pursuant to such Offering, (iii) a maximum aggregate amount of Shares that may be purchased by all Participants pursuant to such Offering, and/or (iv) a maximum aggregate amount of Shares that may be purchased by all Participants on any Purchase Date pursuant to such Offering. If the aggregate purchase of Shares issuable upon exercise of Rights granted under the Offering would exceed any such maximum aggregate amount, then, in the absence of any Board action otherwise, the Board shall make a pro rata allocation of the Shares available in as nearly a uniform manner as shall be practicable and as it shall deem to be equitable.
(d) The purchase price of Shares acquired pursuant to Rights granted under the Plan shall be not less than the lesser of:
(i)an amount equal to eighty-five percent (85%) of the Fair Market Value of the Shares on the Offering Date; or
(ii)an amount equal to eighty-five percent (85%) of the Fair Market Value of the Shares on the applicable Purchase Date.
| |
8. | Participation; Withdrawal; Termination. |
(a)An Eligible Employee may become a Participant in the Plan pursuant to an Offering and may elect to authorize payroll deductions as the means of making Contributions by delivering a participation agreement to the Company within the time specified in the Offering, in such form as the Company provides. Each such agreement shall authorize Contributions of up to the maximum percentage specified by the Board of such Employee’s Earnings during the Offering (as defined in each Offering). The Contributions made for each Participant shall be credited to a bookkeeping account for such Participant under the Plan and shall be deposited with the general funds of the Company, except where applicable law requires that Contributions be deposited with a third party. To the extent provided in the Offering, a Participant may reduce (including to zero) or increase such Contributions.
To the extent provided in the Offering, a Participant may begin such Contributions on or after the beginning of the Offering. To the extent specifically provided in the Offering, in addition to or instead of making Contributions by payroll deductions, a Participant may make Contributions through payment by cash or check prior to a Purchase Date.
(b)At any time during an Offering, a Participant may cease making Contributions and withdraw from the Offering by delivering to the Company a notice of withdrawal in such form as the Company provides. Such withdrawal may be elected at any time prior to the end of the Offering except as provided by the Board in the Offering. Upon such withdrawal from the Offering by a Participant, the Company shall distribute to such Participant all of his or her accumulated Contributions (reduced to the extent, if any, such Contributions have been used to acquire Shares for the Participant) under the Offering, without interest unless otherwise specified in the Offering, and such Participant’s Right in that Offering shall be automatically terminated. A Participant’s withdrawal from an Offering will have no effect upon such Participant’s eligibility to participate in any other Offerings under the Plan but such Participant will be required to deliver a new participation agreement in order to participate in subsequent Offerings under the Plan.
(c)Rights granted pursuant to any Offering under the Plan shall terminate immediately upon cessation of any participating Employee’s employment with the Company or a designated Affiliate for any reason (subject to any post-employment participation period required by law) or other lack of eligibility. The Company shall distribute to such Employee all of his or her accumulated Contributions (reduced to the extent, if any, such Contributions have been used to acquire Shares for the Employee) under the Offering, without interest unless otherwise specified in the Offering.
(d)Rights granted under the Plan shall not be transferable by a Participant otherwise than by will or the laws of descent and distribution, or, if permitted by the Company, by a beneficiary designation as provided in Section 15 and, otherwise during his or her lifetime, shall be exercisable only by the person to whom such Rights are granted.
(e)Unless otherwise specified in an Offering, the Company will have no obligation to pay interest on Contributions.
(a)On each Purchase Date specified therefor in the relevant Offering, each Participant’s accumulated Contributions (without any increase for interest) will be applied to the purchase of Shares up to the maximum amount of Shares permitted pursuant to the terms of the Plan and the applicable Offering, at the purchase price specified in the Offering. No fractional Shares shall be issued upon the exercise of Rights granted under the Plan unless specifically provided for in the Offering.
(b)Unless otherwise specifically provided in the Offering, if any amount of accumulated Contributions remains in a Participant’s account after the purchase of Shares and such remaining amount is less than the amount required to purchase one Share on the final Purchase Date of an Offering, then such remaining amount shall be held in such Participant’s account for the purchase of Shares under the next Offering under the Plan, unless such Participant withdraws from or is not eligible to participate in such next Offering, in which case such amount shall be distributed to such Participant after the final Purchase Date without interest. If the amount of Contributions remaining in a Participant’s account after the purchase of Shares is at least equal to the amount required to purchase one Share on the final Purchase Date of an Offering, then such remaining amount shall be distributed in full to such Participant after the final Purchase Date of such Offering without interest.
(c)No Rights granted under the Plan may be exercised to any extent unless the Shares to be issued upon such exercise under the Plan (including Rights granted thereunder) are covered by an effective registration statement pursuant to the Securities Act and the Plan is in material compliance with all applicable state, foreign and other securities and other laws applicable to the Plan. If on a Purchase Date in any Offering hereunder the Shares are not so registered or the Plan is not in such compliance, no Rights granted under the Plan or any Offering shall be exercised on such Purchase Date, and the Purchase Date shall be delayed until the Shares are subject to such an effective registration statement and the Plan is in such compliance, except that the Purchase Date shall not be delayed more than twelve (12) months and the Purchase Date shall in no event be more than twenty-seven (27) months from the Offering Date. If, on the Purchase Date of any Offering hereunder, as delayed to the maximum extent permissible, the Shares are not so registered or the Plan is not in such compliance, no Rights granted under the Plan or any Offering shall be exercised and all Contributions accumulated during the Offering (reduced to the extent, if any, such Contributions have been used to acquire Shares) shall be distributed to the Participants, without interest unless otherwise specified in the Offering.
| |
10. | Covenants of the Company. |
The Company shall seek to obtain from each federal, state, foreign or other regulatory commission or agency having jurisdiction over the Plan such authority as may be required to grant Rights under the Plan and issue and sell Shares upon exercise of the Rights granted under the Plan. If, after commercially reasonable efforts, the Company is unable to obtain from any such
regulatory commission or agency the authority which counsel for the Company deems necessary for the grant of Rights under the Plan or the lawful issuance and sale of Shares under the Plan, and at a commercially reasonable cost, the Company shall be relieved from any liability for failure to grant Rights under the Plan and/or to issue and sell Shares upon exercise of such Rights.
| |
11. | Use of Proceeds from Shares. |
Proceeds from the sale of Shares pursuant to Rights granted under the Plan shall constitute general funds of the Company.
| |
12. | Rights as a Stockholder. |
A Participant shall not be deemed to be the holder of, or to have any of the rights of a holder with respect to, Shares subject to Rights granted under the Plan unless and until the Participant’s Shares acquired upon exercise of Rights under the Plan are recorded in the books of the Company.
| |
13. | Adjustments upon Changes in Securities; Corporate Transactions. |
(a)If any change is made in, or other events occur with respect to, the Shares subject to the Plan or subject to any Right, without the receipt of consideration by the Company (through merger, consolidation, reorganization, recapitalization, reincorporation, stock dividend, dividend in property other than cash, large nonrecurring cash dividend, stock split, reverse stock split, liquidating dividend, combination of shares, exchange of shares, change in corporate structure or other similar equity restructuring transaction, as that term is used in Statement of Financial Accounting Standards Board Accounting Standards Codification Topic 718 (or any successor thereto)) (a “Capitalization Adjustment”), the Plan will be appropriately adjusted in the class(es) and maximum number of Shares subject to the Plan pursuant to Section 4(a), and the outstanding Rights and Offerings will be appropriately adjusted in the class(es), number of Shares subject to, purchase price applicable to, and purchase limits of such outstanding Rights and Offerings. The Board shall make such adjustments, and its determination shall be final, binding and conclusive. Notwithstanding the foregoing, the conversion of any convertible securities of the Company shall not be treated as a Capitalization Adjustment.
(b)In the event of (i) a dissolution or liquidation of the Company, (ii) a sale, lease or other disposition of all or substantially all of the assets of the Company, (iii) a merger, consolidation or similar transaction in which the Company is not the surviving corporation, (iv) a reverse merger, consolidation or similar transaction in which the Company is the surviving corporation but the Shares outstanding immediately preceding the merger, consolidation or similar transaction are converted by virtue of the merger, consolidation or similar transaction into other property, whether in the form of securities, cash or otherwise, or (v) an acquisition by any Exchange Act Person of the beneficial ownership (within the meaning of Rule 13d-3 promulgated under the Exchange Act, or comparable successor rule) of securities of the Company representing at least fifty percent (50%) of the combined voting power entitled to vote in the election of members of the Board (each, a “Corporate Transaction”), (1) any surviving or acquiring corporation (or its parent company) shall assume Rights outstanding under the Plan or shall substitute similar rights (including a right to acquire the same consideration paid to stockholders in the Corporate Transaction) for those outstanding under the Plan, or (2) in the event any surviving or acquiring corporation (or its parent company) refuses to assume such Rights or to substitute similar rights for those outstanding under the Plan, then, as determined by the Board in its sole discretion, such Rights may continue in full force and effect or the Participants’ accumulated Contributions (exclusive of any accumulated interest which cannot be applied toward the purchase of Shares under the terms of the Offering) may be used to purchase Shares immediately prior to the Corporate Transaction under the ongoing Offering and the Participants’ Rights under the ongoing Offering thereafter terminated.
| |
14. | Amendment, Suspension or Termination of the Plan. |
(a)The Board at any time, and from time to time, may amend the Plan. However, except as provided in Section 13(a) relating to Capitalization Adjustments, stockholder approval will be required for any amendment of the Plan for which stockholder approval is required by applicable law or listing requirements, including any amendment that either (i) materially increases the number of Shares available for issuance under the Plan, (ii) materially expands the class of individuals eligible to become Participants and receive Rights, (iii) materially increases the benefits accruing to Participants under the Plan or materially reduces the price at which Shares may be purchased under the Plan, (iv) materially extends the term of the Plan, or (v) expands the types of awards available for issuance under the Plan, but in each of (i) through (v) above only to the extent stockholder approval is required by applicable law or listing requirements.
(b)The Board in its discretion may suspend or terminate the Plan at any time. Unless sooner terminated, the Plan shall terminate at the time that all of the Shares subject to the Plan’s reserve, as increased and/or adjusted from time to time, have
been issued under the terms of the Plan. No Rights may be granted under the Plan while the Plan is suspended or after it is terminated.
(c)Any benefits, privileges, entitlements and obligations under any outstanding Rights granted before an amendment, suspension or termination of the Plan shall not be materially impaired by any such amendment, suspension or termination except (i) with the consent of the person to whom such Rights were granted, (ii) as necessary to comply with any laws, listing requirements, or governmental regulations (including, without limitation, the provisions of Section 423 of the Code and the regulations and other interpretive guidance issued thereunder relating to Employee Stock Purchase Plans) including, without limitation, any such regulations or other guidance that may be issued or amended after the effective date of the Plan, or (iii) as necessary to obtain or maintain favorable tax, listing, or regulatory treatment. To be clear, the Board may amend outstanding Rights without a Participant’s consent if such amendment is necessary to ensure that the Right and/or the Plan complies with the requirements of Section 423 of the Code.
Notwithstanding anything in the Plan or any Offering to the contrary, the Board will be entitled to: (i) establish the exchange ratio applicable to amounts withheld in a currency other than U.S. dollars; (ii) permit Contributions in excess of the amount designated by a Participant in order to adjust for mistakes in the Company’s processing of properly completed Contribution elections; (iii) establish reasonable waiting and adjustment periods and/or accounting and crediting procedures to ensure that amounts applied toward the purchase of Shares for each Participant properly correspond with amounts withheld from the Participant’s Contributions; (iv) amend any outstanding Rights or clarify any ambiguities regarding the terms of any Offering to enable the Rights to qualify under and/or comply with Section 423 of the Code; and (v) establish other limitations or procedures as the Board determines in its sole discretion advisable that are consistent with the Plan. The actions of the Board pursuant to this paragraph will not be considered to alter or impair any Rights granted under an Offering as they are part of the initial terms of each Offering and the Rights granted under each Offering.
| |
15. | Designation of Beneficiary. |
(a)The Company may, but is not obligated to, permit a Participant to file a written designation of a beneficiary who is to receive any Shares and/or cash, if any, from the Participant’s account under the Plan in the event of such Participant’s death subsequent to the end of an Offering but prior to delivery to the Participant of such Shares and cash. In addition, the Company may, but is not obligated to, permit a Participant to file a written designation of a beneficiary who is to receive any cash from the Participant’s account under the Plan in the event of such Participant’s death during an Offering. The Company may, but is not obligated to, permit the Participant to change such designation of beneficiary at any time by written notice. Any such designation and/or change must be on a form approved by the Company.
(b) In the event of the death of a Participant and in the absence of a beneficiary validly designated under the Plan who is living at the time of such Participant’s death, the Company shall deliver such Shares and/or cash to the executor or administrator of the estate of the Participant, or if no such executor or administrator has been appointed (to the knowledge of the Company), the Company, in its sole discretion, may deliver such Shares and/or cash to the spouse or to any one or more dependents or relatives of the Participant, or if no spouse, dependent or relative is known to the Company, then to such other person as the Company may designate.
| |
16. | Effective Date of Plan. |
The Plan shall become effective as determined by the Board, but no Rights granted under the Plan shall be exercised unless and until the Plan has been approved by the stockholders of the Company within twelve (12) months before or after the date the Plan is adopted by the Board.
| |
17. | Miscellaneous Provisions. |
(a)The Plan and Offering do not constitute an employment contract. Nothing in the Plan or in the Offering will in any way alter the at will nature of a Participant’s employment or be deemed to create in any way whatsoever any obligation on the part of any Participant to continue in the employ of the Company or an Affiliate, or on the part of the Company or an Affiliate to continue the employment of a Participant.
(b)The provisions of the Plan will be governed by the laws of the State of California without resort to that state’s conflicts of laws rules.